Abstract
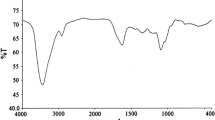
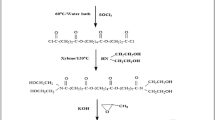
In the present investigation novel nonionic surfactants were synthesized, characterized and tested as inhibitors of the corrosion of carbon steel in 1M HCl solution. The inhibition action of these surfactants was studied by weight loss, galvanostatic polarization and electrochemical impedance spectroscopy. The adsorption of the inhibitors was well described by the Langmuir adsorption isotherm and the adsorption isotherm parameters were determined at different temperatures. The inhibition efficiency was found to rise when increasing the concentration of these compounds and decreasing the temperature. The effect of temperature on the inhibition efficiency of the corrosion process was studied and the values of some activated thermodynamic parameters were calculated to elaborate the mechanism of inhibition. The synthesized nonionic surfactants exhibit good surface and antimicrobial properties.





Similar content being viewed by others
References
Abdallah M, Megahed HE, Sobhi M (2010) Ni+2 cation and imidazole as corrosion inhibitors for carbon steel in sulfuric acid solutions. Monatsh Chem 141:1287–1295
Abdallah M, Megahed HE, Sobhi M (2009) Inhibiting effect of Ni+2 cation +3-methyl pyrazolone as a corrosion inhibitor for carbon steel in sulfuric acid solution. Mater Chem Phys 118:111–117
Sobhi M, Abdallah M, Khairou KS (2012) Sildenafil citrate (Viagra) as corrosion inhibitor for carbon steel in hydrochloric acid solution. Monatsh Chem 143:1379–1387
Negm NA, Aiad IA (2007) Synthesis and characterization of multifunctional surfactants in oil-field protection applications. J Surf Deterg 10:87–92
Mahdavian M, Tehrani-Begha AR, Holmberg K (2011) Comparison of cationic Gemini surfactant and the corresponding monomeric surfactant for corrosion protection of mild steel in hydrochloric acid. J Surf Deterg 14:605–613
Tawfik SM, Sayed A, Aiad I (2012) Corrosion inhibition by some cationic surfactants in oil fields. J Surf Deterg 15:577–585
Yildirim A, Öztürk S, Çetin M (2013) Novel amide-based cationic surfactants as efficient corrosion inhibitors for carbon steel in HCl and H2SO4 media. J Surf Deterg 16:13–23
Fuchs-Godec R (2007) Inhibitory effect of non-ionic surfactants of the Triton-X series on the corrosion of carbon steel in sulphuric acid. Electrochim Acta 52:4974–4981
Hegazy MA, Zaky MF (2010) Inhibition effect of novel nonionic surfactants on the corrosion of carbon steel in acidic medium. Corros Sci 52:1333–1341
Menger FM, Littau CA (1991) Gemini-surfactants: synthesis properties. J Am Chem Soc 113:1451–1452
El-Sawy AA, Mahmoud AA, Shaker NO (1990) Preparation and surface active properties of oxypropylated diol monoester of fatty acids. J Serb Chem Soc 55:395
Findly A (1963) Practical physical chemistry, 6th edn. Longmans, London
Durham K (1961) Surface activity and detergency. MacMillan, London
Draves CZ, Clarkson RG (1931) A new method for evaluation of wetting agents. Am Dyest Report 20:201–208
Takeshi H (1970) Studies of ester containing surfactant: preparation and properties of sodium sulphalkanoates. Bull Chem Soc 43:2236
Ross J, Miles GD (1941) Apparatus for comparison of foaming properties of soaps and detergents. Oil Soap 18:99–102
Amin MS, Eissa AMF, Shaaban AF, El-Sawy AA, El-Sayed R (2004) New heterocycles having double characters; as antimicrobial and surface active agents. Part 1: nonionic compounds from fatty acid isothiocyanate. Grasas Aceites 55:370–377
Aiad IA, Hafiz AA, El-Awady MY, Habib AO (2010) Some imidazoline derivatives as corrosion inhibitors. J Surf Deterg 13:247–254
Li X, Deng S, Fu H (2009) Synergism between red tetrazolium and uracil on the corrosion of cold rolled steel in H2SO4 solution. Corros Sci 51:1344–1355
Yurt A, Ultras S, Dal H (2006) Electrochemical and theoretical investigation on the corrosion of aluminum in acidic solution containing some aldimine compounds. Appl Surf Sci 253:919–925
Larabi L, Harek Y, Benali O, Ghalem S (2005) Hydrazide derivatives as corrosion inhibitors for mild steel in 1M HCl. Prog Org Coat 54:256–262
Zhang S, Tao Z, Li W, Hou B (2009) The effect of some triazole derivatives as inhibitors for the corrosion of mild steel in 1M hydrochloric acid. Appl Surf Sci 255:6757–6763
Tsuru T, Haruyama S, Gijutu B (1978) Corrosion monitor based on impedance method: constraction and its application to homogeneous corrosion. J Jpn Soc Corros Eng 27:573–579
Malik MA, Hashim MA, Nabi F, Al Thabaiti SA, Khan Z (2011) Anti-corrosion ability of surfactants: a review. Int J Electrochem Sci 6:1927–1948
Free ML (2002) Understanding the effect of surfactant aggregation on corrosion inhibition of mild steel in acidic medium. Corros Sci 44:2865–2870
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
About this article
Cite this article
Sobhi, M., El-sayed, R. & Abdallah, M. The Effect of Non Ionic Surfactants Containing Triazole, Thiadiazole and Oxadiazole as Inhibitors of the Corrosion of Carbon Steel in 1M Hydrochloric Acid. J Surfact Deterg 16, 937–946 (2013). https://doi.org/10.1007/s11743-013-1468-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11743-013-1468-y