Abstract
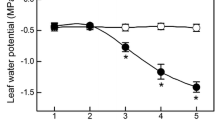
Plants grown at high vapor pressure deficit (VPD) usually present decreased photosynthesis, but stomatal and mesophyll limitation to photosynthesis remain poorly quantified. To better understand the regulation of high VPD on photosynthesis and plant growth in tomatoes, we investigated the limitation of stomatal conductance and mesophyll conductance to photosynthesis and relative importance of stomatal morphology and function in stomatal conductance. Both the net photosynthesis rate and total biomass were significantly limited by high VPD. Meanwhile, stomatal conductance and mesophyll conductance were decreased under high VPD. The stomatal conductance limitation was responsible for 60% of the total photosynthetic limitation. Moreover, a reduction in stomatal density and stomatal size occurred under high VPD, which was significantly correlated with the down-regulation of stomatal conductance. The stomatal morphology contributed to more than half the change in stomatal conductance. Nevertheless, stomatal movement was also an important factor in regulating stomatal conductance. The decrease of hydraulic conductance and transpiration rate with no significant difference in relative water content, leaf water potential, and/or osmotic potential suggested passive hydraulic regulation in the feedforward responses of stomata to high VPD.






Similar content being viewed by others
References
Aliniaeifard S, van Meeteren U (2016) Stomatal characteristics and desiccation response of leaves of cut chrysanthemum (Chrysanthemum morifolium) flowers grown at high air humidity. Sci Hortic 205:84–89. https://doi.org/10.1016/j.scienta.2016.04.025
Barbour MM, Bachmann S, Bansal U, Bariana H, Sharp P (2016) Genetic control of mesophyll conductance in common wheat. New Phytol 209:461–465. https://doi.org/10.1111/nph.13628
Blum A (2017) Osmotic adjustment is a prime drought stress adaptive engine in support of plant production. Plant cell Environ 40:4–10. https://doi.org/10.1111/pce.12800
Bongi G, Loreto F (1989) Gas-exchange properties of salt-stressed olive (Olea europea L.) leaves. Plant Physiol 90:1408–1416. https://doi.org/10.1104/pp.90.4.1408
Brodribb TJ, McAdam SAM (2017) Evolution of the stomatal regulation of plant water content. Plant Physiol 174:639–649. https://doi.org/10.1104/pp.17.00078
Buckley TN (2005) The control of stomata by water balance. New Phytol 168:275–292. https://doi.org/10.1111/j.1469-8137.2005.01543.x
Bunce JA (2006) How do leaf hydraulics limit stomatal conductance at high water vapour pressure deficits? Plant Cell Environ 29:1644–1650. https://doi.org/10.1111/j.1365-3040.2006.01541.x
Campany CE, Tjoelker MG, von Caemmerer S, Duursma RA (2016) Coupled response of stomatal and mesophyll conductance to light enhances photosynthesis of shade leaves under sunflecks. Plant Cell Environ 39:2762–2773. https://doi.org/10.1111/pce.12841
Carins Murphy MR, Jordan GJ, Brodribb TJ (2014) Acclimation to humidity modifies the link between leaf size and the density of veins and stomata. Plant Cell Environ 37:124–131. https://doi.org/10.1111/pce.12136
Christmann A, Weiler EW, Steudle E, Grill E (2007) A hydraulic signal in root-to-shoot signalling of water shortage. Plant J 52:167–174. https://doi.org/10.1111/j.1365-313X.2007.03234.x
Cochard H, Coll L, Le Roux X, Améglio T (2002) Unraveling the effects of plant hydraulics on stomatal closure during water stress in walnut. Plant Physiol 128:282–290. https://doi.org/10.1104/pp.010400
Dow GJ, Bergmann DC, Berry JA (2014) An integrated model of stomatal development and leaf physiology. New Phytol 201:1218–1226. https://doi.org/10.1111/nph.12608
Drake PL, Froend RH, Franks PJ (2013) Smaller, faster stomata: scaling of stomatal size, rate of response, and stomatal conductance. J Exp Bot 64:495–505. https://doi.org/10.1093/jxb/ers347
Du QJ, Zhang DL, Jiao XC, Song XM, Li JM (2018) Effects of atmospheric and soil water status on photosynthesis and growth in tomato. Plant Soil Environ 64:13–19. https://doi.org/10.17221/701/2017-pse
