Abstract
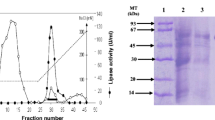
Phospholipase A2 (PLA2) activity was investigated in various tissues of male and female red sea bream. In both male and female fishes, the specific activity of PLA2 in the gills was 70 times higher than that in other tissues, such as the adipose tissue, intestine, and hepatopancreas. Therefore, we tried to purify PLA2 from the gill filaments of red sea bream to near homogeneity by sequential chromatography on Q-Sepharose Fast Flow, Butyl-Cellulofine, and DEAE-Sepharose Fast Flow columns, and by reversed-phase high-performance liquid chromatography. Two minor and one major PLA2, tentatively named G-1, G-2 and G-3 PLA2, were purified, and all showed a single band with an apparent molecular mass of approximately 15 kDa by sodium dodecylsulfate-polyacrylamide gel electrophoresis. The exact molecular mass values of G-1, G-2, and G-3 PLA2 were 14,040, 14,040 and 14,005 Da, respectively. G-1, G-2, and G-3 PLA2 had a Cys 11 and were all identical in N-terminal amino acid sequences from Ala-1 to Glu-56. A full-length cDNA encoding G-3 PLA2 was cloned by reverse transcriptase-polymerase chain reaction and rapid amplification of cDNA ends methods, and G-3 PLA2 was found to be classified to group IB PLA2 from the deduced amino acid sequence. G-1, G-2, and G-3 PLA2 had a pH optimum in an alkaline region at around pH 9–10 and required Ca2+ essentially for enzyme activity, using a mixed-micellar phosphatidylcholine substrate with sodium cholate. These results demonstrate that three group 1 PLA2, G-1, G-2, and G-3 PLA2, are expressed in the gill filaments of red sea bream.
Similar content being viewed by others
Abbreviations
- p-APMSF:
-
p-amidinophenylmethylsulfonyl fluoride
- p-BPB:
-
p-bromophenacyl bromide
- CHAPS:
-
3-[(3-cholamidopropyl) dimethylammoniol-1-propanesulfonate
- CTAB:
-
cetyl trimethylammonium bromide
- HPLC:
-
high-performance liquid chromatography
- MALDI-TOF:
-
matrixassisted laser desorption ionization-time of flight
- PAGE:
-
polyacrylamide gel electrophoresis
- PCR:
-
polymerase chain reaction
- PLA2 :
-
phospholipase(s) A2
- POPC:
-
1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine
- POPE:
-
1-palmitoyl-2-oleoyl-sn-glycero-3-phosphoethanolamine
- POPG:
-
1-palmitoyl-2-oleoyl-sn-glycero-3-phosphoglycerol
- POPS:
-
1-palmitoyl-2-oleoyl-sn-glycero-3-phosphoserine
- RACE:
-
rapid amplification of cDNA ends
- RP-HPLC:
-
reversed-phase-HPLC
- RT:
-
reverse transcriptase
- SDS:
-
sodium lauryl sulfate
- TFA:
-
trifluoroacetic acid
References
Dennis, E.A. (1997) The Growing Phospholipase A2 Superfamily of Signal Transduction Enzymes, Trends Biochem. Sci. 22, 1–2.
Chaminade, B., Le Balle, F., Fourcade, O., Nauze, M., Delagebeaudeuf, C., Gassama-Diagne, A., Simon, M.F., Fauvel, J., and Chap, H. (1999) New Developments in Phospholipase A2, Lipids 34 (Suppl.), S49-S55.
Tischfield, J.A. (1997) A Reassessment of the Low Molecular Weight Phospholipase A2 Gene Family in Mammals, J. Biol. Chem. 272, 17247–17250.
Seilhamer, J.J., Randall, T.L., Yamanaka, M., and Johnson, L.K. (1986) Pancreatic Phospholipase A2: Isolation of the Human Gene and cDNAs from Porcine Pancreas and Human Lung, DNA 5, 519–527.
Tojo, H., Ono, T., Kuramitsu, S., Kagamiyama, H., and Okamoto, M. (1988) A Phospholipase A2 in the Supernatant Fraction of Rat Spleen, J. Biol. Chem. 263, 5724–5731.
Sakata, T., Nakamura, E., Tsuruta, Y., Tamaki, M., Teraoka, H., Tojo, H., and Ono, T. (1989) Presence of Pancreatic Type Phospholipase A2 mRNA in Rat Gastric Mucosa and Lung, Biochim. Biophys. Acta 1007, 124–126.
Tojo, H., Ono, T., and Okamoto, M. (1991) Spleen Phospholipase A2, Methods Enzymol. 197, 390–399.
Tojo, H., Ono, T., and Okamoto, M. (1993) Reverse-Phase High-Performance Liquid Chromatographic Assay of Phospholipases: Application of Spectrophotometric Detection to Rat Phospholipase A2 Isozymes, J. Lipid Res. 34, 837–844.
