Abstract
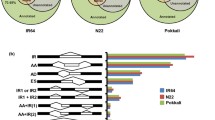
We used the Affymetrix rice genome array to study the gene expression change in response to low- (T1 treatment), middle- (T2 treatment), and high-osmotic stresses (T3 treatment) in rice root at the transcription level. Our experiment results showed that osmotic stress could induce some genes in rice root with a total of 2,082 genes being regulated transcriptionally by at least one osmotic stress. There was little overlap between the genes responding to the low-, middle-, and high-osmotic stresses. A total of 190 transcripts overlapped among all three osmotic stress treatments with 112 genes being up-regulated and 78 transcripts being down-regulated. A total of 113, 159, and 132 transcription factors (TFs) transcripts were regulated at the transcription level by T1, T2, and T3 treatments, respectively, and the expression of 296 genes encoding TFs was changed in response to at least one osmotic stress treatment. 47, 49, and 32 TFs transcripts overlapped between T1 and T2, T1 and T3, and T2 and T3 treatments, and 20 TFs overlapped among all treatments.





Similar content being viewed by others
References
Aharoni A, Dixita S, Jetterb R, Thoenesa E, Arkela GV, Pereiraa A (2004) The SHINE clade of AP2 domain transcription factors activates wax biosynthesis, alters cuticle properties, and confers drought tolerance when overexpressed in Arabidopsis. Plant Cell 16:2463–2480. doi:10.1105/tpc.104.022897
Allison LA (2000) The role of sigma factors in plastid transcription. Biochimie 82(6–7):537–548. doi:10.1016/S0300-9084(00)00611-8
Aoyama T, Oka A (2003) Cytokinin signal transduction in plant cells. J Plant Res 116(3):221–231. doi:10.1007/s10265-003-0094-6
Birnbaum K, Shasha DE, Wang JY, Jung WJ, Lambert GM, Galbraith DW, Benfey PN (2003) A gene expression map of the Arabidopsis root. Science 302:1956–1960. doi:10.1126/science.1090022
Cominelli E, Galbiati M, Vavasseur A, Conti L, Sala T, Vuylsteke M, Leonhardt N, Dellaporta SL, Tonelli C (2005) A guard-cell-specific MYB transcription factor regulates stomatal movements and plant drought tolerance. Curr Biol 15:1196–1200. doi:10.1016/j.cub.2005.05.048
Gergely I, Korcak RF, Faust M (1980) Polyethylene glycol induced water stress effects on apple seedlings. Am Soc Hortic Sci 105:854–857
Haake V, Cook D, Riechmann JL, Pineda O, Thomashow MF, Zhang JZ (2002) Transcription factor CBF4 is a regulator of drought adaptation in Arabidopsis. Plant Physiol 130:639–648. doi:10.1104/pp.006478
Heath LS, Ramakrishnan N, Sederoff RR, Whetten RW, Chevone BI, Struble CA, Jouenne VY, Dawei Chen, Zyl LV, Grene R (2002) Studying the functional genomics of stress responses in loblolly pine with the Express microarray experiment management system. Comp Funct Genomics 3:226–243. doi:10.1002/cfg.169
Himanen K, Vuylsteke M, Vanneste S, Vercruysse S, Boucheron E, Alard P, Chriqui D, Montagu MV, Inze D, Beeckman T (2004) Transcript profiling of early lateral root initiation. Proc Natl Acad Sci USA 104:5146–5151. doi:10.1073/pnas.0308702101
Kaufmann MR, Eckard AN (1971) Evaluation of water stress control with polyethylene glycols by analysis of guttation. Plant Physiol 47(4):453–456. doi:10.1104/pp.47.4.453
Kawasaki S, Borchert C, Deyholos M, Wang H, Brazille S, Kawai K, Galbraith D, Bohnert HJ (2001) Gene expression profiles during the initial phase of salt stress in rice. Plant Cell 13:889–905
Kim S, Kang JY, Cho DI, Park JH, Kim SY (2004) ABF2, an ABRE binding bZIP factor, is an essential component of glucose signaling and its overexpression affects multiple stress tolerance. Plant J 40:75–87. doi:10.1111/j.1365-313X.2004.02192.x
Kreps JA, Wu Y, Chang HS, Zhu T, Wang X, Harper JF (2002) Transcriptome changes for Arabidopsis in response to salt, osmotic, and cold stress. Plant Physiol 130:2129–2141. doi:10.1104/pp.008532
