Abstract
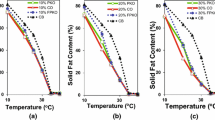
Using high-pressure homogenization to generate different droplet size distributions, the nucleation and crystallization of two fat systems [lard or a plam stearin/canola oil blend (PSCO)] were compared in bulk and emulsified form. Droplet size reduction decreased the final volume fraction of solid fat primarily in the lard system vs. the PSCO system, with a greater reduction in volume fraction using the homogenization regime that led to smaller droplets. Homogeneous and heterogeneous models showed that the nucleation rate generally decreased with a reduction in droplet size. However, the Gibbs surface energy (γ) was significantly underestimated using the homogeneous model, whereas the heterogeneous model fit the data adequately (P<0.05). The temperature sensitivity of the calculated impurity concentration in all emulsified systems was droplet size dependent. The Avrami model showed the emulsified fats to have lower Avrami indices relative to the bulk fat as well as lower crystallization rate constants. Differences in the Avrami indices and the rate constants were more pronounced in the bulk and emulsified PSCO compared with lard.
Similar content being viewed by others
References
Boistelle, R., Fundamentals of Nucleation and Crystal Growth, in Crystallization and Polymorphism of Fats and Fatty Acids, edited by N. Garti and K. Sato, Marcel Dekker, New York, 1988, pp. 189–226.
Rousset, P., Modeling Crystallization Kinetics of Triacylglycerols, in Physical Properties of Lipids, edited by A.G. Marangoni and S.S. Narine, Marcel Dekker, New York, 2002, pp. 1–35.
Hindle, S., M.J.W. Povey, and K. Smith, Kinetics of Crystallization in n-Hexadecane and Cocoa Butter Oil-in-Water Emulsions Accounting for Droplet Collision Mediated Nucleation, J. Colloid Interface Sci. 232:370–380 (2000).
Skoda, W., and M. Van den Tempel, Crystallization of Emulsified TAGs, J. Colloid Sci. 18:568–584 (1963).
Kloek, W., P. Walstra, and T. van Vliet, Nucleation Kinetics of Emulsified Triglyceride Mixtures, J. Am. Oil. Chem. Soc. 77:643–652 (2000).
Walstra, P., and E.C.H. van Beresteyn, Crystallization of Milk Fat in the Emulsified State, Neth. Milk Dairy J. 29:35–65 (1975).
Awad, T., Y. Hamada, and K. Sato, Effects of Addition of Diacylglycerols on Fat Crystallization in Oil-in-Water Emulsions, Eur. J. Lipid Sci. Technol. 103:735–741 (2001).
Kaneko, N., T. Horie, S. Ueno, J. Yano, T. Katsuragi, and K. Sato, Impurity Effects on Crystallization Rates of n-Hexadecane in Oil-in-Water Emulsions, J. Crystal Growth 197:263–270 (1999).
Hodate, Y., S. Ueno, J. Yano, T. Katsuragi, Y. Tezuka, T. Tagawa, N. Yoshimoto, and K. Sato, Ultrasonic Velocity Measurement of Crystallization of Palm Oil in Oil-in-Water Emulsions, Colloids Surf. A. 128:217–224 (1997).
Campbell, S.D., H.D. Goff, and D. Rousseau, Comparison of Crystallization Properties of a Palm Stearin/Canola Oil Blend and Lard in Bulk and Emulsified Form, Food Res. Int. 35:935–944 (2002).
Official Methods and Recommended Practices of the AOCS, 5th edn., AOCS Press, Champaign, 1997.
Kashchiev, D., N. Kaneko, and K. Sato, Kinetics of Crystallization in Polydisperse Emulsions, J. Colloid Interface Sci. 208:167–177 (1998).
Kashchiev, D., D. Clausse, and C. Jolivet-Dalmazzone. Crystallization and Critical Supercooling of Disperse Liquids, 165:148–153 (1994).
Avrami, M., Kinetics of Phase Change I. General Theory, J. Chem. Phys. 7:1103–1112 (1939).
Avrami, M., Kinetics of Phase Change II. Transformation-Time Relations for Random Distribution of Nuclei, 8:212–224 (1940).
Sharples, A., Overall Kinetics of Crystallization, in Introduction to Polymer Crystallization, Edward Arnold, London, 1966, pp. 44–59.
Kashchiev, D., Nucleation: Basic Theory with Applications, Butterworth-Heinemann, Oxford, 2000, pp. 366–368.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Campbell, S.D., Douglas Goff, H. & Rousseau, D. Modeling the nucleation and crystallization kinetics of a palm stearin/canola oil blend and lard in bulk and emulsified form. J Amer Oil Chem Soc 81, 213–219 (2004). https://doi.org/10.1007/s11746-004-0884-9
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11746-004-0884-9