Kortesuo, P.T., Hietaranta, A.J., Jamia, M., Hirsimaki, P., and Nevalainen, T.J. (1993) Rat Pancreatic Phospholipase A2. Purification, Localization, and Development of an Enzyme Immunoassay, Int. J. Pancreatol. 13, 111–118.
Nevalainen, T.J., and Haapanen, T.J. (1993) Distribution of Pancreatic (Group I) and Synovial-type (Group II) Phospholipases A2 in Human Tissues, Inflammation 17, 453–464.
Hara, S., Kudo, I., Komatani, T., Takahashi, K., Nakatani, Y., Natori, Y., Ohshima, M., and Inoue, K. (1995) Detection of Two 14 kDa Phospholipase A2 Isoforms in Rat Kidney: Their Role in Eicosanoid Synthesis, Biochim. Biophys. Acta 1257, 11–17.
Aarsman, A.J., Schalkwijk, C.G., Neys, F.W., Iijima, N., Wherret, J.R., and van den Bosch, H. (1996) Purification and Characterization of Ca2+-Dependent Phospholipase A2 from Rat Kidney, Arch. Biochem. Biophys. 331, 95–103.
Ohara, O., Ishizaki, J., and Arita, H. (1995) Structure and Function of Phospholipase A2 Receptor, Prog. Lipid Res., 34, 117–138.
Lambeau, G.H., Cupillard, L., and Ladzunski, M. (1997) Membrane Receptors for Venom Phospholipase A2, in Venom Phospholipase A 2 Enzymes: Structure Function and Mechanism (Kini, R.M., eds.), pp. 389–412, John Wiley & Sons, Chichester.
Kundu, G.C., and Mukherjee, A.B. (1997) Evidence That Porcine Pancreatic Phospholipase A2 via Its High Affinity Receptor Stimulates Extracellular Matrix Invasion by Normal and Cancer Cells, J. Biol. Chem. 272, 2346–2353.
Hanasaki, K., Yokota, Y., Ishizaki, J., Itoh, T., and Arita, H. (1997) Resistance to Endotoxic Shock in Phospholipase A2 Receptor-Deficient Mice, J. Biol. Chem. 272, 32792–32797.
Neas, N.P., and Hazel, J.R. (1984) Temperature-Dependent Deacylation of Molecular Species of Phosphatidylcholine by Microsomal Phospholipase A2 of Thermally Acclimated Rainbow Trout, Salmo gairdneri, Lipids 19, 258–263.
Neas, N.P., and Hazel, J.R. (1985) Partial Purification and Kinetic Characterization of the Microsomal Phospholipase A2 from Thermally Acclimated Rainbow Trout (Salmo gairdneri), J. Comp. Physiol. 155B, 461–469.
Audley, M.A., Shetty, K.J., and Kinsella, J.E. (1978) Isolation and Properties of Phospholipase A from Pollock Muscle, J. Food Sci. 43, 1771–1775.
Aaen, B., Jessen, F., and Jensen, B. (1995) Partial Purification and Characterization of a Cellular Acidic Phospholipase A2 from Cod (Gadus morhua), Comp. Biochem. Physiol. 110B, 547–554.
Zambonino Infante, J.L., and Cahu, C.L. (1999) High Dietary Lipid Levels Enhance Digestive Tract Maturation and Improve Dicentrarchus labrax Larval Development, J. Nutr. 129, 1195–2000.
Iijima, N., Nakamura, M., Uematsu, K., and Kayama, M. (1990) Partial Purification and Characterization of Phospholipase A2 from the Hepatopancreas of Red Sea Bream, Nippon Suisan Gakkaishi 56, 1331–1339.
Iijima, N., Chosa, S., Uematsu, K., Goto, T., Hoshita, T., and Kayama, M. (1997) Purification and Characterization of Phospholipase A2 from the Pyloric Caeca of Red Sea Bream, Pagrus major, Fish Physiol. Biochem. 16, 487–498.
Ono, H., and Iijima, N. (1998) Purification and Characterization of Phospholipase A2 Isoforms from the Hepatopancreas of Red Sea Bream, Pagrus major, Fish Physiol. Biochem. 18, 135–147.
Uematsu, K., Kitano, M., Morita, M., and Iijima, N. (1992) Presence and Ontogeny of Intestinal and Pancreatic Phospholipase A2-like Proteins in the Red Sea Bream, Pagrus major. An Immunocytochemical Study, Fish Physiol. Biochem. 9, 427–438.
Laemmli, U.K. (1970) Cleavage of Structural Proteins During the Assembly of Bacteriophage T4, Nature 227, 680–685.
Heinrickson, R.L. (1991) Dissection and Sequence Analysis of Phospholipase A2, Methods Enzymol. 197, 201–214.
Fleer, E.A.M., Verheij, H.M., and de Haas, G.H. (1978) The Primary Structure of Bovine Pancreatic Phospholipase A2, Eur. J. Biochem. 82, 261–269.
