Abstract
The purpose of this study was to determine the content of selected phenolic compounds in white mustard, buckwheat, spring barley, oat and rye grown under field conditions. Moreover, the allelopathic efficiency of these compounds was evaluated by sensitivity of Echinochloa crus-galli. The aromatic acids: trans-cinnamic, salicylic, ferulic, chlorogenic, p-hydroxybenzoic, protocatechuic, p-coumaric and vanillic were separated from crop plants by TLC and determined spectrophotometrically.
Differences in concentrations of analysed compounds were observed for most of the examined plant species. The highest concentration was noticed for cinnamic acid and ranged from 360 µg·g−1 DW in rye to 2770 µg·g−1 DW in spring barley. The relatively high concentration was noticed for ferulic acid (from 73.8 µg·g−1 DW in buckwheat to 1046 µg·g−1 DW in spring barley) and p-coumaric acid (from 50 µg·g−1 DW in oat to 1499 µg·g−1 DW in buckwheat). The observed differences in the phenolics content between two successive vegetation seasons can reflect the effect of abiotic and biotic environmental factors on the phenolics level in studied plants.
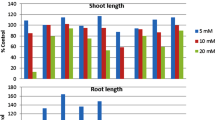
In the greenhouse experiment the effect of particular compounds on the growth of Echinochloa crus-galli was also studied. It has been found that the examined phenolics, and especially trans-cinnamic acid and mixture of phenolic compounds, significantly inhibit the growth of Echinochloa crus-galli. The obtained results may contribute to the explanation of the biological activity of some phenolic compounds.
Similar content being viewed by others
Abbreviations
- TLC:
-
thin — layer chromatography
- AE:
-
allelopathic efficiency
- x, x/2, x/4:
-
concentrations of the phenolic compounds
References
Bate N.J., Orr, J., Ni W., Merom A., Nadler-Hassar T., Doerner P.W., Dixon R.A., Lamb C.J., Elkind Y. 1994. Quantitative relationship between phenylalanine ammonia-lyase levels and phenylpropanoid accumulation in transgenic tobacco identifies a rate-determining step in natural product synthesis. In: Proceedings National Academy of Science, USA, 91: 7608–7612.
Blum U. 1999. Designing laboratory plant debris-soil bioassays: some reflections. In: Inderjit, Dakshini K.M.M., Foy C.L. (eds). Principles and practices in plant ecology: allelochemical interactions. CRC, Boca Raton, pp. 17–23.
Caldwell M.M., Robberecht R., Flint S.D. 1983. Internal filters: Prospects for UV-acclimation in higher plants. Physiol. Plant., 58: 445–450.
Delalonde M., Barret Y., Coumans M.P. 1996. Development of phenolic compounds in maize anthers (Zea mays) during cold pretreatment prior to androgenesis. J. Plant Physiol., 149: 612–616.
Duke S.O., Dayan F.E., Romagni J.G., Rimando A.M. 2000. Natural products as sources of herbicides: current status and future trends. Weed Res., 40: 99–111.
Duke S.O. 1985. Biosynthesis of phenolic compounds. Chemical manipulation in higher plants. In: Thompson A.C. (ed.). The chemistry of allelopathy: Biochemical interactions among plants. Am. Chem. Soc. Symp. Ser. 268. Am. Chem. Soc., Washington, DC., pp. 113–131.
Einhellig F.A. 1996. Interactions involving allelopathy in cropping system. Agr. J., 88: 886–893.
Einhellig F.A., Rasmussen J.A., Hejl A.M., Souza I.F. 1993. Effects of root exudate sorgoleone on photosynthesis. J. Chem. Ecol., 19: 369–375.
Gonzalez V.M., Kazimir J., Nimbal C., Weston L.A., Cheniae G.M. 1997. Inhibition of a photosystem II electron transfer reaction by the natural product sorgoleone. J. Agric. Food Chem., 45: 1415–1421.
Guenzi W.D., McCalla T.M. 1966. Phenolic acid in oats, wheat, sorghum, and corn residues and their phytotoxixity. Agr. J., 58: 303–304.
Guinn G., Eindenbock M.P. 1982. Catechin and condensed tannin contents of leaves and bolls of cotton in relation to irrigation and boll load. Crop Sci., 22: 614–616.
Hao Z., Charles D.J., Yu L., Simon J.E. 1999. Purification and characterization of a phenylalanine ammonia-lyase from Ocimum basilicum. Phytochem., 43: 735–739.
Harborne J.B. 1985. Phenolics and plant defence. Annu. Proc. Phytochem. Soc. Europe., 25: 395–408.
Horner J.D. 1990. Nonlinear effects of water deficits on foliar tannin concentration. Bioche. Syst. Ecol., 18: 211–213.
