Abstract
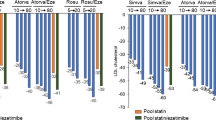
Policosanol is a poorly absorbed nutritional supplement used primarily for cholesterol management. Findings from previous trials evaluating the effects of policosanol are mixed with early data reporting positive lipid effects while more recent studies indicate negligible efficacy. We hypothesized that re-formulating policosanol would result in an improvement in major lipoproteins and possibly provide some explanation for previously conflicting trial data. Our primary objectives were to assess the efficacy and safety of modified-policosanol (MP) on the major lipoproteins among hyperlipidemic subjects receiving background statin therapy or as monotherapy. This 8-week clinical trial consisted of 3 arms. Subjects receiving chronic statin therapy (N = 36) were randomized in a double-blind design to MP 20 mg daily or placebo. In the third arm, subjects not receiving statin therapy (N = 18) were assigned open-label MP 20 mg daily. The utilization of MP when added to background statin therapy or as monotherapy resulted in no significant changes in major lipoproteins (all p > 0.05). The MP therapy was well tolerated with no major adverse events reported. Consistent with recent clinical trial data, MP demonstrated an excellent safety profile but produced no significant effects on major lipoproteins when used as monotherapy or when given with concomitant statin therapy.
Similar content being viewed by others
Abbreviations
- ANOVA:
-
Analysis of variance
- ATP-III:
-
Adult Treatment Panel III
- CHD:
-
Coronary heart disease
- HDL-C:
-
High-density lipoprotein cholesterol
- HMG CoA:
-
Hydroxymethylglutaryl Coenzyme A
- HRT:
-
Hormone replacement therapy
- LDL-C:
-
Low-density lipoprotein cholesterol
- Lp(a):
-
Lipoprotein a
- MP:
-
Modified policosanol
- NCEP:
-
National Cholesterol Education Program
- SPSS:
-
Statistical Package for the Social Sciences
- VLDL:
-
Very low-density lipoprotein cholesterol
References
National Cholesterol Education Program Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults: Third Report of the National Cholesterol Education Program (NCEP) (2002) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III) final report (see comment). Circulation 106:3143-3421
Grundy SM, Cleeman JI, Merz CN, Brewer HB Jr, Clark LT, Hunninghake DB, Pasternak RC, Smith SC Jr, Stone NJ, National Heart LaBI et al (2004) Implications of recent clinical trials for the National Cholesterol Education Program Adult Treatment Panel III guidelines (erratum appears in Circulation. 2004 Aug 10;110(6):763). Circulation 110:227-239
Davidson MH (2002) Combination therapy for dyslipidemia: safety and regulatory considerations. Am J Cardiol 90:20
Kamstrup PR, Tybjaerg-Hansen A, Steffensen R (2009) Nordestgaard BG: Genetically elevated lipoprotein (a) and increased risk of myocardial infarction. JAMA 301:2331–2339
Janikula M (2002) Policosanol: a new treatment for cardiovascular disease? Altern Med Rev 7:203–217
Menendez R, Amor AM, Gonzalez RM, Fraga V, Mas R (1996) Effect of policosanol on the hepatic cholesterol biosynthesis of normocholesterolemic rats. Biol Res 29:253–257
Menendez R, Amor AM, Rodeiro I, Gonzalez RM, Gonzalez PC, Alfonso JL, Mas R (2001) Policosanol modulates HMG-CoA reductase activity in cultured fibroblasts. Arch Med Res 32:8–12
Gouni-Berthold I, Berthold HK (2002) Policosanol: clinical pharmacology and therapeutic significance of a new lipid-lowering agent. Am Heart J 143:356–365
Castano G, Fernandez L, Mas R, Illnait J, Fernandez J et al (2005) Effects of addition of policosanol to omega-3 fatty acid therapy on the lipid profile of patients with type II hypercholesterolaemia. Drugs R D 6:207–219
Castano G, Fernandez L, Mas R, Illnait J, Fernandez JC et al (2003) Comparison of the effects of policosanol and atorvastatin on lipid profile and platelet aggregation in patients with dyslipidaemia and type 2 diabetes mellitus. Clinic Drug Investig 23:639–650
Castano G, Mas R, Fernandez JC, Fernandez L, Alvarez E, Lezcay M (2000) Efficacy and tolerability of policosanol compared with lovastatin in patients with type II hypercholesterolemia and concomitant coronary risk factors. Curr Ther Res 61:137–146
Castano G, Mas R, Fernandez L, Illnait J, Mendoza S, Gamez R, Fernandez J, Mesa M (2005) A comparison of the effects of D-003 and policosanol (5 and 10 mg/day) in patients with type II hypercholesterolemia: a randomized, double-blinded study. Drugs Exp Clin Res 31(Suppl):31–44
