Abstract
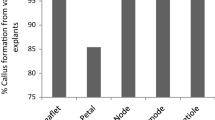
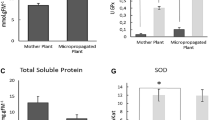
Ephedra foliata, (Gymnosperm) is a pharmaceutically important plant known for the last 5,000 years and has a number of medicinal properties. We describe here for the first time, a method for plant regeneration from callus established from axillary buds as explant, with the aim of optimizing alkaloids production in vitro. The tissue cultures initiated are being maintained for the last 3 years on Murashige and Skoog (Physiol Plant 15:473–497, 1962) medium containing 0.5 mg l−1 each of 2, 4-D and Kin. Maintained callus cultures exhibited regeneration potential and maximum number (23.5 ± 0.44 shoots per culture vessel) of shoots with an average height (4.94 ± 0.23 cm) was achieved on MS medium containing combination of 0.25 mg l−1 each of Kin, BA and 0.1 mg l−1 of NAA. About 84.9 % regenerated shoots were rooted under ex vitro conditions on Soilrite®, if their base was treated with 500 mg l−1 of IBA for 5 min. The rooted plantlets were successfully acclimatized under greenhouse conditions with ≈80 % survival rate. We analyzed alkaloid contents of tissue culture raised plants/callus as affected by the different concentrations and combination of two additives, i.e., l-phenylalanine and IBA. The alkaloid production was higher in the in vitro grown cultures than field-grown plants. Highest alkaloid content was recorded in callus culture on M5 medium having 0.5 mg l−1 each of 2, 4-D and Kin, 100 mg l−1 l-phenylalanine and 5 mg l−1 IBA. The present protocol may be applicable for the large-scale cultivation of E. foliata and selection of cell line having higher secondary metabolite contents of this pharmaceutically important threatened plant species.


Similar content being viewed by others
Abbreviations
- 2, 4-D:
-
2, 4-Dichlorophenoxyacetic acid
- BA:
-
6-Benzyladenine
- IBA:
-
Indole-3-butyric acid
- Kin:
-
Kinetin
- MS:
-
Murashige and Skoog (1962) medium
- NAA:
-
α-Naphthalene acetic acid
- PFD:
-
Photon flux density
- PGRs:
-
Plant growth regulators
- RH:
-
Relative humidity
- TLC:
-
Thin layer chromatography
References
Ahmad M (2004) Standardization of plant-based medicine. Int Chem Pharm Med J 1:87–101
Bhojwani SS, Dantu PK (2013) Plant tissue culture: an introductory text. Springer, India
Bonga JM, Klimaszewska KK, von Aderkas P (2010) Recalcitrance in clonal propagation, in particular of conifers. Plant Cell Tissue Organ Cult 100:241–254
Caveney S, Charlet DA, Freitag H, Maier-stolte M, Starratt AN (2001) New observations on the secondary chemistry of world Ephedra (Ephedraceae). Amer J Bot 88:1199–1208
Chandra S, Bandopadhyay R, Kumar V, Chandra R (2010) Acclimatization of tissue cultured plantlets: from laboratory to land. Biotech Lett 32:1199–1205
Cheng ZJ, Wang L, Sun W, Zhang Y, Zhou C, Su YH, Li W, Sun TT, Zhao XY, Li XG, Cheng Y, Zhao Y, Xie Q, Zhang XS (2013) Pattern of auxin and cytokinin responses for shoot meristem induction results from the regulation of cytokinin biosynthesis by auxin response factor 3. Plant Physiol 161:240–251
Chou TQ (1926) Ephedrine and its salts—isolation and properties. J Biol Chem 70:109–115
Coenen C, Lomax TL (1997) Auxin-cytokinin interactions in higher plants: old problems and new tools. Trend Plant Sci 2:351–356
Collin HA (2001) Secondary product formation in plant tissue cultures. Plant Growth Regul 34:119–134
Cromwell BT (1955) The alkaloids. In: Peach K, Tracey MV (eds) Modern methods of plant analysis. Springer, Berlin
Debergh PC, Maene LJ (1981) A scheme for commercial propagation of ornamental plants by tissue culture. Sci Hortic 14:335–345
