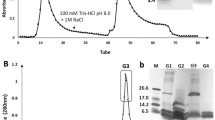
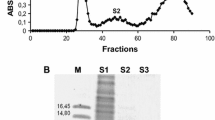
Abstract
This study aimed to detect and characterize antimicrobial proteins, especially antimicrobial peptides (AMPs) from leaves and roots of Capsicum annuum and to evaluate their inhibitory activities against different phytopathogenic fungi and the bacterium Xanthomonas euvesicatoria. Two methodologies were used for the extraction of peptides from leaves and roots of C. annuum: acid and ethanolic extraction. Extracts were subjected to reversed-phase chromatography on HPLC. The extraction and purification procedures were analysed by uni- and bi-dimensional electrophoresis in tricine gels. Our results show that alcoholic and acid extracts from both tissues can inhibit the growth of the phytopathogenics fungi C. lindemuthianum and C. gloeosporioides. The acid extracts from both tissues are active against X. euvesicatoria and only leaf extracts displayed specific inhibitory activity towards trypsin and α-amylase activity. The data compiled here aim to contribute to establish the multiplicity of potential uses of plant AMPs for the control of pests and pathogens of agricultural relevance.










Similar content being viewed by others
Abbreviations
- AMPs:
-
Antimicrobial peptides
- LTP:
-
Lipid-transfer proteins
- BAPNA:
-
N-Benzoyl-dl-arginyl-p-nitroanilide
References
Altschul SF, Madden TL, Schäffer AA, Zhang J, Zhang Z, Miller W, Lipman DJ (1997) Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res 25:3389–3402
Bard GCV, Nascimento VV, Oliveira AE, Rodrigues R, Da Cunha M, Vasconcelos IM, Carvalho AO, Gomes VM (2014) Vicilin-like peptides from Capsicum baccatum L. seeds are α-amylase inhibitors and exhibit antifungal activity against important yeasts in medical mycology. Biopolymers 101:335–343
Bard GCV, Nascimento VV, Ribeiro SFF, Rodrigues R, Pereles J, Teixeira-Ferreira A, Carvalho AO, Fernandes KV, Gomes VM (2015) Characterization of peptides from Capsicum annum hybrid seeds with inhibitory activity against α-amylase, serine proteinases and fungi. Protein J 34:122–129
Baumann C, Kampfer U, Schurch S, Schaller J, Largiader C, Nentwig W, Kuhn-Nentwig L (2010) Ctenidins: antimicrobial glycine-rich peptides from the hemocytes of the spider Cupiennius salei. Cell Mol Life Sci 67:2787–2798
Benko-Iseppon AM, Galdino SL, Calsa T Jr, Kido EA, Tossi Belarmino LC, Crovella S (2010) Overview on plant antimicrobial peptides. Curr Protein Pept Sci 11:181–188
Bernfeld P (1955) Amylase α and β. Methods Enzymol 1:149–158. https://doi.org/10.1016/0076-6879(55)01021-5
Bjellqvist B, Kristina EK, Righetti PG, Gianazza E, Görg A, Westermeir R, Postel W (1982) Isoelectric focusing in immobilized pH gradients: principle, methodology and some applications. J Biochem Biophys Methods 6:317–339
Broekaert WF, Terras FRG, Cammue BPA, Vanderleyden J (1990) An automated quantitative assay for fungal growth inhibition. FEMS Microbiol Lett 69:55–59
Broekaert WF, Cammue BPA, De Bolle MFC, Thevisssem K, De Samblanx GW, Osborn RW, Nielson K (1997) Antimicrobial peptides from plants. Plants Sci 16:297–323
Brogden K (2005) Antimicrobial peptides: pore formers or metabolic inhibitors in bacteria? Nat Rev Microbiol 3:238–250
Carvalho AO, Gomes VM (2009) Plant defensins—prospects for the biological functions and biotechnological properties. Peptides 30:1007–1020
Carvalho AO, Gomes VM (2011) Plant defensins and defensin-like peptides-biological activities and biotechnological applications. Curr Pharm Des 17:4270–4293
Castro MS, Fontes W (2005) Plant defense and antimicrobial peptides. Protein Pept Lett 12:13–18
Claeson P, Göransson U, Johansson S, Luijendijk T, Bohlin L (1998) Fractionation protocol for the isolation of polypeptides from plant biomass. J Nat Prod 61:77–81
Cruz LP, Ribeiro SFF, Carvalho AO, Vasconcelos IM, Rodrigues R, Da Cunha M, Gomes VM (2010) Isolation and partial characterization of a novel lipid transfer protein (LTP) and antifungal activity of peptides from chilli pepper seeds. Protein Pept Lett 17:311–318
