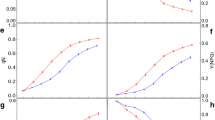
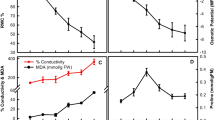
Abstract
Greenhouse and field experiments were carried out to evaluate the potential of specific fluorescence emission parameters for the detection of a temporary water deficit in selected sugar beet (Beta vulgaris L.) genotypes. Changes in the plant physiology due to reduced water availability were recorded with a multiparameter fluorescence sensor in addition to destructive and non-invasive reference analysis. Our results show that an insufficient water supply is followed by only slight changes of the UV-excited blue fluorescence. However, significant alterations due to desiccation were detected in several chlorophyll fluorescence parameters measured after excitation with UV, green and red light. In the scope of our activities, the relevance of the green light source for the fluorescence excitation became evident and enabled to characterize cultivar-specific reactions during dehydration and re-watering period. A field experiment was conducted to validate the data collected in the greenhouse. As proven, several days of low water supply led to effects similar to those observed in the greenhouse study. Our results indicate that the far-red fluorescence, as well as the simple and complex fluorescence ratios having the chlorophyll fluorescence as basis, is the appropriate parameter to evaluate physiological responses of sugar beet plants exposed to a short-term, temporary water deficit.





Similar content being viewed by others
References
Bagatta M, Pacifico D, Mandolino G (2008) Evaluation of the osmotic adjustment response within the genus Beta. J Sugar Beet Res 45:119–133
Ben Ghozlen N, Cerovic ZG, Germain C, Toutain S, Latouche G (2010) Non-destructive optical monitoring of grape maturation by proximal sensing. Sensors 10:10040–10068
Berger B, Parent B, Tester M (2010) High-throughput shoot imaging to study drought responses. J Exp Bot 61:3519–3528
Bloch D, Hoffmann CM, Märländer B (2006) Impact of water supply on photosynthesis, water use and carbon isotope discrimination of sugar beet genotypes. Eur J Agron 24:218–225
Boyer JS (1982) Plant productivity and environment. Science 218:443–448
Brodersen CR, Vogelmann TC (2010) Do changes in light direction affect absorption profiles in leaves? Funct Plant Biol 37:403–412
Buschmann C, Lichtenthaler HK (1998) Principles and characteristics of multi-colour fluorescence imaging of plants. J Plant Physiol 152:297–314
Buschmann C, Langsdorf G, Lichtenthaler HK (2000) Imaging of the blue, green and red fluorescence emission of plants: an overview. Photosynthetica 38:483–491
Cerovic ZG, Morales F, Moya I (1994) Time-resolved studies of blue-green fluorescence of leaves, mesophyll and chloroplasts of sugar beet (Beta vulgaris L.). Biochim Biophys Acta 1188:58–68
Cerovic ZG, Samson G, Morales F, Tremblay N, Moya I (1999) Ultraviolet-induced fluorescence for plant monitoring: present state and prospects. Agronomie 19:543–578
Chaerle L, Van Der Straeten D (2001) Seeing is believing: imaging techniques to monitor plant health. Biochim Biophys Acta 1519:153–166
Chimenti CA, Pearson J, Hall AJ (2002) Osmotic adjustment and yield maintenance under drought in sunflower. Field Crop Res 75:235–246
Clover GRG, Smith HG, Azam-Ali SN, Jaggard KW (1999) The effects of drought on sugar beet growth in isolation and in combination with beet yellows virus infection. J Agric Sci 133:251–261
Cornic G, Masacci A (1996) Leaf photosynthesis under drought stress. In: Baker NR (ed) Photosynthesis and the environment. Kluwer, Dordrecht, pp 347–366
Dahn HG, Günther KP, Lüdeker W (1999) Characterization of drought stress of maize and wheat by means of spectral resolved laser induced fluorescence. EARSeL Adv Remote Sens 1:12–19
D’Ambrosio N, Szabó K, Lichtenthaler HK (1992) Increase of the chlorophyll fluorescence ratio F690/F735 during the autumnal chlorophyll breakdown. Radiat Environ Biophys 31:51–62
Davies WJ, Zhang J (1991) Root signals and the regulation of growth and development in plants in drying soils. Annu Rev Plant Physiol Plant Mol Biol 42:55–70
Flexas J, Medrano H (2002) Drought-inhibition of photosynthesis in C3 plants: stomatal and non-stomatal limitation revisited. Ann Bot 89:183–189
Hák R, Lichtenthaler HK, Rinderle U (1990) Decrease of the chlorophyll fluorescence ratio F690/F730 during greening and development of leaves. Radiat Environ Biophys 29:329–336
Havaux M (1992) Stress tolerance of photosystem II in vivo: antagonistic effects of water, heat, and photoinhibition stresses. Plant Physiol 100:424–432
Hura T, Hura K, Grzesiak M, Rezepka A (2007) Effect of long-term drought stress on leaf gas exchange and fluorescence parameters in C3 and C4 plants. Acta Physiol Plant 29:103–113
Hura T, Hura K, Grzesiak S (2009a) Leaf dehydration induces different content of phenolics and ferulic acid in drought-resistant and -sensitive genotypes of spring triticale. Z Naturforsch C 64:85–95
Hura T, Hura K, Grzesiak S (2009b) Possible contribution of cell-wall-bound ferulic acid in drought resistance and recovery in triticale seedlings. J Plant Physiol 166:1720–1733
Jones HG, Schofield P (2008) Thermal and other remote sensing of plant stress. Gen Appl Plant Physiol (special issue) 34:19–32
