Abstract
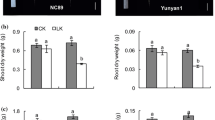
Several lines of evidence suggest that nitric oxide (NO) and hydrogen peroxide (H2O2) are important signal molecules involved in plant development and other physiological processes. Marigold (Tagetes erecta L. ‘Marvel’) was used to understand the role and relationship of NO and H2O2 in adventitious root development of plants. The results showed that the effects of H2O2 or NO on adventitious root organogenesis of explants were dose dependent, with maximal biological responses at 200 μM H2O2 or 50 μM NO donor sodium nitroprusside (SNP). The results also indicated the importance of both putative NO synthase (NOS)-like and nitrate reductase (NR) enzymes, which might be responsible for the production of NO in explants during rooting. Additionally, guanosine 3′, 5′ -cyclic monophosphate (cGMP) was involved in NO- induced root formation of marigold, but it was not involved in H2O2- mediated rooting process. The root number and length of explants treated with NO and H2O2 simultaneously were significantly higher than those of explants treated with H2O2 or NO alone. Moreover, NO treatments enhanced endogenous H2O2 levels in hypocotyls. Together, these results indicate that NO and H2O2 play crucial roles in the adventitious root development of marigold explants both synergistically and independently.








Similar content being viewed by others
References
Beligni MV, Lamattina L (2001) Nitric oxide: a nontraditional regulator of plant growth. Trends Plant Sci 6:508–509
Bright J, Desikan R, Hancock JT, Weir IS, Neill SJ (2006) ABA-induced NO generation and stomatal closure in Arabidopsis are dependent on H2O2 synthesis. Plant J 45:113–122
Correa-Aragunde NM, Graziano ML, Lamattina L (2004) Nitric oxide plays a central role in determining lateral root development in tomato. Planta 218(6):900–905
Correa-Aragunde NM, Graziano ML, Chevalier C, Lamattina L (2006) Nitric oxide modulates the expression of cell cycle regulatory genes during lateral root formation in tomato. J Exp Bot 57(3):581–588
Crawford NM, Galli M, Tischner R, Heimer YM, Okamoto M, Mack A (2006) Response to Zemojtel et al.: plant nitric oxide synthase: back to square one. Trends Plant Sci 11:526–527
Delledonne M, Xia YJ, Dixon RA, Lamb C (1998) Nitric oxide functions as a signal in plant disease resistance. Nature 394:585–588
Desikan R, Griffiths R, Hancock J, Neill SJ (2002) A new role for an old enzyme: nitrate reductase-mediated nitric oxide generation is required for abscisic acid-induced stomatal closure in Arabidopsis thaliana. Proc Natl Acad Sci USA 99:16314–16318
Desikan R, Cheung MK, Bright J, Henson D, Hancock JT, Neill SJ (2004) ABA, hydrogen peroxide and nitric oxide signaling in stomatal guard cells. J Exp Bot 55(395):205–212
Dordas C, Rivoal J, Hill RD (2003) Plant haemoglobins, nitric oxide and hypoxic stress. Ann Bot 91:173–178
Dunand C, Crèvecoeur M, Penel C (2007) Distribution of superoxide and hydrogen peroxide in Arabidopsis root and their influence on root development: possible interaction with peroxidases. New Phytol 174:332–341
Durner J, Wendehenne D, Klessig DF (1998) Defence gene induction in tobacco by nitric oxide, cyclic GMP, and cyclic ADP-ribose. Proc Natl Acad Sci USA 17:10328–10333
Flores T, Todd CD, Tovar-Mendez A, Dhanoa PK, Correa-Aragunde N, Hoyos ME, Brownfield DM, Mullen RT, Lamattina L, Polacco JC (2008) Arginase-negative mutants of arabidopsis exhibit increased nitric oxide signaling in root development. Plant Physiol 147:1936–1946
