Abstract
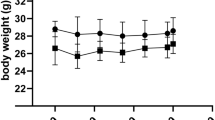
The aim of this study was to compare two different strategies to elevate brain, retina, liver, and heart docosahexaenoic acid (DHA) levels in guinea pigs. Fist, we used an increasing dose of α-linolenic acid (AIA) relative to a constant linoleic acid (LA) intake, and second, we used two levels of dietary DHA provided in conjunction with dietary arachidonic acid (AA). The percentage DHA and AA of total phospholipids in retina, liver, and heart, and in the brain phosphotidylethanolamine and phosphatidylcholine was studied in female pigmented guinea pigs (3 wk old) fed one of five semisynthetic diets containing 10% (w/w) lipid for 12 wk. The LA content in the diets was constant (17% of total fatty acids), with the ALA content varying from 0.05% (diet SFO), to 1% (diet Mix), and to 7% (diet CNO). Two other diets LCP) and LCP3) had a constant LA/ALA ratio (17.5∶1) but varied in the levels of dietary AA and DHA supplementation. Diet LCP1 was structured to closely replicate the principal long chain polyunsaturated fatty acids (PUFA) found in human breast milk and contained 0.9% AA and 0.6% DHA (% of total fatty acids) whereas diet LCP3 contained 2.7% AA and 1.8% DHA. At the end of the study, animals were sacrificed and tissues taken for fatty acid analyses. We found no significant effects of diets on the growth of guinea pigs. Diets containing ALA has profoundly different effects on tissue fatty acid compositions compared with diets which contained the long chain PUFA (DHA and AA). In the retina and brain phospholipids, high-ALA diets or dietary DHA supplementation produced moderate relative increases in DHA levels. There was no change in retinal or brain AA proportions following dietary AA supplementation, even at the highest level. This was in contrast to liver and heart where tissue DHA proportions were low and AA predominated. In these latter tissues, dietary ALA had little effect on tissue DHA proportions although the proportion of AA was slightly depressed at the highest dietary ALA intake, but dietary DHA and AA supplements led to large increases (up to 10-fold) in the proportions of these PUFA. Tissue uptake of dietary AA and DHA appeared maximal for the LCP1 diet (replicate of breast milk) in the heart. There were no significant changes in the plasma levels of 11-dehydrothromboxane B2 (a thromboxane A2 metabolite), for any diet. The data confirm that dietary ALA is less effective than dietary DHA supplementation (on a gram/gram basis) in increasing tissue DHA levels and that tissues vary greatly in their response to exogenous AA and DHA, with the levels of these long chain metabolites being most resistant to change in the retina and brain compared with liver and heart. Dietary DHA markedly increased tissue DHA proportions in both liver and heart, whereas the major effect of dietary AA was in the liver. Future studies of the effects of dietary DHA and AA supplementation should examine a variety of tissues rather than focusing only on neural tissue.
Similar content being viewed by others
Abbreviations
- ALA:
-
α-linolenic acid
- AA:
-
arachidonic acid
- DHA:
-
docosahexaenoic acid 22∶6n−3
- LA:
-
linoleic acid
- PUFA:
-
polyunsaturated fatty acids
References
Crawford, M.A., Costeloe, K., Ghebremeskel, K., and Phylactos, A. (1998) The Inadequacy of the Essential Fatty Acid Content of Present Preterm Feeds, Eur. J. Pediatr. 157S, 23–27.
Jensen, C.L., Chen, H., Fraley, J.K., Anderson, R.E., and Heird, W.C. (1996) Biochemical Effects of Dietary Linoleic/α-Linolenic Acid Ratio in Term Infants, Lipids 31, 107–113.
Carlson, S.E., Werkman, S.H., and Tolley, E.A. (1996) The Effect of Long Chain n−3 Fatty Acid Supplementation on Visual Acuity and Growth of Preterm Infants With and Without Bronchopulmonary Dysplasia, Am. J. Clin. Nutr. 63, 687–697.
Birch, E.E., Hoffman, D.R., Uauy, R., Birch, D.G., and Prestidge, C. (1998) Visual Acuity and the Essentiality of Docosahexaenoic Acid and Arachidonic Acid in the Diet of Term Infants, Pediatr. Res. 44, 201–209.