Puijk, W.C., Verheij, H.M., and de Haas, G.H. (1977) The Primary Structure of Phospholipase A2 from Porcine Pancreas. A Reinvestigation, Biochim. Biophys. Acta 492, 254–259.
Joubert, F.J., and Taljaard, N. (1980) Purification, Some Properties and Amino Acid Sequences of Two Phospholipase A (CM-II and CM-III) from Naja naja kaouthia Venom, Eur. J. Biochem. 112, 493–499.
Verheij, H.M., Slotboom, A.J., and de Haas, G.H. (1981) Structure and Function of Phospholipase A2, Rev. Physiol. Biochem. Pharmacol. 91, 91–203.
Van den Bosch, H. (1982) Phospholipases, in Phospholipids (Hawthorne, J.N. and Ansell, G.B., eds.), pp. 313–357. Elsevier Biomedical, Amsterdam.
de Haas, G.H., Postema, N.M., Nieuwenhuizen, W., and van Deenen, L.L.M. (1967) Purification and Properties of Phospholipase A from Porcine Pancreas, Biochim. Biophys. Acta 159, 103–117.
Nielsen, H., Brunak, S., and von Heijne, G. (1999) Machine Learning Approaches for the Prediction of Signal Peptides and Other Protein Sorting Signals, Protein Eng. 12, 3–9.
Halban, P.A., and Irminger, J.C. (1994) Sorting and Processing of Secretory Proteins, Biochem. J. 299, 1–18.
Noel, J.P., Deng, T., Hamilton, K.J., and Tsai, M.-D. (1990) Phospholipase A2 Engineering. 3. Replacement of Lysine-56 by Neutral Residues Improves Catalytic Potency Significantly, Alters Substrate Specificity, and Clarifies the Mechanism of Interfacial Recognition, J. Am. Chem. Soc. 112, 3704–3706.
Noel, J.P., Bingman, C.A., Deng, T., Dupureur, C.M., Hamilton, K.J., Jiang, R.-T., Kwak, J.-G., Sekharudu, C., Sundaralingam, M., and Tsai, M.-D. (1991) Phospholipase A2 Engineering. X-ray Structural and Functional Evidence for the Interaction of Lysine-56 with Substrates, Biochemistry 30, 11801–11811.
Lugtigheid, R.B., Otten-Kuipers, M.A., Verheij, H.M., and de Haas, G.H. (1993) Arginine 53 Is Involved in Head-Group Specificity of the Active Site of Porcine Pancreatic Phospholipase A2, Eur. J. Biochem. 213, 517–522.
Beiboer, S.H., Franken, P.A., Cox, R.C., and Verheij, H.M. (1995) An Extended Binding Pocket Determines the Polar Head Group Specificity of Porcine Pancreatic Phospholipase A2, Eur. J. Biochem. 231, 747–753.
Han, S.K., Yoon, E.T., Scott, D.L., Sigler, P.B., and Cho, W. (1997) Structural Aspects of Interfacial Adsorption. A Crystallographic and Site-directed Mutagenesis Study of the Phospholipase A2 from the Venom of Agkistrodon piscivorus piscivorus, J. Biol. Chem. 272, 3573–3582.
Janssen, M.J.W., Vermeulen, L., Van der Helm, H.A., Aarsman, A.J., Slotbloom, A.J., and Egmond, M.R. (1999) Enzymatic Properties of Rat Group IIA and V Phospholipases A2 Compared, Biochim. Biophys. Acta 1440, 56–72.
Rogers, J., Yu, B.Z., Tsai, M.D., Berg, O.G., and Jain, M.K. (1998) Cationic Residues 53 and 56 Control the Anion-induced Interfacial kcat Activation of Pancreatic Phospholipase A2, Biochemistry 37, 9549–56.
Takasaki, C., Yutani, F., and Kajiyashiki, T. (1990) Amino Acid Sequences of Eight Phospholipases A2 from the Venom of Australian King Brown Snake, Pseudechis australis, Toxicon 28, 329–339.
Puijk, W.C., Verheij, H.M., Weitzes, P., and de Haas, G.H. (1979) The Amino Acid Sequence of the Phospholipase A2 Isoenzyme from Porcine Pancreas, Biochim. Biophys. Acta 580, 411–415.
Chang, T.M., Chang, C.H., Wagner, D.R., and Chey, W.Y. (1999) Porcine Pancreatic Phospholipase A2 Stimulates Secretin Release from Secretin-Producing Cells, J. Biol. Chem. 274, 10758–10764.
Laurent, P. (1984) Gill Internal Morphology, in Gills (Hoar, W.S., and Randall, D.J., eds.), pp. 73–183, Academic Press, Inc., Orlando.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Iijima, N., Uchiyama, S., Fujikawa, Y. et al. Purification, characterization, and molecular cloning of group I phospholipases A2 from the gills of the red sea bream, Pagrus major . Lipids 35, 1359–1371 (2000). https://doi.org/10.1007/s11745-000-0653-4
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11745-000-0653-4