Foreman J, Demidchik V, Bothwell JHF, Mylona P, Miedema H, Torres MA, Linstead P, Costa S, Brownlee C, Jones JDG, Davies JM, Dolan L (2003) Reactive oxygen species produced by NADPH oxidase regulate plant cell growth. Nature 422:442–446
Guo F, Okamoto M, Crawford NM (2003) Identification of a plant nitric oxide synthase gene involved in hormonal signaling. Science 302:100–103
Hu X, Neill SJ, Tang Z, Cai W (2005) Nitric oxide mediates gravitropic bending in soybean roots. Plant Physiol 137:663–670
Hung KT, Hsu YT, Kao CH (2006) Hydrogen peroxide is involved in methyl jasmonate-induced senescence of rice leaves. Physiol Plant 127:293–303
Jasid S, Simontacchi M, Bartoli CG, Puntarulo S (2006) Chloroplasts as a nitric oxide cellular source. Effect of reactive nitrogen species on chloroplastic lipids and proteins. Plant Physiol 142:1246–1255
Joo JH, Bae YS, Lee JS (2001) Role of auxin-induced reactive oxygen species in root gravitropism. Plant Physiol 3:1055–1060
Kolbert Z, Bartha B, Erdei L (2008) Exogenous auxin-induced NO synthesis is nitrate reductase-associated in Arabidopsis thaliana root primordia. J Plant Physiol 165:967–975
Kopyra M, Gwód EA (2004) The role of nitric oxide in plant growth regulation and responses to abiotic stresses. Acta Physiol Plant 4:459–472
Kuźniak E, Urbanek H (2000) The involvement of hydrogen peroxide in plant responses to stresses. Acta Physiol Plant 2:195–203
Larkindalea J, Huang BR (2004) Thermotolerance and antioxidant systems in Agrostis stolonifera: involvement of salicylic acid, abscisic acid, calcium, hydrogen peroxide, and ethylene. J Plant Physiol 161:405–413
Lee S, Choi H, Suh S, Doo IS, Oh KY, Choi EJ, Taylor ATS, Low PS, Lee Y (1999) Oligogalacturonic acid and chitosan reduce stomatal aperture by inducing the evolution of reactive oxygen species from guard cells of tomato and Commelina communis. Plant Physiol 121:147–152
Lei Y, Yin C, Li C (2007) Adaptive responses of Populus przewalskii to drought stress and SNP application. Acta Physiol Plant 6:519–526
Li SW, Xue LG, Xu SJ, Feng HY, An LZ (2007) Hydrogen peroxide involvement in formation and development of adventitious roots in cucumber. Plant Growth Regul 52:173–180
Li Y, Yin H, Wang Q, Zhao XM, Du YG, Li FL (2009) Oligochitosan induced Brassica napus L. production of NO and H2O2 and their physiological function. Carbohydr Polym 4:612–617
Lombardo MC, Graziano ML, Polacco JC, Lamattina L (2006) Nitric oxide functions as a positive regulator of root hair development. Plant Signal Behav 1:28–33
Neill S, Barros R, Bright J, Desikan R, Hancock J, Harrison J, Morris P, Ribeiro D, Wilson I (2008) Nitric oxide, stomatal closure, and abiotic stress. J Exp Bot 59(2):165–176
Ottenschläger I, Wolff P, Wolverton C, Bhalero RP, Sandberg G, Ishikawa H, Evans ML, Palme K (2003) Gravity-regulated differential auxin transport from columella to lateral root cap cells. Proc Natl Acad Sci USA 100:2987–2991
Pagnussat GC, Simontacchi M, Puntarulo S, Lamattina L (2002) Nitric oxide is required for root organogenesis. Plant Physiol 129:954–956
Pagnussat GC, Lanteri ML, Lamattina L (2003) Nitric oxide and cyclic GMP are messengers in the indole acetic acid-induced adventitious rooting process. Plant Physiol 132:1241–1248