Deshpande SR, Josekutty PC, Prathapasenan G (1998) Plant regeneration from axillary buds of a mature tree of Ficus religiosa. Plant Cell Rep 17:571–573
El-Moneim Hegazi GA, El Lamey TM (2011) Callus induction and extraction of Ephedrine from Ephedra alata Decne. cultures. Amer Euras J Agric Environ Sci 11:19–25
Hazarika BN (2003) Acclimatization of tissue-cultured plants. Curr Sci 85:1704–1712
Jayanand B, Sudarsanam G, Sharma KK (2003) An efficient protocol for the regeneration of whole plants of chickpea (Cicer arietinum L.) by using axillary meristem explants derived from in vitro germinated seedlings. In Vitro Cell Dev Biol Plant 39:171–179
Lodha D, Rathore N, Kataria V, Shekhawat NS (2014) In vitro propagation of female Ephedra foliata Boiss. & Kotschy ex Boiss.: an endemic and threatened Gymnosperm of the Thar Desert. Physiol Mol Biol Plant 20:375–383
Mousavi B, Parsaeimehr A, Irvani N (2011) Influences of growth regulators on callus induction, ephedrine and pseudoephedrine contents and chemical analysis of mature embryo of Ephedra strobilacea. AAB Bioflux 3:39–45
Muller D, Leyser O (2011) Auxin, cytokinin and the control of shoot branching. Ann Bot 107:1203–1212
Munir C, Iqbal R, Nasim B (1982) Spectrophotometric determination of ephedrine with ninhydrin. J Chem Soc Pak 4:19–23
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:473–497
O’Dowd NA, McCauley PG, Richardson DHS, Wilson G (1993) Callus production, suspension culture and in vitro alkaloid yields of Ephedra. Plant Cell Tissue Organ Cult 34:149–155
O’Dowd NA, McCauley PG, Wilson G, Parnell JAN, Kavanagh TAK, McConnell DJ (1998) X Ephedra species: in vitro culture, micropropagation and the production of ephedrine and other alkaloids. In: Bajaj YPS (ed) Biotechnology in agriculture and forestry 41; Medicinal and aromatic plants X. Springer, Berlin Heidelberg, pp 154–193
Parsaeimehr A, Sargsyan E (2013) Ephedra alkaloids-Alkaloids derived by amination reaction: phenylalanine derived. In: Ramawat KG, Henry M, Mérillon JM (eds) Natural products: phytochemistry, botany and metabolism of alkaloids, phenolics and terpenes. Springer, Berlin Heidelberg, pp 909–922
Parsaeimehr A, Sargsyan E, Javidnia K (2010) Influence of plant growth regulators on callus induction, growth, chlorophyll, ephedrine and pseudoephedrine contents in Ephedra procera. J Med Plant Res 4:1308–1317
Parsaeimehr A, Sargsyan E, Javidnia K (2012) Optimization the usage of l-phenylalanine as a precursor to induce ephedrine and pseudoephedrine production in Ephedra procera Fisch et CA Mey suspension culture. Russ J Biopharm 4:3–10
Patel AK, Agarwal T, Phulwaria M, Kataria V, Shekhawat NS (2014) An efficient in vitro plant regeneration system from leaf of mature plant of Leptadenia reticulata (Jeewanti): a life giving endangered woody climber. Ind Crop Prod 52:499–505
Phulwaria M, Shekhawat NS, Rathore JS, Singh RP (2013) An efficient in vitro regeneration and ex vitro rooting of Ceropegia bulbosa Roxb.—a threatened and pharmaceutical important plant of Indian Thar Desert. Ind Crop Prod 42:25–29
Rai MK, Asthana P, Jaiswal VS, Jaiswal U (2010) Biotechnological advances in guava (Psidium guajava L.): recent developments and prospects for further research. Tree Struct Funct 24:1–12
Ramawat KG (1978) On the growth of Ephedra tissues in culture with special references to secondary products. PhD thesis, Department of Botany, Jai Narain Vyas University, Jodhpur, India
Ramawat KG, Arya HC (1979) Effect of amino acids on Ephedrine production in Ephedra gerardiana callus culture. Phytochem 18:484–485
Ranaweeraa KK, Gunasekarab MTK, Eeswara JP (2013) Ex vitro rooting: a low cost micropropagation technique for Tea (Camellia sinensis (L.) O.Kuntz) hybrids. Sci Hortic 155:8–14