Maron LG, Kirst M, Mao C, Milner MJ, Menossi M, Kochian LV (2008) Transcriptional profiling of aluminum toxicity and tolerance responses in maize roots. New Physiologist 179:116–128
Oh SJ, Song SI, Kim YS, Jang HJ, Kim SY, Kim M, Kim YK, Nahm BH, Kim JK (2005) Arabidopsis CBF3/DREB1A and ABF3 in transgenic rice increased tolerance to abiotic stress without stunting growth. Plant Physiol 138:341–351. doi:10.1104/pp.104.059147
Ozturk ZN, Talame V, Deyholos M, Michalowski CB, Galbraith DW, Gozukirmizi N, Tuberosa R, Bohnert HJ (2002) Monitoring large-scale changes in transcript abundance in drought-and salt-stressed barley. Plant Mol Biol 48:551–573. doi:10.1023/A:1014875215580
Paget MS, Helmann JD (2003) The sigma70 family of sigma factors. Genome Biol 4(1):203–224. doi:10.1186/gb-2003-4-1-203
Passardi F, Longet D, Penel C, Dunand C (2004) The class III peroxidase multigenic in land plants family in rice and its evolution. Phytochemistry 65:1879–1893. doi:10.1016/j.phytochem.2004.06.023
Qin F, Sakuma Y, Li J, Liu Q, Li YQ, Shinozaki K, Yamaguchi-Shinozaki K (2004) Cloning and functional analysis of a novel DREB1/CBF transcription factor involved in cold-responsive gene expression in Zea mays L. Plant Cell Physiol 45:1042–1052. doi:10.1093/pcp/pch118
Seki M, Narusaka M, Abe H, Kasugac M, Yamaguchi-Shinozakic K, Carnincid P, Hayashizakid Y, Shinozaki K (2001) Monitoring the expression patterns of 1300 Arabidopsis genes under drought and cold stresses by using a full-length cDNA microarray. Plant Cell 13:61–72
Shinozaki K, Yamaguchi-Shinozaki K (2000) Molecular responses to dehydration and low temperature: differences and cross-talk between two stress signaling pathways. Curr Opin Plant Biol 3:217–223
Shinozaki K, Yamaguchi-Shinozaki K, Seki M (2003) Regulatory network of gene expression in the drought and cold stress responses. Curr Opin Plant Biol 6:410–417. doi:10.1016/S1369-5266(03)00092-X
Tran LS, Nakashima K, Sakuma Y, Simpson SD, Fujita Y, Maruyama K, Fujita M, Seki M, Shinozaki K, Yamaguchi-Shinozaki K (2004) Isolation and functional analysis of Arabidopsis stress inducible NAC transcription factors that bind to a drought responsive cis-element in the early responsive to dehydration stress 1 promoter. Plant Cell 16:2481–2498. doi:10.1105/tpc.104.022699
Vinocur B, Altman A (2005) Recent advances in engineering plant tolerance to abiotic stress: achievements and limitations. Curr Opin Biotechnol 16:123–132. doi:10.1016/j.copbio.2005.02.001
Yu J et al (2005) The genomes of Oryza sativa: a history of duplications. PLoS Biol 3:e38. doi:10.1371/journal.pbio.0030038
Zhou JL, Wang XF, Jiao YL, Qin YH, Liu XG, He K, Chen C, Ma LG, Wang J, Xiong LZ, Zhang QF, Fan LM, Deng XW (2007) Global genome expression analysis of rice in response to drought and high-salinity stresses in shoot, flag leaf, and panicle. Plant Mol Biol 63:591–608. doi:10.1007/s11103-006-9111-1
Acknowledgments
This work was supported in part by the Innovation Grant of the Chinese Academy of Sciences (No. 0551033) and the National Nature Science Foundation of China (Grant no. 30671273). During the preparation of this paper helpful comments were provided by Dr. Xia Xin-jie and Dr. Pedro Rocha (Institute of Subtropical Agriculture, The Chinese Academy of Sciences).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Communicated by M. Stobiecki.
Electronic supplementary material
Below is the link to the electronic supplementary material.
11738_2009_291_MOESM2_ESM.doc
Table S2 GO analysis of overlap genes regulated by three osmotic stress treatments in rice root under microarray data (DOC 108 kb)
Rights and permissions
About this article
Cite this article
Ma, Tc., Chen, Rj., Yu, Rr. et al. Differential global genomic changes in rice root in response to low-, middle-, and high-osmotic stresses. Acta Physiol Plant 31, 773–785 (2009). https://doi.org/10.1007/s11738-009-0291-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11738-009-0291-6