Abstract
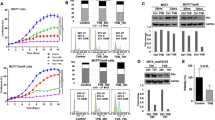
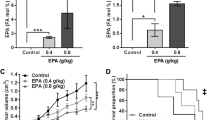
Docosahexanoic acid (DHA) and eicosapentanoic acid (EPA) have been shown to possess anti-carcinogenic properties in mammary cancers, both in vitro and in vivo. The objective of this study was to investigate the effect of treating three different breast cancer cell lines with DHA or EPA on cellular growth, chemotherapy efficacy, and CD95 expression and localization in the cell. MDA-MB-231, MCF-7 and SKBr-3 cells were incubated with EPA or DHA with or without chemotherapy agents [doxorubicin (dox), Herceptin]. Cell growth was assessed by WST-1 assay and CD95 expression was investigated using flow cytometry, Western blotting and confocal microscopy. DHA and EPA inhibited the growth of all three breast cancer cell lines in a dose-dependent fashion (P < 0.05). DHA, and to a lesser extent EPA, induced the movement and raft clustering of CD95 in the cell membrane (via confocal microscopy) and the surface expression (via flow cytometry) in MDA-MB-231 cells. Neither fatty acid altered the growth/metabolic activity of the non-transformed MCF-12A breast cell line. Pre-treatment with DHA, but not EPA, improved the efficacy of dox in estrogen receptor negative MDA-MB-231 cells (P < 0.05), but not in the other two cell lines. Pre-treating cells with DHA increased CD95 surface expression (threefold) and the plasma membrane raft content of CD95 (2fold) and FADD (>4-fold) after dox treatment, compared to dox treatment alone (P < 0.05). This study demonstrated that pre-treatment of estrogen receptor negative MDA-MB-231 cells with DHA increased the anti-cancer effects of dox and presents evidence to suggest that this may be mediated in part by CD95-induced apoptosis.




Similar content being viewed by others
Abbreviations
- DAPI:
-
4′,6′-Diamidino-2-phenylindole
- DHA:
-
Docosahexanoic acid
- DISC:
-
Death-inducing signaling complex
- EPA:
-
Eicosapentanoic acid
- ER:
-
Estrogen receptor
- FADD:
-
Fas-associated death domain-containing protein
- LNA:
-
Linoleic acid
- OLA:
-
Oleic acid
- PUFA:
-
Polyunsaturated fatty acid(s)
References
Bougnoux P, Maillard V, Chajes V (2005) Omega-6/omega-3 polyunsaturated fatty acids ratio and breast cancer. World Rev Nutr Diet 94:158–165
Karmali RA, Marsh J, Fuchs C (1984) Effect of omega-3 fatty acids on growth of a rat mammary tumor. J Natl Cancer Inst 73:457–461
Rose DP, Connolly JM (1999) Omega-3 fatty acids as cancer chemopreventive agents. Pharmacol Ther 83:217–244
Schley PD, Jijon HB, Robinson LE, Field CJ (2005) Mechanisms of omega-3 fatty acid-induced growth inhibition in MDA-MB-231 human breast cancer cells. Breast Cancer Res Treat 92:187–195
Schley PD, Brindley DN, Field CJ (2007) (n-3) PUFA alter raft lipid composition and decrease epidermal growth factor receptor levels in lipid rafts of human breast cancer cells. J Nutr 137:548–553
Bernard-Gallon DJ, Vissac-Sabatier C, Antoine-Vincent D, Rio PG, Maurizis J-C, Fustier P, Bignon Y-J (2002) Differential effects of n-3 and n-6 polyunsaturated fatty acids on BRCA1 and BRCA2 gene expression in breast cell lines. Br J Nutr 87:281–289
Baumgartner M, Sturlan S, Roth E, Wessner B, Bachleitner-Hofmann T (2004) Enhancement of arsenic trioxide-mediated apoptosis using docosahexaenoic acid in arsenic trioxide-resistant solid tumor cells. Int J Cancer 112:707–712
Menendez JA, Ropero S, Mehmi I, Atlas E, Colomer R, Lupu R (2004) Overexpression and hyperactivity of breast cancer-associated fatty acid synthase (oncogenic antigen-519) is insensitive to normal arachidonic fatty acid-induced suppression in lipogenic tissues but it is selectively inhibited by tumoricidal alpha-linolenic and gamma-linolenic fatty acids: a novel mechanism by which dietary fat can alter mammary tumorigenesis. Int J Oncol 24:1369–1383
Biondo PD, Brindley DN, Sawyer MB, Field CJ (2008) The potential for treatment with dietary long-chain polyunsaturated n-3 fatty acids during chemotherapy. J Nutr Biochem 19:787–796
