Abstract
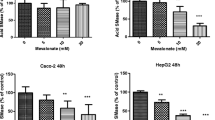
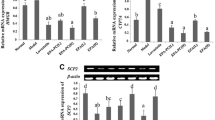
Ezetimibe inhibits cholesterol absorption in the intestine. Sphingomyelin has strong interactions with cholesterol. We investigated the effects of ezetimibe on Sphingomyelinase (SMase) expression in intestine and liver. After feeding rats with ezetimibe (5 mg/kg per day) for 14 days, acid SMase activities in the liver and in the proximal part of small intestine were reduced by 34 and 25%, respectively. Alkaline SMase (alk-SMase) was increased in the proximal part of the small intestine. Administration of lower doses of ezetimibe reduced acid SMase only in the liver by 14% (P < 0.05). In cell culture studies, ezetimibe decreased acid SMase activity in Hep G2 and Caco-2 cells dose-dependently. The reductions were more rapid for Hep G2 cells than for Caco-2 cells. Western blot showed that acid SMase protein was decreased in both Hep G2 and Caco-2 cells by 100 μM ezetimibe. The SM content was increased in Hep G2 cells but not Caco-2 cells, and total cholesterol content was increased in both cell lines 24 h after stimulation with 100 μM ezetimibe. Mevastatin, the inhibitor of cholesterol synthesis, induced a mild increase in acid SMase activity in Hep G2 cells but not Caco-2 cells. Following the reduction of acid SMase, ezetimibe at high dose slightly increased alk-SMase activity. In conclusion, the study demonstrates an inhibitory effect of ezetimibe on acid SMase activity and expression in both liver and intestine.






Similar content being viewed by others
Abbreviations
- Alk-SMase:
-
Alkaline sphingomyelinase
- NPC1:
-
Niemann–Pick C1
- NPC1L1:
-
Niemann–Pick C1 like 1 protein
- SM:
-
Sphingomyelin
- SMase:
-
Sphingomyelinase
References
Bays HE, Neff D, Tomassini JE, Tershakovec AM (2008) Ezetimibe: cholesterol lowering and beyond. Expert Rev Cardiovasc Ther 6:447–470
Altmann SW, Davis H R Jr, Zhu LJ, Yao X, Hoos LM, Tetzloff G, Iyer SP, Maguire M, Golovko A, Zeng M, Wang L, Murgolo N, Graziano MP (2004) Niemann–Pick C1 like 1 protein is critical for intestinal cholesterol absorption. Science 303:1201–1204
Davis H R Jr, Zhu LJ, Hoos LM, Tetzloff G, Maguire M, Liu J, Yao X, Iyer SP, Lam MH, Lund EG, Detmers PA, Graziano MP, Altmann SW (2004) Niemann–Pick C1 like 1 (NPC1L1) is the intestinal phytosterol and cholesterol transporter and a key modulator of whole-body cholesterol homeostasis. J Biol Chem 279:33586–33592
Davies JP, Levy B, Ioannou YA (2000) Evidence for a Niemann–Pick C (NPC) gene family: identification and characterization of NPC1L1. Genomics 65:137–145
Deushi M, Nomura M, Kawakami A, Haraguchi M, Ito M, Okazaki M, Ishii H, Yoshida M (2007) Ezetimibe improves liver steatosis and insulin resistance in obese rat model of metabolic syndrome. FEBS Lett 581:5664–5670
Zuniga S, Molina H, Azocar L, Amigo L, Nervi F, Pimentel F, Jarufe N, Arrese M, Lammert F, Miquel JF (2008) Ezetimibe prevents cholesterol gallstone formation in mice. Liver Int 28:935–947
Slotte JP, Bierman EL (1988) Depletion of plasma-membrane sphingomyelin rapidly alters the distribution of cholesterol between plasma membranes and intracellular cholesterol pools in cultured fibroblasts. Biochem J 250:653–658
Gupta AK, Rudney H (1991) Plasma membrane sphingomyelin and the regulation of HMG-CoA reductase activity and cholesterol biosynthesis in cell cultures. J Lipid Res 32:125–136
Byers DM, Morgan MW, Cook HW, Palmer FB, Spence MW (1992) Niemann–Pick type II fibroblasts exhibit impaired cholesterol esterification in response to sphingomyelin hydrolysis. Biochim Biophys Acta 1138:20–26
Chen H, Born E, Mathur SN, Johlin F C Jr, Field FJ (1992) Sphingomyelin content of intestinal cell membranes regulates cholesterol absorption. Evidence for pancreatic and intestinal cell sphingomyelinase activity. Biochem J 286:771–777
Nyberg L, Duan RD, Nilsson A (2000) A mutual inhibitory effect on absorption of sphingomyelin and cholesterol. J Nutr Biochem 11:244–249
Nilsson Å, Duan R-D (1999) Alkaline sphingomyelinases and ceramidases of the gastrointestinal tract. Chem Phys Lipids 102:97–105
Duan RD (2006) Alkaline sphingomyelinase: an old enzyme with novel implications. Biochim Biophys Acta 1761:281–291
Spence MW, Callahan JW (1989) Sphingomyelin-cholesterol lipidoses: the Niemann–Pick group of diseases. In: Scriver CR, Beaudet AL, Sly WS, Valle D (eds) The metabolic basis of inherited diseases. McGraw-Hill, New York, pp 1655–1676