Fanourakis D, Heuvelink E, Carvalho SMP (2013) A comprehensive analysis of the physiological and anatomical components involved in higher water loss rates after leaf development at high humidity. J Plant Physiol 170:890–898. https://doi.org/10.1016/j.jplph.2013.01.013
Fernandes-Silva AA, López-Bernal Á, Ferreira TC, Villalobos FJ (2016) Leaf water relations and gas exchange response to water deficit of olive (cv. Cobrançosa) in field grown conditions in Portugal. Plant Soil 402:191–209. https://doi.org/10.1007/s11104-015-2786-9
Franks PJ, Beerling DJ (2009) Maximum leaf conductance driven by CO2 effects on stomatal size and density over geologic time. Proc Natl Acad Sci 106:10343–10347 https://doi.org/10.1073/pnas.0904209106
Galle A, Florez-Sarasa I, Tomas M, Pou A, Medrano H, Ribas-Carbo M, Flexas J (2009) The role of mesophyll conductance during water stress and recovery in tobacco (Nicotiana sylvestris): acclimation or limitation? J Exp Bot 60:2379–2390. https://doi.org/10.1093/jxb/erp071
Galmés J, Ochogavía JM, Gago J, Roldán EJ, Cifre J, Conesa M (2013) Leaf responses to drought stress in Mediterranean accessions of Solanum lycopersicum: anatomical adaptations in relation to gas exchange parameters. Plant Cell Environ 36:920–935. https://doi.org/10.1111/pce.12022
Grassi G, Magnani F (2005) Stomatal, mesophyll conductance and biochemical limitations to photosynthesis as affected by drought and leaf ontogeny in ash and oak trees. Plant Cell Environ 28:834–849. https://doi.org/10.1111/j.1365-3040.2005.01333.x
Harley PC, Loreto F, Di Marco G, Sharkey TD (1992) Theoretical considerations when estimating the mesophyll conductance to CO2 flux by analysis of the response of photosynthesis to CO2. Plant Physiol 98:1429–1436. https://doi.org/10.1104/pp.98.4.1429
Hubbard RM, Ryan MG, Stiller V, Sperry JS (2001) Stomatal conductance and photosynthesis vary linearly with plant hydraulic conductance in ponderosa pine. Plant Cell Environ 24:113–121. https://doi.org/10.1046/j.1365-3040.2001.00660.x
Laisk AK (1977) Kinetics of photosynthesis and photorespiration in C3-plants. Nauka, Moscow
Lake JA, Walker HJ, Cameron DD, Lomax BH (2017) A novel root-to-shoot stomatal response to very high CO2 levels in the soil: electrical, hydraulic and biochemical signalling. Physiol Plant 159:433–444. https://doi.org/10.1111/ppl.12525
Lawson T, Blatt MR (2014) Stomatal size, speed, and responsiveness impact on photosynthesis and water use efficiency. Plant Physiol 164:1556–1570. https://doi.org/10.1104/pp.114.237107
Liu F, Jensen CR, Andersen MN (2003) Hydraulic and chemical signals in the control of leaf expansion and stomatal conductance in soybean exposed to drought stress. Funct Plant Biol 30:65–73. https://doi.org/10.1071/FP02170
Liu YB, Qin LJ, Han LZ, Xiang Y, Zhao DG (2015a) Overexpression of maize SDD1 (ZmSDD1) improves drought resistance in Zea mays L. by reducing stomatal density. Plant Cell Tissue Organ Cult 122:147–159. https://doi.org/10.1007/s11240-015-0757-8
Liu YY, Song J, Wang M, Li N, Niu CY, Hao GY (2015b) Coordination of xylem hydraulics and stomatal regulation in keeping the integrity of xylem water transport in shoots of two compound-leaved tree species. Tree Physiol 35:1333–1342. https://doi.org/10.1093/treephys/tpv061
Luo YH, Strain BR (1992) Alteration of components of leaf water potential and water content in velvetleaf under the effects of long-term humidity difference. Plant Physiol 98:966–970
Macková J, Vašková M, Macek P, Hronková M, Schreiber L, Šantrůček J (2013) Plant response to drought stress simulated by ABA application: changes in chemical composition of cuticular waxes. Environ Exp Bot 86:70–75. https://doi.org/10.1016/j.envexpbot.2010.06.005
Martins SCV, McAdam SAM, Deans RM, DaMatta FM, Brodribb TJ (2016) Stomatal dynamics are limited by leaf hydraulics in ferns and conifers: results from simultaneous measurements of liquid and vapour fluxes in leaves. Plant Cell Environ 39:694–705. https://doi.org/10.1111/pce.12668
McAdam SAM, Brodribb TJ (2016) Linking turgor with ABA biosynthesis: implications for stomatal responses to vapor pressure deficit across land plants. Plant Physiol 171:2008–2016. https://doi.org/10.1104/pp.16.00380