Willatts, P., Forsyth, L.S., Dimodugno, M.K., Varma, S., and Colvin, M. (1998) Effect of Long Chain Polyunsaturated Fatty Acids in Infant Formula on Problem Solving at 10 Months of Age, Lancet, 352, 688–691.
Farquharson, J., Jamieson, E.C., Abbasi, K.A., Patrick, W.J.A., Logan, R.W., and Cockburn, F. (1995) Effect of Diet on the Fatty Acid Composition of the Major Phospholipids of Infant Cerebral Cortex, Arch. Dis. Child. 72, 198–203.
Makrides, M., Neumann, M., Byard, R.W. Simmer, K., and Gibson, R.A. (1994) Fatty Acid Composition of Brain, Retina and Erythrocytes in Breast- and Formula-Fed Infants, Am. J. Clin. Nutr. 60, 189–194.
Neuringer, M., Connor, W.E., Lin, D.S., Barstad, L., and Luck, S. (1986) Biochemical and Functional Effects of Prenatal and Postnatal Omega-3 Fatty Acid Deficiency on Retina and Brain in Rhesus Monkeys, Proc. Natl. Acad. Sci. USA 83, 4021–4025.
Benolken, R.M., Anderson, R.E., and Wheeler, T.G. (1973) Membrane Fatty Acids Associated with the Electrical Response in Visual Excitation, Science 182, 1253–1254.
Weisinger, H.S., Vingrys, A.J., and Sinclair, A.J. (1996) The Effect of Docosahexaenoic Acid on the Electroretinogram of the Guinea Pig, Lipids, 31, 65–70.
Clark, K.J., Makrides, M., Neumann, M.A., and Gibson, R.A. (1992) Determination of the Optimal Ratio of Linoleic Acid to α-Linolenic Acid in Infant Formulas, J. Pediatr. 120(S), 151–158.
Makrides, M., and Gibson, R.A. (1996) Defining the LCPUFA Requirement of Term Infants. in Recent Developments in Infant Nutrition, (Bindel, J.G., Goedhart, A.C., and Visser, H.K.A., eds.) pp. 202–211, Kluwer Academic Publishers, Great Britain.
Clandinin, M.T., Van Aerde, J.E., Parrott, A., Field, C.J., Euler, A.R., and Lien, E.L. (1997) Assessment of the Efficacious Dose of Arachidonic and Docosahexaenoic Acids in Preterm Infant Formutas: Fatty Acid Composition of Erythrocyte Membrane Lipids, Pediatr. Res. 42, 819–825.
Boyle, F.G., Yuhas, R.J., Goldberg, K., and Lien, E.L. (1998) Interaction of n−3 Long-Chain Polyunsaturated Fatty Acids with n−6 Fatty Acids in Suckled Rat Pups, Lipids 33, 243–250.
Sinclair, A.J., O'Dea, K., Dunstan, G., Ireland, P.D., and Niall, M. (1987) Effects on Plasma Lipids and Fatty Acid Composition of Very Low Fat Diets Enriched with Fish or Kangaroo Meat, Lipids 22, 523–529.
Fine, J.B., and Sprecher, H. (1982) Unidimensional Thin-Layer Chromatography of Phospholipids on Boric Acid-Impregnated Plates, J. Lipid Res. 23, 660–663.
Anding, R.H., and Hwang, D.H. (1986) Effects of Dietary Linolenate on the Fatty Acid Composition of Brain Lipids in Rats, Lipids, 21, 697–701.
Craig-Schmidt, M.C., and Huang, M.C. (1998) Interaction of n−6 and n−3 Fatty Acids: Implications for Supplementation of Infant Formula with Long Chain Polyunsaturated Fatty Acids, in Lipids In Infunt Nutrition, (Huang, Y.S., and Sinclair, A.J. eds.) pp. 63–84, AOCS Press, Champaign.
Litman, B.J., and Mitchell, D.C. (1996) A Role for Phospholipids in Modulating Membrane Protein Function, Lipids, 31, S193-S198.
Poling, J.S., Karanian, J.W., Salem, N., and Vicini, S. (1993) Time- and Voltage-Dependent Block of Delayed Rectifier Potassium Channels by Docosahexaenoic Acid, Mol. Pharmacol. 47, 381–390.
Craig-Schmidt, M.C., Stieh, K.E., and Lien, E.L. (1996) Retinal Fatty Acids of Piglets Fed Docosahexaenoic and Arachidonic Acids from Microbial Sources, Lipids 31, 53–59.