Grammatikos SI, Subbaiah PV, Victor TA, Miller WM (1994) n-3 and n-6 fatty acid processing and growth effects in neoplastic and non-cancerous human mammary epithelial cell lines. Br J Cancer 70:219–227
Chajes V, Sattler W, Stranzl A, Kostner GM (1995) Influence of n-3 fatty acids on the growth of human breast cancer cells in vitro: relationship to peroxides and vitamin-E. Breast Cancer Res Treat 34:199–212
Begin ME, Ells G, Horrobin DF (1988) Polyunsaturated fatty acid-induced cytotoxicity against tumor cells and its relationship to lipid peroxidation. J Natl Cancer Inst 80:188–194
Yamamoto D, Kiyozuka Y, Adachi Y, Takada H, Hioki K, Tsubura A (1999) Synergistic action of apoptosis induced by eicosapentaenoic acid and TNP-470 on human breast cancer cells. Breast Cancer Res Treat 55:149–160
Tsujita-Kyutoku M, Yuri T, Danbara N, Senzaki H, Kiyozuka Y, Uehara N, Takada H, Hada T, Miyazawa T, Ogawa Y, Tsubura A (2004) Conjugated docosahexaenoic acid suppresses KPL-1 human breast cancer cell growth in vitro and in vivo: potential mechanisms of action. Breast Cancer Res 6:R291–R299
Simons K, Toomre D (2000) Lipid rafts and signal transduction. Nat Rev Mol Cell Biol 1:31–39
Shaw AS (2006) Lipid rafts: now you see them, now you don’t. Nat Immunol 7:1139–1142
Foster LJ, De Hoog CL, Mann M (2003) Unbiased quantitative proteomics of lipid rafts reveals high specificity for signaling factors. Proc Natl Acad Sci USA 100:5813–5818
Brown DA, London E (1998) Functions of lipid rafts in biological membranes. Annu Rev Cell Dev Biol 14:111–136
Hatala MA, Rayburn J, Rose DP (1994) Comparison of linoleic acid and eicosapentaenoic acid incorporation into human breast cancer cells. Lipids 29:831–837
Rose DP, Connolly JM, Rayburn J, Coleman M (1995) Influence of diets containing eicosapentaenoic or docosahexaenoic acid on growth and metastasis of breast cancer cell in nude mice. J Natl Cancer Inst 87:587–592
Robinson LE, Clandinin MT, Field CJ (2002) The role of dietary long-chain n-3 fatty acids in anti-cancer immune defense and R3230AC mammary tumor growth in rats: influence of diet fat composition. Breast Cancer Res Treat 73:145–160
Rockett BD, Franklin A, Harris M, Teague H, Rockett A, Shaikh SR (2011) Membrane raft organization is more sensitive to disruption by (n-3) PUFA than nonraft organization in EL4 and B cells. J Nutr 141:1041–1048
Bougnoux P, Germain E, Chajes V, Hubert B, Lhuillery C, Le FO, Body G, Calais G (1999) Cytotoxic drugs efficacy correlates with adipose tissue docosahexaenoic acid level in locally advanced breast carcinoma. Br J Cancer 79:1765–1769
Bougnoux P, Hajjaji N, Ferrasson MN, Giraudeau B, Couet C, Le FO (2009) Improving outcome of chemotherapy of metastatic breast cancer by docosahexaenoic acid: a phase II trial. Br J Cancer 101:1978–1985
Siddiqui RA, Harvey KA, Xu Z, Bammerlin EM, Walker C, Altenburg JD (2011) Docosahexaenoic acid: a natural powerful adjuvant that improves efficacy for anticancer treatment with no adverse effects. BioFactors 37:399–412
Barnhart BC, Alappat EC, Peter ME (2003) The CD95 type I/type II model. Semin Immunol 15:185–193
Fulda S, Strauss G, Meyer E, Debatin KM (2000) Functional CD95 ligand and CD95 death-inducing signaling complex in activation-induced cell death and doxorubicin-induced apoptosis in leukemic T cells. Blood 95:301–308
Bollinger CR, Teichgraber V, Gulbins E (2005) Ceramide-enriched membrane domains. Biochim Biophys Acta 1746:284–294
Kim HS, Lee YS, Kim DK (2009) Doxorubicin exerts cytotoxic effects through cell cycle arrest and Fas-mediated cell death. Pharmacology 84:300–309
Blanckaert V, Ulmann L, Mimouni V, Antol J, Brancquart L, Chenais B (2010) Docosahexaenoic acid intake decreases proliferation, increases apoptosis and decreases the invasive potential of the human breast carcinoma cell line MDA-MB-231. Int J Oncol 36:737–742
Patterson RE, Flatt SW, Newman VA, Natarajan L, Rock CL, Thomson CA, Caan BJ, Parker BA, Pierce JP (2011) Marine fatty acid intake is associated with breast cancer prognosis. J Nutr 141:201–206