Shekhawat MS, Shekhawat NS (2011) Micropropagation of Arnebia hispidissima (Lehm). DC. and production of alkannin from callus and cell suspension culture. Acta Physiol Plant 33:1445–1450
Shekhawat NS, Phulwaria M, Harish, Rai MK, Kataria V, Shekhawat S, Gupta AK, Rathore NS, Vyas M, Rathore N, Vibha JB, Choudhary SK, Patel AK, Lodha D, Modi R (2012a) Bioresearches of fragile ecosystem/desert. Proc Natl Acad Sci India 82:319–334
Shekhawat S, Shekhawat NS, Choudhary SK, Harish, Kataria V (2012b) Peduncle: a potential source of competent cells for plant regeneration in Pearl millet (Pennisetum glaucum). Phytomorphol 62:1–11
Shukor MFA, Ismail I, Zainal Z, Noor NM (2013) Development of a Polygonum minus cell suspension culture system and analysis of secondary metabolites enhanced by elicitation. Acta Physiol Plant 35:1675–1689
Singh AK (2004) Endangered economic species of Indian desert. Genet Res Crop Evol 51:371–380
Singh V, Pandey RP (1998) Ethanobotany of Rajasthan. Scientific Publisher, Jodhpur 112
Su YH, Liu YB, Zhang Xian-Sheng (2011) Auxin–cytokinin interaction regulates meristem development. Mol Plant 4:616–625
Supriyanto Rohr R (1994) In vitro regeneration of plantlets of Scots pine (Pinus sylvestris) with mycorrhizal roots from subcultured callus initiated from needle adventitious buds. Can J Bot 72:1144–1150
Tapia N, Zamilpa A, Bonfill M, Ventura E, Cruz-Vega D, Villar AD, Cruz-Sosa F, Osuna L (2013) Effect of the culture medium and biotic stimulation on taxane production in Taxus globosa Schltdl in vitro cultures. Acta Physiol Plant 35:3447–3455
Trigiano RN, Gray DJ (2000) Plant tissue culture concepts and laboratory exercises, 2nd edn. CRC Press, Boca Raton
Vallerand AL (1993) Effects of ephedrine/xanthines on thermogenesis and cold tolerance. Int J Obes 17:53–56
Van Der Krieken WM, Breteler H, Visser MHM, Mavridou D (1993) The role of conversion of IBA into IAA on root regeneration in apple: introduction of a test system. Plant Cell Rep 12:203–206
von Arnold S, Sabala I, Bozhkov P, Dyachok J, Filonova L (2002) Developmental pathways of somatic embryogenesis. Plant Cell Tissue Organ Cult 69:233–249
Yamasaki K, Tamaki T, Uzawa S, Sankawa U, Shibata S (1973) Participation of C6-C1, unit in the biosynthesis of Ephedrine in Ephedra. Phytochem 12:2877–2882
Acknowledgments
Authors (DL and AKP) gratefully acknowledge the financial support from Council of Scientific and Industrial Research (CSIR), New Delhi in the form of Junior and Senior Research Fellowship. We are thankful to Dr. Mahendra Bhati, Department of Chemistry, Jai Narain Vyas University, Jodhpur (India) for his duly support in determination of MP of obtained crystals. Authors are also thankful to Dr. Kamini Dinesh (Professor of English) for the language improvement of the present manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by M. H. Walter.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Online Resource 1 (ESM_1)
Figures showing extraction and confirmation of alkaloid contents from callus culture of E. foliata. Supplementary material 1 (PDF 261 kb)
Online Resource 2 (ESM_2)
Table showing identification of product extracts of E. foliata by TLC. Supplementary material 2 (PDF 123 kb)
Rights and permissions
About this article
Cite this article
Lodha, D., Patel, A.K., Rai, M.K. et al. In vitro plantlet regeneration and assessment of alkaloid contents from callus cultures of Ephedra foliata (Unth phog), a source of anti-asthmatic drugs. Acta Physiol Plant 36, 3071–3079 (2014). https://doi.org/10.1007/s11738-014-1677-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11738-014-1677-7