Dias GB, Gomes VM, Moraes TMS, Zottich UP, Rabelo GR, Carvalho AO, Moulin M, Gonçalves LSA, Rodrigues R, Da Cunha M (2013a) Characterization of Capsicum species using anatomical and molecular data. Genet Mol Res 12:488–6501
Dias GB, Gomes VM, Zottich UP, Ribeiro SFF, Carvalho AO, Rodrigues R, Machado OLT, Fernandes KVS, Ferreira ATS, Perales J, Da Cunha M (2013b) Isolation, characterization and antifungal activity of proteinase inhibitors from Capsicum chinense Jacq. seeds. Protein J 32:15–26
Diz MS, Carvalho AO, Rodrigues R, Neves-Ferreira AG, Da Cunha M, Alves EW, Okorokova-Façanha AL, Oliveira MA, Perales J, Machado OL, Gomes VM (2006) Antimicrobial peptides from chili pepper seeds causes yeast plasma membrane permeabilization and inhibits the acidification of the medium by yeast cells. Biochim Biophys Acta 1760:1323–1332
Diz MS, Carvalho AO, Ribeiro SFF, Da Cunha M, Beltramini LM, Nascimento VV, Machado OLT, Gomes VM (2011) Characterisation, immunolocalisation and antifungal activity of a lipid transfer protein from chili pepper (Capsicum annuum) seeds with novel α-amylase inhibitory properties. Physiol Plant 142:233–246
Edman P (1950) Method for determination of the amino acid sequence in peptides. Acta Chem Scand 4:283–293
Egorov TA, Odintsova TI, Pukhalsky VA, Grishin EV (2005) Diversity of wheat anti-microbial peptides. Peptides 26:2064–2073
Fardim JM, Carvalho LP, Nascimento VV, Melo EJTM, Gomes VM, Machado OLT, Carvalho AO (2016) Biochemical purification of proteins from bertholletia excelsa seeds and their antileishmanial action in vitro. World J Pharm Res 5:273–300
Filho RL, Romeiro RS (2009) Sensibilidade de Xanthomonas vesicatoria a antibióticos para desenvolvimento de um meio semi-seletivo. Rev Tróp Ciênc Agric Biol 3:28
Gadge PP, Wagh SK, Shaikh FK, Tak RD, Padul MV, Kachole MS (2015) A bifunctional α-amylase/trypsin inhibitor from pigeonpea seeds: purification, biochemical characterization and its bio-efficacy against Helicoverpa armigera. Pestic Biochem Physiol 125:17–25
Gomes MTR, Oliva ML, Lopes MTP, Sala CE (2011) Plant proteinases and inhibitors: an overview of biological function and pharmacological activity. Curr Protein Pept Sci 12:417–436
Görg A, Postel W, Günther S (1988) The current state of two-dimensional electrophoresis with immobilized pH gradients. Electrophoresis 9:531–546
Hancock RE, Sahl HG (2006) Antimicrobial and host-defense peptides as new anti-infective therapeutic strategies. Nat Biotechnol 24:15511–11557
Islamov RA, Furusov OV (2007) Bifunctional inhibitor of alpha-amylase/trypsin from wheat grain. Prikl Biokhim Mikrobiol 43(4):419–423
Kouassi CK, Koffi-Nevry R, Guillaume LY, Yéssé ZN, Koussémon M, Kablan T, Athanase KK (2012) Profiles of bioactive compounds of some pepper fruit (Capsicum L.) varieties grown in Côte d’Ivoire. Innov Rom Food Biotechnol 11:23–31
Lai Y, Gallo RL (2009) AMPed up immunity: how antimicrobial peptides have multiple roles in immune defense. Trends Immunol 30:131–141
Laskowski NJ Jr, Kato I (1980) Protein inhibitor of proteinases. Annu Rev Biochem 49:593–626
Macedo ML, Garcia VA, Freire M, Richardson M (2007) Characterization of a Kunitz trypsin inhibitor with a single disulfide bridge from seeds of Inga laurina (SW.) Willd. Phytochemistry 68:1104–1111
Mishra M, Mahajan N, Tamhane VA, Kulkarni MJ, Baldwin IT, Gupta VS, Giri AP (2012)) Stress inducible proteinase inhibitor diversity in Capsicum annuum. BMC Plant Biol 12:217–222
Moguel-Salazar F, Brito-Argáez L, Díaz-Brito M, Islas-Flores IA (2011) A review of a promising therapeutic and agronomical alternative: antimicrobial peptides from Capsicum sp. Afr J Biotechnol 10:19918–19928
Mosolov VV, Valueva TA (2005) Proteinase inhibitors and their function in plants: a review. Appl Biochem Microbiol 41:227–246
Moulim MM, Rodrigues R, Ribeiro SFF, Gonçalves LSA, Bento CS, Sudré CP, Vasconcelos IM, Gomes VM (2014) Trypsin inhibitors from Capsicum baccatum var. pendulum leaves involved in Pepper yellow mosaic virus resistance. Genet Mol Res 13:9229–9243
Nagaoka I, Hirota S, Yomogida S, Ohwada A, Hirata M (2000) Synergistic actions of antibacterial neutrophil defensins and cathelicidins. Inflamm Res 49:73–79
