Abstract
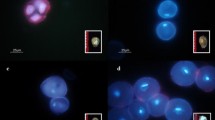
The aim of the research was to make a preliminary determination of the effectiveness of the induction of haploids in Capsicum frutescens L. In order to induce androgenesis red and yellow fruit forms of species were used, each bred by the researchers on their own. The experiment was performed in October. Anther cultures were conducted according to a modified method developed by Dumas et al. (1981) for C. annuum L. The anthers were laid on CP medium containing 0.01 mg dm−3 2.4-D and 0.01 mg dm−3 kinetin, with the addition of 0.5 g dm−3 of activated carbon and 5 mg×dm−3 of silver nitrate, solidified with 8 g dm−3 of agar. The cultures were incubated in the dark at 35 deg C for 8 days. Next they were transferred to 25 deg C under a 12-hour photoperiod. After 14 days of induction, anthers were transferred to R1 medium supplemented with 0.1 mg dm−3 kinetin. Obtained embryos were subsequently transplanted onto V3 hormone-free medium and well growing plants were planted in greenhouses. The efficiency of androgenesis for both C. frutescens L. forms was relatively low and it did not exceed 5%. The ploidy level of the resulting plants was determined by flow-cytometric analysis. The regenerants consisted of about equal numbers of haploids and diploids. Additionally, among plants regenerated from anthers of yellow fruit forms, two mixoploids were observed.
Similar content being viewed by others
References
Alegbejo M. D., Orakwue F. C. 2002. Characteristics of some pepper cultivars commonly grown in Nigeria. Capsicum and Eggplant Newsletter, 21: 22–24.
Carvalho S. I. C., Bianchetti L. B., Henz G. P. 2003. Germplasm collection of Capsicum spp. maintained by EMBRAPA Hortalicas. Capsicum and Eggplant Newsletter, 22: 17–20.
Dumas de Vaulx R., Chambonnet D., Pochard E. 1981. Culture in vitro d’anthéres de piment (Capsicum annuum L.) amélioration des taux d’obtention de plantes chez différents génotypes par des traitements á +35 °C. Agronomie, 1: 859–864.
Dolcet-Sanjuan R., Claveria E., Huerta A. 1997. Androgenesis in Capsicum annuum L.: effect of carbohydrate and carbon dioxide enrichment. J. Amer. Soc. Hort. Sci., 122: 468–475.
Galbraith D. W., Harkins K. R., Maddox J. M., Ayres N. M., Sharma D. P., Firoozabady E. 1983. Rapid flow cytometry analysis of the cell cycle in intact plant tissues. Science, 220: 1049–1051.
Kristiansen K., Andersen S.B. 1993. Effects of donor plant, temperature, photoperiod and age on anther culture response of Capsicum annuum L. Euphytica, 67: 105–109.
Li C. L., Jiang Z. R. 1990. A new sweet pepper cultivar, Haihua 3, developed by anther culture. Acta Hort. Sinica, 17: 39–44.
Maheshwari S. C., Rashid A., Tyagi A. K. 1982. Haploids from pollen grains — retrospect and prospect. Amer. J. Bot., 69: 865–879.
Matsubara S., Yamamoto M., Jo M. H., Murakami K. 1998. Embryoid and callus formation from microspores by anther culture from July to November in pepper (Capsicum annuum L.). Scientific Reports of the Faculty of Agriculture Okayama University, 87: 117–122.
Mitykó J., Andrásfalvy A., Csillery G., Fári M. 1995. Anther-culture response in different genotypes and F1 hybrids of pepper (Capsicum annuum L.). Plant Breed., 114: 78–80.
Mitykó J., Fári M. 1997. Problems and results of doubled haploid plant production in pepper (Capsicum annuum L.) via anther- and microspore culture. Acta Hort., 447, ISHS 1997.
Nervo G., Carannante G., Azzimonti M. T., Rotino G. L. 1995. Use of anther culture method in pepper breeding: factors affecting plantlets production. M. Terzi et al. (eds.). Current Issues in Plant Molecular and Cellular Biology, 155–160.
Nowaczyk L., Nowaczyk P. 2004a. Soft-flesh fruits of Capsicum frutescens L. as a raw material for processing. Annales UMCS, Sec. E, 59, 4: 1823–1828.
Nowaczyk L., Nowaczyk P. 2004b. Hot and sweet soft-flesh hybrids between Capsicum annuum L. and Capsicum frutescens L. Acta Hort. (in press)
Nowaczyk P., Olszewska D., 1999. The differences of interspecific hybrid seeds size within Capsicum genus. Acta Biol. Crac., Vol. 41, 1,55.
Regner F. 1996. Anther and microspore culture in Capsicum. In Vitro Haploid Production in Higher Plants, 3: 77–89.
Sibi M., Dumas de Vaulx R., Chambonnet D. 1979. Obtention de plantes haploides par androgenése in vitro chez le piment (Capsicum annuum L.). Ann. Amélior. Plantes, 29: 583–606.
Sreelathakumary I., Rajmony L. 2003. Variability, heritability and genetic advance in bird pepper (Capsicum frutescens L.). Capsicum and Eggplant Newsletter, 22: 51–54.
Vagera J. 1990. Pepper (Capsicum ssp.): In Vitro Induction of Haploids. Biotech. In Agri. Forestry, 12: 374–392.
Vagera J., Havranek P. 1985. In vitro induction of androgenesis in Capsicum annuum L. and its genetic aspects. Biol. Plant., 27: 10–21.
Venczel G., Mitykó J. 1995. In vitro haploid research and practical utilisation in pepper (Capsicum annuum L.) preservation. Zöldségtermesztési Kutató Intézet Bulletinje, 27: 145–156.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Nowaczyk, P., Kisiała, A. & Olszewska, D. Induced androgenesis of Capsicum frutescens L.. Acta Physiol Plant 28, 35–39 (2006). https://doi.org/10.1007/s11738-006-0066-2
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11738-006-0066-2