Castano G, Mas R, Fernandez L, Illnait J, Mesa M, Alvarez E, Lezcay M (2003) Comparison of the efficacy and tolerability of policosanol with atorvastatin in elderly patients with type II hypercholesterolaemia. Drugs Aging 20:153–163
Chen JT, Wesley R, Shamburek RD, Pucino F, Csako G (2005) Meta-analysis of natural therapies for hyperlipidemia: Plant sterols and stanols versus policosanol. Pharmacotherapy 25:171–183
Fernandez JC, Mas R, Castano G, Menendez R, Alvarez E et al (2001) Comparison of the efficacy, safety and tolerability of policosanol versus fluvastatin in elderly hypercholesterolemic women. Clinic Drug Invest 21:103–113
Marcello S, Gladstein J, Tesone P, Mas R (2000) Effects of bezafibrate plus policosanol or placebo in patients with combined dyslipidemia: a pilot study. Curr Ther Res 61:346–357
Alcocer LCA, Mas R, Fernandez L (1997) A comparative study of policosanol vs acipimox in patients with type II hypercholesterolemia. Data on File @ http://www.policosanol.com
Cubeddu LX, Cubeddu RJ, Heimowitz T, Restrepo B, Lamas GA, Weinberg GB (2006) Comparative lipid-lowering effects of policosanol and atorvastatin: a randomized, parallel, double-blind, placebo-controlled trial. Am Heart J 152:982.e1–982.e5
Berthold HK, Unverdorben S, Degenhardt R, Bulitta M, Gouni-Berthold I (2006) Effect of policosanol on lipid levels among patients with hypercholesterolemia or combined hyperlipidemia: a randomized controlled trial. JAMA 295:2262–2269
Lin Y, Rudrum M, van der Wielen RP, Trautwein EA, McNeill G, Sierksma A, Meijer GW (2004) Wheat germ policosanol failed to lower plasma cholesterol in subjects with normal to mildly elevated cholesterol concentrations. Metabolism 53:1309–1314
Dulin MF, Hatcher LF, Sasser HC, Barringer TA (2006) Policosanol is ineffective in the treatment of hypercholesterolemia: a randomized controlled trial. Am J Clin Nutr 84:1543–1548
Francini-Pesenti F, Beltramolli D, Dall’acqua S, Brocadello F (2008) Effect of sugar cane policosanol on lipid profile in primary hypercholesterolemia. Phytother Res 22:318–322
Francini-Pesenti F, Brocadello F, Beltramolli D, Nardi M, Caregaro L (2008) Sugar cane policosanol failed to lower plasma cholesterol in primitive, diet-resistant hypercholesterolaemia: a double blind, controlled study. Complement Ther Med 16:61–65
Greyling A, De Witt C, Oosthuizen W, Jerling JC (2006) Effects of a policosanol supplement on serum lipid concentrations in hypercholesterolaemic and heterozygous familial hypercholesterolaemic subjects. Br J Nutr 95:968–975
Kassis AN, Jones PJ (2006) Lack of cholesterol-lowering efficacy of Cuban sugar cane policosanols in hypercholesterolemic persons. Am J Clin Nutr 84:1003–1008
Kassis AN, Jones PJ (2008) Changes in cholesterol kinetics following sugar cane policosanol supplementation: a randomized control trial. Lipids Health Dis 7:17
Reiner Z, Tedeschi-Reiner E, Romic Z (2005) Effects of rice policosanol on serum lipoproteins, homocysteine, fibrinogen and C-reactive protein in hypercholesterolaemic patients. Clinic Drug Invest 25:701–707
Marinangeli CP, Kassis AN, Jain D, Ebine N, Cunnane SC, Jones PJ (2007) Comparison of composition and absorption of sugarcane policosanols. Br J Nutr 97:381–388
Hargrove JL, Greenspan P, Hartle DK (2004) Nutritional significance and metabolism of very long chain fatty alcohols and acids from dietary waxes. Exp Biol Med (Maywood) 229:215–226
Porter CJ, Pouton CW, Cuine JF, Charman WN (2008) Enhancing intestinal drug solubilisation using lipid-based delivery systems. Adv Drug Deliv Rev 60:673–691
Kulkarni KR (2006) Cholesterol profile measurement by vertical auto profile method. Clin Lab Med 26:787–802
Acknowledgments
This study was supported by the University of Kansas General Research Fund allocation #23011083 and by Marcor Development Corporation (Carlstadt, NJ). We would also like to express our sincere gratitude to David Mueller, R.Ph., for his diligence and expertise in pharmaceutical compounding and Roger Rajewski Ph.D. for his expertise and assistance in product formulation.
Conflict of interest
Drs. Backes, Gibson, Ruisinger and Moriarty––all report no conflicts of interest with submitted material.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Backes, J.M., Gibson, C.A., Ruisinger, J.F. et al. Modified-Policosanol Does Not Reduce Plasma Lipoproteins in Hyperlipidemic Patients When Used Alone or in Combination with Statin Therapy. Lipids 46, 923–929 (2011). https://doi.org/10.1007/s11745-011-3591-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11745-011-3591-8