Abstract
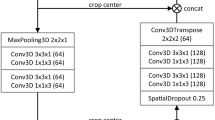
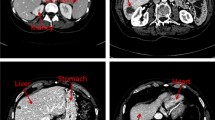
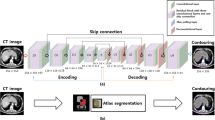
Automatic segmentation of the liver and hepatic lesions from abdominal 3D computed tomography (CT) images is fundamental tasks in computer-assisted liver surgery planning. However, due to complex backgrounds, ambiguous boundaries, heterogeneous appearances and highly varied shapes of the liver, accurate liver segmentation and tumor detection are still challenging problems. To address these difficulties, we propose an automatic segmentation framework based on 3D U-net with dense connections and globally optimized refinement. Firstly, a deep U-net architecture with dense connections is trained to learn the probability map of the liver. Then the probability map goes into the following refinement step as the initial surface and prior shape. The segmentation of liver tumor is based on the similar network architecture with the help of segmentation results of liver. In order to reduce the influence of the surrounding tissues with the similar intensity and texture behavior with the tumor region, during the training procedure, I × liverlabel is the input of the network for the segmentation of liver tumor. By doing this, the accuracy of segmentation can be improved. The proposed method is fully automatic without any user interaction. Both qualitative and quantitative results reveal that the proposed approach is efficient and accurate for liver volume estimation in clinical application. The high correlation between the automatic and manual references shows that the proposed method can be good enough to replace the time-consuming and non-reproducible manual segmentation method.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
A S Haugen, S Eirik, S K Almeland, N Sevdalis, et al. Effect of the World Health Organization Checklist on Patient Outcomes: A Stepped Wedge Cluster Randomized Controlled Trial, Annals of Surgery, 2015, 261(5): 821–828.
S M Macédo, A G Talita, J D Feltenberger, S H S Santos. The role of renin-angiotensin system modulation on treatment and prevention of liver diseases, Peptides, 2014, 62: 189–196.
L A Torre, F Bray, R L Siegel, J Ferlay, et al. Global cancer statistics, CA: A Cancer Journal for Clinicians, 2015, 65(2): 87–108.
L Ruskó, G Bekes, M Fidrich. Automatic segmentation of the liver from multi- and single-phase contrast-enhanced CT images, Medical Image Analysis, 2009, 13(6): 871–882.
R Susomboon, D Raicu, J Furst, D Channin. Automatic Single-Organ Segmentation in Computed Tomography Images, Proceedings of the 6th IEEE International Conference on Data Mining, 2006: 1081–1086.
J Y Yan, L H Schwartz, B S Zhao. Semiautomatic segmentation of liver metastases on volumetric CT images, Medical Physics, 2015, 42(11): 6283–6293.
F Liu, B S Zhao, P K Kijewski, L Wang, H Lawrence. Liver segmentation for CT images using GVF snake, Medical Physics, 2005, 32(12): 3699–3706.
C Y Xu, J L Prince. Snakes, shapes, and gradient vector flow, IEEE Transactions on Image Processing A Publication of the IEEE Signal Processing Society, 1998, 7(3): 359–369.
L Massoptier, S Casciaro. A new fully automatic and robust algorithm for fast segmentation of liver tissue and tumors from CT scans, European Radiology, 2008, 18(8): 1658–1665.
S Al-Shaikhli, M Y Yang, B Rosenhahn. Automatic 3D Liver Segmentation Using Sparse Representation of Global and Local Image Information via Level Set Formulation, Computer Science, 2015.
J K Wang, Y Z Cheng, C Y Guo, Y D Wang, S Tamura. ShapeCintensity prior level set combining probabilistic atlas and probability map constrains for automatic liver segmentation from abdominal CT images, International Journal of Computer Assisted Radiology and Surgery, 2015, 11(5): 817–826.
Y Boykov, G Funka-Lea. Graph Cuts and Efficient N-D Image Segmentation, International Journal of Computer Vision, 2006, 70(2): 109–131.
Y Boykov, M P Jolly. Interactive graph cuts for optimal boundary and region segmentation of objects in n-d images, Proceedings of the IEEE International Conference on Computer Vision, 2001: 105–112.
C Y Li, X Y Wang, S Eberl, M Fulham, et al. A Likelihood and Local Constraint Level Set Model for Liver Tumor Segmentation from CT Volumes, IEEE Transactions on Biomedical Engineering, 2013, 60(10): 2967–2977.
J L Peng, F F Dong, Y M Chen, D X Kong. A region-appearance-based adaptive variational model for 3D liver segmentation, Medical Physics, 2014, 41(4): 043502.
J L Peng, Y Wang, D X Kong. Liver segmentation with constrained convex variational model, Pattern Recognition Letters, 2014, 43(1): 81–88.
A Ben-Cohen, I Diamant, E Klang, M Amitai, H Greenspan. Fully convolutional network for liver segmentation and lesions detection, Springer International Publishing, 2016, 10008: 77–85.
P F Christ, M E A Elshaer, F Ettlinger, S Tatavarty, et al. Automatic Liver and Lesion Segmentation in CT Using Cascaded Fully Convolutional Neural Networks and 3D Conditional Random Fields, International Conference on Medical Image Computing and Computer-Assisted Intervention, Springer, Cham, 2016, 9901: 415–423.
F Lu, F Wu, P J Hu, Z Y Peng, et al. Automatic 3D liver location and segmentation via convolutional neural networks and graph cut, International Journal of Computer Assisted Radiology and Surgery, 2016, 12(2): 171–182.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by the National Natural Science Foundation of China(12090020,12090025) and Zhejiang Provincial Natural Science Foundation of China(LSD19H180005).
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the articles Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the articles Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Hong, Y., Mao, Xw., Hui, Ql. et al. Automatic liver and tumor segmentation based on deep learning and globally optimized refinement. Appl. Math. J. Chin. Univ. 36, 304–316 (2021). https://doi.org/10.1007/s11766-021-4376-3
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11766-021-4376-3