Woods, J., Ward, G., and Salem, N. (1996) Is Docosahexaenoic Acid Necessary in Infant Formula? Evaluation of High Linolenate Diets in the Neonatal Rat, Pediatr. Res. 40, 687–694.
Connor, W.E., and Neuringer, M. (1988) The Effects of n−3 Fatty Acid Deficency and Repletion upon the Fatty Acid Composition and Function of Brain and Retina Prog. Clin. Biol. Res. 282, 275–295.
Arbuckle, L.D., Rioux, F.M., Mackinnon, M.J., and Innis, S.M. (1992) Formula α-Linolenic (18∶3n−3) and Linoleic (18∶2n−6) Acid Influence Neonatal Piglet Liver and Brain Saturated Fatty Acids, as Welt as Docosahexaenoic Acid (22∶6n−3), Biochim. Biophys. Acta 1125, 262–267.
Su, M.-H., Keswick, L.A., and Brenna, J.T. (1996) Increasing Dietary Linoleic Acid in Young Rat Increases and Then Decreases Docosahexaenoic Acid in Retina but Not in Brain, Lipids 31, 1289–1298.
Sinclair, A.J. (1975) The Incorporation of Radioactive Polyunsaturated Fatty Acids into Liver and Brain of Developing Rat, Lipips 10, 175–184.
Guesnet, P., Alasnier, C., Alessandri, J.M., and Durand, G. (1997) Modifying the n−3 Fatty Acid Content of the Maternal Dietto Determine the Requirement of the Fetal and Suckling Rat, Lipids 32, 527–534.
Winters, B.L., Yeh, S.M., and Yeh, Y.Y. (1994) Linolenic Acid Provides a Source of Docosahexaenoic Acid for Artificially Reared Rat Pups, J. Nutr. 124, 1654–1659.
Bourre, J.M., Dumont, O.L., Piciotti, M.J., Clement, M.E., and Durand, G.A. (1997) Comparison of Vegetable and Fish Oil in the Provision of n−3 Polyunsaturated Fatty Acids for Nervous Tissue and Selected Organs, J. Nutr. Biochem. 8, 472–478.
Horrobin, D.F., Huang, Y.S., Cunnane, S.C., and Manku, M.S. (1984) Essential Fatty Acids in Plasma, Red Blood Cells and Liver Phospholipids in Common Laboratory Animals as Compared to Humans, Lipids 19, 806–811.
Willis, A.L. (1981) Unanswered Questions in Essential Fatty Acid and Prostaglandin Research, Prog. Lipid Res. 20, 839–850.
Takasaki, W., Nakagawa, A., Asai, F., Ushiyama, S., Sugidachi, A., Matsuda, K., Oshima, T., and Tanaka, Y. (1993) Superiority of Plasma 11-Dehydrothromboxane B2 to TXB2 as an Index of in vivo TX Formation in Rabbits After Dosing with CS-518, a TX Synthase Inhibitor, Thromb. Res. 71, 69–76.
Dobbing, J., and Sands, J. (1970) Growth and Development of the Brain and Spinal Cord of the Guinea Pig, Brain Res. 17, 115–123.
Huang, J., Wyse, J.P., and Spira, A.W. (1990) Ontogenesis of the Electroretinogram in a Precocial Mammal, the Guinea Pig (Cavia porcellus), Comp. Biochem. Physiol. 95, 149–155.
Graur, D., Hide, W.A., and Hsiun, W. (1991) Is the Guinea-Pig a Rodent? Nature, 351, 649–652.
Li, W.H., Hide, W.A., Zharkikh, A., Ma, D.P., and Graur, D. (1992) The Molecular Taxonomy and Evolution of the Guinea Pig, J. Hered. 83, 174–181.
Emken E.A., Adlof, R.O., Duval, S.M., and Nelson, G.J. (1998) Effect of Dietary Arachidonic Acid on Metabolism of Deuterated Linoleic Acid by Adult Male Subjects, Lipids 33, 471–480.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Abedin, L., Lien, E.L., Vingrys, A.J. et al. The effects of dietary α-linolenic acid compared with docosahexaenoic acid on brain, retina, liver, and heart in the guinea pig. Lipids 34, 475–482 (1999). https://doi.org/10.1007/s11745-999-0387-3
Received:
Issue Date:
DOI: https://doi.org/10.1007/s11745-999-0387-3