Inderjit 1996. Plant phenolics in allelopathy. Bot. Rev., 62: 186–202.
Inderjit, Duke S.O. 2003. Ecophysiological aspects of allelopathy. Planta, 217: 529–539.
Inderjit, Keating K.I. 1999. Allelopathy: principles, procedures, progress and promises for biological control. Adv. Agr., 67: 141–231.
Jones D.H. 1984. Phenylalanine ammonia-lyase: regulation of its induction and its role in plant development. Phytochem., 23: 1349–1359.
Mattice J., Dilday R.H., Gbur E.E., Skulman B.W. 2001. Inhibition of barnyardgrass growth with rice. Agr. J., 93: 8–11.
Olofsdotter M., Navarez D.C. 1996. Allelopathic rice for Echinochloa crus-galli control. In: Proceedings 2nd International Weed Control Congress, Copenhagen, Denmark, 1175–1181.
Olofsdotter M., Rebulanan M., Madrid A., Wang D.L., Navarez D., Olk D.C. 2002. Why phenolic acids are unlikely primary allelochemicals in rice. J. Chem. Ecol., 28: 229–242.
Piskorz B. 1997. Oddziaływanie allelopatyczne chwastnicy jednostronnej (Echinochloa crus-galli L.) na niektóre rośliny warzywne. I. Wpływ wodnych wyciagówz chwastnicy jednostronnej na kiełkowanie ogórka, pomidora i rzodkiewki. Zesz. Probl. Post. Nauk Roln., 452: 153–165.
Putnam A.R., De Frank J., Barnes J.P. 1983. Exploitation of allelopathy for weed control in annual and perennial cropping systems. J. Chem. Ecol., 9: 1001–1010.
Rice E.L. 1984. Allelopathy. Second Edition, Academic Press, New York, pp. 422.
Roll H. 1986. Yield of maize as related to the duration of Echinochloa crus-galli and Amaranthus retroflexus in maize stand. Pam. Puł., 87: 155–170.
Romagni J.G., Allen S.N., Dayan F.E. 2000. Allelopathic effects of volatile cineoles on two weedy plant species. J. Chem. Ecol., 26: 303–313.
Rösler J., Krekel F., Amrhein N., Schmid J. 1997. Maize phenylalanine ammonia-lyase has tyrosine ammonia-lyase activity. Plant Physiol., 113: 175–179.
Ruiz J.M., Bretones G., Baghour M., Belakbir A., Romero L. 1998. Relationship between boron and phenolic metabolism in tobacco leaves. Phytochem., 48: 269–272.
Ruiz J.M., Garcia P.C., Rivero R.M., Romero L. 1999. Response of phenolic metabolism to the application of carbendazim plus boron in tobacco. Physiol. Plant., 106: 151–157.
Schweiger J., Lang M., Lichtenthaler H.K. 1996. Differences in fluorescence excitation spectra of leaves between stressed and non-stressed plants. J. Plant Physiol., 148: 536–547.
Sène M., Dorè T., Gallet Ch. 2001. Relationship between biomass and phenolic production in grain sorghum grown under different conditions. Agr. J., 93: 49–54.
Smith G.D., Doan N.T. 1999. Cyanobacterial metabolites with bioactivity against photosynthesis in cyanobacteria, algae and higher plants. J. Appl. Phycol., 11: 337–344.
Stupnicka-Rodzynkiewicz E., Dubert F., Hochól T., Hura T., Lepiarczyk A., Stoklosa A. 2004. Possibility of application of plants allelopathy for weed control. (in Polish) Zesz. Probl. Post. Nauk Roln., 496: 343–355.
Tempel A.S. 1981. Field studies of the relationship between herbivore damage and tannin content in bracken (Pteridium aquilinum Kuhn.). Oecologia, 51: 97–106.
Yenish J.P., Worsham A.D., York A.C. 1996. Cover crops for herbicide replacement in no-tillage corn (Zea mays). Weed Technol., 10: 815–821.
Vangessel M.J., Renner K.A. 1990. Redroot pigweed (Amaranthus retroflexus) and barnzard grass (Echinochloa crus-galli) interference in potatoes (Solanum tuberosum). Weed Sci., 38: 338–343.
Wu H., Pratley J., Lemerle D., Haig T. 1999. Crop cultivars with allelopathic capability. Weed Res., 39: 171–180.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hura, T., Dubert, F., Dąbkowska, T. et al. Quantitative analysis of phenolics in selected crop species and biological activity of these compounds evaluated by sensitivity of Echinochloa crus-galli . Acta Physiol Plant 28, 537–545 (2006). https://doi.org/10.1007/s11738-006-0049-3
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11738-006-0049-3