Duan RD, Bergman T, Xu N, Wu J, Cheng Y, Duan J, Nelander S, Palmberg C, Nilsson A (2003) Identification of human intestinal alkaline sphingomyelinase as a novel ecto-enzyme related to the nucleotide phosphodiesterase family. J Biol Chem 278:38528–38536
Lillienau J, Crombie DL, Munoz J, Longmire-Cook SJ, Hagey LR, Hofmann AF (1993) Negative feedback regulation of the ileal bile acid transport system in rodents. Gastroenterology 104:38–46
Duan RD, Verkade HJ, Cheng Y, Havinga R, Nilsson A (2007) Effects of bile diversion in rats on intestinal sphingomyelinases and ceramidase. Biochim Biophys Acta 1771:196–201
Heider JG, Boyett RL (1978) The picomole determination of free and total cholesterol in cells in culture. J Lipid Res 19:514–518
Duan RD, Nilsson A (2000) Sphingolipid hydrolyzing enzymes in the gastrointestinal tract. Methods Enzymol 311:276–286
Cheng Y, Tauschel HT, Nilsson Å, Duan RD (1999) Administration of ursodeoxycholic acid increases the activities of alkaline sphingomyelinase and caspase-3 in rat colon. Scand J Gastroenterol 34:915–920
Bligh EH, Dyer WJ (1959) A rapid method for total lipid extraction and purification. Can J Biochem Physiol 37:911–918
Ezzet F, Krishna G, Wexler DB, Statkevich P, Kosoglou T, Batra VK (2001) A population pharmacokinetic model that describes multiple peaks due to enterohepatic recirculation of ezetimibe. Clin Ther 23:871–885
Jenkins RW, Canals D, Hannun YA (2009) Roles and regulation of secretory and lysosomal acid sphingomyelinase. Cell Signal 21:836–846
Davis HR, Veltri EP (2007) Zetia: inhibition of Niemann–Pick C1 like 1 (NPC1L1) to reduce intestinal cholesterol absorption and treat hyperlipidemia. J Atheroscler Thromb 14:99–108
Yu L (2008) The structure and function of Niemann–Pick C1-like 1 protein. Curr Opin Lipidol 19:263–269
Reagan J W Jr, Hubbert ML, Shelness GS (2000) Posttranslational regulation of acid sphingomyelinase in Niemann–Pick type C1 fibroblasts and free cholesterol-enriched Chinese hamster ovary cells. J Biol Chem 275:38104–38110
Repa JJ, Turley SD, Quan G, Dietschy JM (2005) Delineation of molecular changes in intrahepatic cholesterol metabolism resulting from diminished cholesterol absorption. J Lipid Res 46:779–789
Gatt S, Bierman EL (1980) Sphingomyelin suppresses the binding and utilization of low density lipoproteins by skin fibroblasts. J Biol Chem 255:3371–3376
Nilsson A, Duan RD (2006) Absorption and lipoprotein transport of sphingomyelin. J Lipid Res 47:154–171
Tani M, Ito M, Igarashi Y (2007) Ceramide/sphingosine/sphingosine 1-phosphate metabolism on the cell surface and in the extracellular space. Cell Signal 19:229–237
Mari M, Fernandez-Checa JC (2007) Sphingolipid signalling and liver diseases. Liver Int 27:440–450
Mari M, Colell A, Morales A, Paneda C, Varela-Nieto I, Garcia-Ruiz C, Fernandez-Checa JC (2004) Acidic sphingomyelinase downregulates the liver-specific methionine adenosyltransferase 1A, contributing to tumor necrosis factor-induced lethal hepatitis. J Clin Invest 113:895–904
Kirschnek S, Paris F, Weller M, Grassme H, Ferlinz K, Riehle A, Fuks Z, Kolesnick R, Gulbins E (2000) CD95-mediated apoptosis in vivo involves acid sphingomyelinase. J Biol Chem 275:27316–27323
Arundel C, Lewis JH (2007) Drug-induced liver disease in 2006. Curr Opin Gastroenterol 23:244–254
Stolk MF, Becx MC, Kuypers KC, Seldenrijk CA (2006) Severe hepatic side effects of ezetimibe. Clin Gastroenterol Hepatol 4:908–911
van Heyningen C (2005) Drug-induced acute autoimmune hepatitis during combination therapy with atorvastatin and ezetimibe. Ann Clin Biochem 42:402–404
Florentin M, Liberopoulos EN, Elisaf MS (2008) Ezetimibe-associated adverse effects: what the clinician needs to know. Int J Clin Pract 62:88–96
Devlin CM, Leventhal AR, Kuriakose G, Schuchman EH, Williams KJ, Tabas I (2008) Acid sphingomyelinase promotes lipoprotein retention within early atheromata and accelerates lesion progression. Arterioscler Thromb Vasc Biol 28:1723–1730
Acknowledgments
The work was supported by grants from Albert Påhlsson Foundation, Swedish Cancer Foundation, Swedish Research Council, and the Research Foundation of Lund University Hospital. Merck Sharp & Dohme (Sweden) AB is thanked for providing ezetimibe and financial support for this study.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Cheng, Y., Liu, F., Wu, J. et al. Ezetimibe Inhibits Expression of Acid Sphingomyelinase in Liver and Intestine. Lipids 44, 897–906 (2009). https://doi.org/10.1007/s11745-009-3343-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11745-009-3343-1