Kang KS, Wang P, Yamabe N, Fukui M, Jay T, Zhu BT (2010) Docosahexaenoic acid induces apoptosis in MCF-7 cells in vitro and in vivo via reactive oxygen species formation and caspase 8 activation. PLoS One 5:e10296
Shaikh IA, Brown I, Schofield AC, Wahle KW, Heys SD (2008) Docosahexaenoic acid enhances the efficacy of docetaxel in prostate cancer cells by modulation of apoptosis: the role of genes associated with the NF-kappaB pathway. Prostate 68:1635–1646
Sawyer M, Field CJ (2010) Possible mechanisms for omega-3 (n-3) the anti-tumour action. In: Carviello G, Serini S (eds) Diet and Cancer. Springer, New York
Wendel M, Heller AR (2009) Anticancer actions of omega-3 fatty acids—current state and future perspectives. Anticancer Agents Med Chem 9:457–470
Rickert RC, Jellusova J, Miletic AV (2011) Signaling by the tumor necrosis factor receptor superfamily in B-cell biology and disease. Immunol Rev 244:115–133
Grassme H, Jekle A, Riehle A, Schwarz H, Berger J, Sandhoff K, Kolesnick R, Gulbins E (2001) CD95 signaling via ceramide-rich membrane rafts. J Biol Chem 276:20589–20596
Blank N, Schiller M, Krienke S, Wabnitz G, Ho AD, Lorenz HM (2007) Cholera toxin binds to lipid rafts but has a limited specificity for ganglioside GM1. Immunol Cell Biol 85:378–382
Kenworthy AK, Petranova N, Edidin M (2000) High-resolution FRET microscopy of cholera toxin B-subunit and GPI-anchored proteins in cell plasma membranes. Mol Biol Cell 11:1645–1655
Shaikh SR, Rockett BD, Salameh M, Carraway K (2009) Docosahexaenoic acid modifies the clustering and size of lipid rafts and the lateral organization and surface expression of MHC class I of EL4 cells. J Nutr 139:1632–1639
Alquobaili F, Miller SA, Muhie S, Day A, Jett M, Hammamieh R (2010) Estrogen receptor-dependent genomic expression profiles in breast cancer cells in response to fatty acids. J Carcinog 8:17
Fan YY, Ly LH, Barhoumi R, McMurray DN, Chapkin RS (2004) Dietary docosahexaenoic acid suppresses T cell protein kinase C theta lipid raft recruitment and IL-2 production. J Immunol 173:6151–6160
Altenburg JD, Siddiqui RA (2009) Omega-3 polyunsaturated fatty acids down-modulate CXCR4 expression and function in MDA-MB-231 breast cancer cells. Mol Cancer Res 7:1013–1020
Vibet S, Goupille C, Bougnoux P, Steghens JP, Gore J, Maheo K (2008) Sensitization by docosahexaenoic acid (DHA) of breast cancer cells to anthracyclines through loss of glutathione peroxidase (GPx1) response. Free Radic Biol Med 44:1483–1491
Ruth MR, Proctor SD, Field CJ (2009) Feeding long-chain n-3 polyunsaturated fatty acids to obese leptin receptor-deficient JCR:lA-cp rats modifies immune function and lipid-raft fatty acid composition. Br J Nutr 101:1341–1350
Carpinteiro A, Dumitru C, Schenck M, Gulbins E (2008) Ceramide-induced cell death in malignant cells. Cancer Lett 264:1–10
Ogretmen B, Hannun YA (2004) Biologically active sphingolipids in cancer pathogenesis and treatment. Nat Rev Cancer 4:604–616
Kawase M, Watanabe M, Kondo T, Yabu T, Taguchi Y, Umehara H, Uchiyama T, Mizuno K, Okazaki T (2002) Increase of ceramide in adriamycin-induced HL-60 cell apoptosis: detection by a novel anti-ceramide antibody. Biochim Biophys Acta 1584:104–114
Dawood S, Ueno NT, Valero V, Woodward WA, Buchholz TA, Hortobagyi GN, Gonzalez-Angulo AM, Cristofanilli M (2011) Identifying factors that impact survival among women with inflammatory breast cancer. Ann Oncol 23(4):870–875
Acknowledgments
Study funding provided by the Canadian Institutes of Health Research (CIHR). J. Ewaschuk was a recipient of a CIHR post-doctoral fellowship.
Conflict of interest
The authors have no conflict of interest to report.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Ewaschuk, J.B., Newell, M. & Field, C.J. Docosahexanoic Acid Improves Chemotherapy Efficacy by Inducing CD95 Translocation to Lipid Rafts in ER− Breast Cancer Cells. Lipids 47, 1019–1030 (2012). https://doi.org/10.1007/s11745-012-3717-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11745-012-3717-7