Nguyen LT, Haney EF, Vogel HJ (2011) The expanding scope of antimicrobial peptide structures and their modes of action. Trends Biotechnol 29:464–472
Nicolas P (2009) Multifunctional host defense peptides: intracellular-targeting antimicrobial peptides. FEBS J 276:6483–6496
O’Farrel PH (1975) High resolution two-dimensional electrophoresis of proteins. J Biol Chem 250:4007–4021
Ohtsubo K, Richardson M (1992) The amino acid sequence of a 20 kDa bifunctional subtilisin/alpha-amylase inhibitor from bran of rice (Oryza sativa L.) seeds. FEBS Lett 309(1):68–72
Payan F (2004) Structural basis for the inhibition of mammalian and insect alpha-amylases by plant protein inhibitors. Biochim Biophys Acta 1696:171–180
Pelegrini PB, Del Sarto RP, Silva ON, Franco OL, Grossi-de-As MF (2011) Antibacterial peptides from plants: what they are and how they probably work. Biochem Int 2011:250–349
Peters BM, Shirtliff ME, Jabra-Rizk MA (2010) Antimicrobial peptides: primeval molecules or future drugs? PLoS Pathog 6:e1001067
Ribeiro SF, Carvalho AO, Da Cunha M, Rodrigues R, Cruz LP, Melo VM, Vasconcelos IM, Melo EJ, Gomes VM (2007) Isolation and characterization of novel peptides from chilli pepper seeds: antimicrobial activities against pathogenic yeasts. Toxicon 50:600–611
Ribeiro SF, Silva MS, Da Cunha M, Carvalho AO, Dias GB, Rabelo G, Mello EO, Santa-Catarina C, Rodrigues R, Gomes VM (2012) Capsicum annuum L. trypsin inhibitor as a template scaffold for new drug development against pathogenic yeast. Antonie Van Leeuwenhoek 101:657–670
Richins RD, Micheletto S, O’Connell MA (2010) Gene expression profiles unique to chile (Capsicum annuum L.) resistant to Phytophthora root rot. Plant Sci 178:192–201
Riva-Souza EM, Rodrigues R, Pereira MG, Sudré CP, Karasawa M (2004) Inheritance of bacterial spot disease in Capsicum annuum L. The CBAB 4:490–494
Schägger H, von Jagow G (1987) Tricine-sodium dodecyl sulfate-polyacrylamide gel electrophoresis for the separation of proteins in the range from 1 to 100 kDa. Anal Biochem 166:368–379
Silva SM, Koehnlein EA, Bracht A, Castoldi R, Morais RG, Baesso ML, Peralta RA, Souza CGM, Sá-Nakanishi BA, Sá-Nakanishi AB, Peralta RM (2014) Inhibition of salivary and pancreatic α-amylases by a pinhão coat (Araucaria angustifolia) extract rich in condensed tannin. Food Res Int 56:1–8
Svensson B, Fukuda k, Nielsen PK, Bønsager BC (2004) Proteinaceous alfa-amylase inhibitors. Biochim Biophys Acta 1696:145–156
Taveira GB, Mathias LS, Motta OV, Machado OLT, Rodrigues R, Carvalho AO, Teixeira- Ferreira A, Perales J, Vasconcelos IM, Gomes VM (2014) Thionin-like peptides from Capsicum annuum fruits with high activity against human pathogenic bacteria and yeasts. Biopolymers 102:30–39
Thevissen K, Terras FRG, Broekaert WF (1999) Permeabilization of fungal membranes by plant defensins inhibits fungal growth. Appl Environ Microbiol 65:5451–5458
Thevissen K, Warnecke DC, François IEJA., Leipelt M, Heinz E, Ott C, Zährinder U, Thomma BPHJ., Ferket KA (2004) Defensins from insects and plants interact with fungal glucosylceramides. J Biol Chem 279:3900–3905
Zottich U, Da Cunha M, Carvalho AO, Dias GB, Silva NC, Santos IS, do Nacimento VV, Miguel EC, Machado OL, Gomes VM (2011) Purification, biochemical characterization and antifungal activity of a new lipid transfer protein (LTP) from Coffea canephora seeds with α-amylase inhibitor properties. Biochim Biophys Acta 1810:375–383
Acknowledgements
We acknowledge the financial support of the Brazilian agencies CNPq (305495/2017-8), FAPERJ (E-26/202.735/2016), and CAPES through the CAPES/Toxinology project.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Z.-L. Zhang.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
da Silva Pereira, L., do Nascimento, V.V., de Fátima Ferreira Ribeiro, S. et al. Characterization of Capsicum annuum L. leaf and root antimicrobial peptides: antimicrobial activity against phytopathogenic microorganisms. Acta Physiol Plant 40, 107 (2018). https://doi.org/10.1007/s11738-018-2685-9
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11738-018-2685-9