Lang M, Stober F, Lichtenthaler HK (1991) Fluorescence emission spectra of plant leaves and plant constituents. Radiat Environ Biophys 30:333–347
Lang M, Lichtenthaler HK, Sowinska M, Heisel F, Miehe JA (1996) Fluorescence imaging of water and temperature stress in plant leaves. J Plant Physiol 148:613–621
Lichtenthaler HK, Rinderle U (1988) The role of chlorophyll fluorescence in the detection of stress conditions in plants. Crit Rev Anal Chem 19:29–85
Lichtenthaler HK, Schweiger J (1998) Cell wall bound ferulic acid, the major substance of the blue-green fluorescence emission of plants. J Plant Physiol 152:272–282
Lichtenthaler HK, Subhash N, Wenzel O, Miehé JA (1997) Laser-induced imaging of blue/red and blue/far-red fluorescence ratios, F440/F690 and F440/F740, as a means of early stress detection in plants. In: Proceedings of the international geoscience and remote sensing symposium IGARSS ‘97. Singapore, IEEE/USA, pp 1799–1801
Meyer S, Cartelat A, Moya I, Cerovic ZG (2003) UV-induced blue-green and far-red fluorescence along wheat leaves: a potential signature for leaf ageing. J Exp Bot 54:757–769
Morales F, Cerovic ZG, Moya I (1996) Time-resolved blue-green fluorescence of sugar beet (Beta vulgaris L.) leaves: spectroscopic evidence for the presence of ferulic acid as the main fluorophore in the epidermis. Biochim Biophys Acta 1273:251–262
Morales F, Cerovic ZG, Moya I (1998) Time-resolved blue-green fluorescence of sugar beet leaves. Temperature-induced changes and consequences for the potential use of blue-green fluorescence as a signature for remote sensing of plants. Aust J Plant Physiol 25:325–334
Ober ES, Clark CJA, Le Bloa M, Royal A, Jaggard KW, Pidgeon JD (2004) Assessing the genetic resources to improve drought tolerance in sugar beet: agronomic traits of diverse genotypes under droughted and irrigated conditions. Field Crop Res 90:213–234
Ober ES, Le Bloa M, Clark CJA, Royal A, Jaggard KW, Pidgeon JD (2005) Evaluation of physiological traits as indirect selection criteria for drought tolerance in sugar beet. Field Crop Res 91:231–249
Schweiger J, Lang M, Lichtenthaler HK (1996) Differences in fluorescence excitation spectra of leaves between stressed and non-stressed plants. J Plant Physiol 148:536–547
Stober F, Lichtenthaler HK (1993a) Studies on the constancy of the blue and green fluorescence yield during the chlorophyll fluorescence induction kinetics (Kautsky effect). Radiat Environ Biophys 32:357–365
Stober F, Lichtenthaler HK (1993b) Characterization of the laser-induced blue, green and red fluorescence signatures of leaves of wheat and soybean leaves grown under different irradiance. Physiol Plantarum 88:696–704
Yordanov I, Velikova V, Tsonev T (2003) Plant responses to drought and stress tolerance. Bulgarian J Plant Physiol (special issue) pp 187–206
Acknowledgments
The authors are grateful to Dr. Britta Schulz and the company KWS Saat AG for providing the seeds of sugar beet, the company Force-A for the technical assistance with the fluorescence sensor, and Dr. Uwe Rascher (Forschungszentrum Jülich) for the fruitful discussions. This study is part of the CROPSENSe.net research project “Networks of excellence in agricultural and nutrition research”, which is financially supported by the German Federal Ministry of Education and Research (BMBF 0315529) and the European Union for regional development (z1011bc001a).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by J.-H. Liu.
Electronic supplementary material
Below is the link to the electronic supplementary material.
11738_2012_1213_MOESM1_ESM.docx
Supplementary Fig. 1. Influence of water supply on the far-red fluorescence (FRF) recorded after green excitation light (G) on the sugar beet cultivars Pauletta (a), Berenika (b), Cesira (c) and Mauricia (d) cultivated in greenhouse. Measurements were regularly taken on marked leaves between 59 and 105 DAS. Grey regions in the graphs illustrate the periods where the water supply was stopped in the case of the non-irrigated plants. Values indicate mean ± SE (n ≥ 8). Asterisks indicate significant differences with a P ≤ 0.05 (t test) between leaves of irrigated (control) and seasonal non-irrigated (stressed) plants for each cultivar and measuring day (DOCX 2064 kb)
11738_2012_1213_MOESM2_ESM.docx
Supplementary Fig. 2. Influence of a seasonal water shortage on the blue fluorescence (BF) excited with UV-light on the four sugar beet genotypes Pauletta (a), Berenika (b), Cesira (c) and Mauricia (d). Leaves were measured at day 104 and 108 after sowing of plants cultivated under field conditions. Columns indicate mean ± SE (n = 96). Asterisks mean significant values with a P ≤ 0.05 (t test) between leaves of irrigated (control) and rainfed (stressed) plants for each cultivar and measuring day (DOCX 38 kb)
Rights and permissions
About this article
Cite this article
Leufen, G., Noga, G. & Hunsche, M. Physiological response of sugar beet (Beta vulgaris) genotypes to a temporary water deficit, as evaluated with a multiparameter fluorescence sensor. Acta Physiol Plant 35, 1763–1774 (2013). https://doi.org/10.1007/s11738-012-1213-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11738-012-1213-6