Pagnussat CG, Lanteri ML, Lombardo MC, Lamattina L (2004) Nitric oxide mediates the indole acetic acid induction activation of a mitogenactivated protein kinase cascade involved in adventitious root development. Plant Physiol 135:279–286
Pei ZM, Murata Y, Benning G, Thomine S, Klüsener B, Allen GJ, Grill E, Schroeder JI (2000) Calcium channels activated by hydrogen peroxide mediate abscisic acid signalling in guard cells. Nature 406(6797):731–734
Rentel MC, Lecourieux D, Ouaked F, Usher SL, Petersen L, Okamoto H, Knight H, Peck SC, Grierson CS, Hirt H, Knight MR (2004) OXI1 kinase is necessary for oxidative burst-mediated signalling in Arabidopsis. Nature 427:858–861
Rockel P, Strube F, Rockel A, Wildt J, Kaiser WM (2002) Regulation of nitric oxide (NO) production by plant nitrate reductase in vivo and in vitro. J Exp Bot 53(366):103–110
Schopfer P, Plachy C, Frahry G (2001) Release of reactive oxygen intermediates (superoxide radicals, hydrogen peroxide, and hydroxyl radicals) and peroxidase in germinating radish seeds controlled by light, gibberellin, and abscisic acid. Plant Physiol 125:1591–1602
She XP, Song XG, He JM (2004) The role and relationship of nitric oxide and hydrogen peroxide in light/dark-regulated stomatal movement in Vicia faba. Acta Bot Sin 46:1292–1300
Sheng YW, Jin W, Min ZY, Ya QW, Bo L, Qiong SL, Ping YL, Hua SW, Xin S (2004) Salicylic acid modulates aluminum-induced oxidative stress in roots of Cassiatora. Acta Bot Sin 46:819–828
Stǒhr C, Stremlau S (2006) Formation and possible roles of nitric oxide in plant roots. J Exp Bot 57(3):463–470
Stǒhr C, Strube F, Marx G, Ullrich WR, Rockel P (2001) A plasma membrane-bound enzyme of tobacco roots catalyses the formation of nitric oxide from nitrite. Planta 212(5–6):835–841
Su GX, Zhang WH, Liu YL (2006) Involvement of hydrogen peroxide generated by polyamine oxidative degradation in the development of lateral roots in soybean. J Integr Plant Biol 48(4):426–432
Suita K, Kiryu T, Sawada M, Mitsui M, Nakagawa M, Kanamaru K, Yamagata H (2009) Cyclic GMP acts as a common regulator for the transcriptional activation of the flavonoid biosynthetic pathway in soybean. Planta 229(2):402–413
Xu MJ, Dong JF, Zhang XB (2008) Signal interaction between nitric oxide and hydrogen peroxide in heat shock-induced hypericin production of Hypericum perforatum suspension cells. Sci China C Life Sci 8:676–686
Zhang X, Zhang L, Dong FC, Gao JF, Galbraith DW, Song CP (2001) Hydrogen peroxide is involved in abscisic acid-induced stomatal closure in Vicia faba. Plant Physiol 126:1438–1448
Zhang A, Jiang M, Zhang J, Ding H, Xu S, Hu X, Tan M (2007) Nitric oxide induced by hydrogen peroxide mediates abscisic acid-induced activation of the mitogen-activated protein kinase cascade involved in antioxidant defense in maize leaves. New Phytol 175:36–50
Acknowledgments
This work was supported by the National Natural Science Foundation of China (No. 40501076); and Key Scientific and Technological Project of Lanzhou city, China (No. 05-1-39; 07-1-04). Authors are grateful to the editors and the anonymous reviewers for their valuable comments and help.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by B. Barna.
Rights and permissions
About this article
Cite this article
Liao, W., Xiao, H. & Zhang, M. Role and relationship of nitric oxide and hydrogen peroxide in adventitious root development of marigold. Acta Physiol Plant 31, 1279–1289 (2009). https://doi.org/10.1007/s11738-009-0367-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11738-009-0367-3