McAdam SAM, Sussmilch FC, Brodribb TJ (2016) Stomatal responses to vapour pressure deficit are regulated by high speed gene expression in angiosperms. Plant Cell Environ 39:485–491. https://doi.org/10.1111/pce.12633
Niinemets Ü, Díaz-Espejo A, Flexas J, Galmés J, Warren CR (2009) Role of mesophyll diffusion conductance in constraining potential photosynthetic productivity in the field. J Exp Bot 60:2249–2270. https://doi.org/10.1093/jxb/erp036
Perez-Martin A, Flexas J, Ribas-Carbó M, Bota J, Tomás M, Infante JM, Diaz-Espejo A (2009) Interactive effects of soil water deficit and air vapour pressure deficit on mesophyll conductance to CO2 in Vitis vinifera and Olea europaea. J Exp Bot 60:2391–2405. https://doi.org/10.1093/jxb/erp145
Qiu C, Ethier G, Pepin S, Dubé P, Desjardins Y, Gosselin A (2017) Persistent negative temperature response of mesophyll conductance in red raspberry (Rubus idaeus L.) leaves under both high and low vapour pressure deficits: a role for abscisic acid? Plant. Cell Environ 40:1940–1959. https://doi.org/10.1111/pce.12997
Rodriguez-Dominguez CM, Buckley TN, Egea G, de Cires A, Hernandez-Santana V, Martorell S, Diaz-Espejo A (2016) Most stomatal closure in woody species under moderate drought can be explained by stomatal responses to leaf turgor. Plant Cell Environ 39:2014–2026. https://doi.org/10.1111/pce.12774
Sagardoy R et al (2010) Stomatal and mesophyll conductances to CO2 are the main limitations to photosynthesis in sugar beet (Beta vulgaris) plants grown with excess zinc. New Phytol 187:145–158. https://doi.org/10.1111/j.1469-8137.2010.03241.x
Savvides A, Fanourakis D, van Ieperen W (2012) Co-ordination of hydraulic and stomatal conductances across light qualities in cucumber leaves. J Exp Bot 63:1135–1143. https://doi.org/10.1093/jxb/err348
Simonin KA, Burns E, Choat B, Barbour MM, Dawson TE, Franks PJ (2015) Increasing leaf hydraulic conductance with transpiration rate minimizes the water potential drawdown from stem to leaf. J Exp Bot 66:1303–1315. https://doi.org/10.1093/jxb/eru481
Tanaka Y, Fujii K, Shiraiwa T (2010) Variability of leaf morphology and stomatal conductance in soybean cultivars. Crop Sci 50:2525–2532. https://doi.org/10.2135/cropsci2010.02.0058
Tardieu F, Parent B, Simonneau T (2010) Control of leaf growth by abscisic acid: hydraulic or non-hydraulic processes? Plant. Cell Environ 33:636–647. https://doi.org/10.1111/j.1365-3040.2009.02091.x
Tomeo NJ, Rosenthal DM (2017) Variable mesophyll conductance among soybean cultivars sets a tradeoff between photosynthesis and water-use-efficiency. Plant Physiol 174:241–257. https://doi.org/10.1104/pp.16.01940
Warren C (2006) Estimating the internal conductance to CO2 movement. Funct Plant Biol 33:431–442. https://doi.org/10.1071/FP05298
Warren CR (2008) Soil water deficits decrease the internal conductance to CO2 transfer but atmospheric water deficits do not. J Exp Bot 59:327–334. https://doi.org/10.1093/jxb/erm314
Xie C et al (2012) Overexpression of MtCAS31 enhances drought tolerance in transgenic Arabidopsis by reducing stomatal density. New Phytol 195:124–135. https://doi.org/10.1111/j.1469-8137.2012.04136.x
Xu Z, Zhou G (2008) Responses of leaf stomatal density to water status and its relationship with photosynthesis in a grass. J Exp Bot 59:3317–3325. https://doi.org/10.1093/jxb/ern185
Zhang DL, Zhang ZD, Li JM, Chang YB, Du QJ, Pan TH (2015) Regulation of vapor pressure deficit by greenhouse micro-fog systems improved growth and productivity of tomato via enhancing photosynthesis during summer season. PloS One 10:e0133919. https://doi.org/10.1371/journal.pone.0133919
Acknowledgements
This work was supported by the National Science Foundation of China [31471916].
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
Additional information
Communicated by P. Sowinski.
Rights and permissions
About this article
Cite this article
Du, Q., Xing, G., Jiao, X. et al. Stomatal responses to long-term high vapor pressure deficits mediated most limitation of photosynthesis in tomatoes. Acta Physiol Plant 40, 149 (2018). https://doi.org/10.1007/s11738-018-2723-7
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11738-018-2723-7