Abstract
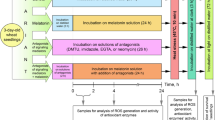
We studied the alterations in phenolic compounds in grape seeds during their stratification and germination under optimal conditions (+25 °C) and at low temperature (+10 °C). Biological materials in the study were seeds of Vitis riparia. Phenolic compounds were extracted from defatted seeds using 80 % methanol or 80 % acetone. The content of total phenolics was determined with the Folin-Ciocalteau reagent, while the content of tannins was determined by vanillin assay and the protein (BSA) precipitation method. The RP-HPLC method was used to determine phenolic compounds (phenolic acids, catechins) in the extracts. High amounts of tannins, catechins, gallic acid and lesser amounts of p-coumaric acid were found in the seeds. The content of total phenolics in acetone extracts was higher than that obtained using methanol. The amounts of phenolic acids and tannins found in V. riparia seeds after stratification were much lower. It may confirm a possible role of these compounds in dormancy of V. riparia seeds. After 72 h of low temperature treatment, inhibition of grape root growth and biochemical changes in seeds were detected. The chilling stimulated increased accumulation of some phenolic compounds (free gallic acid and catechins) in the seeds. These substances can protect plants against some abiotic stressors.
Similar content being viewed by others
References
Amarowicz R., Piskuła M., Honke J., Rudnicka B., Troszy ska A., Kozłowska H. 1995. Extraction of phenolic compounds from lentil seeds (Lens culinaris) with various solvents, Pol. J. Food Nutr. Sci. 4/85: 53–62.
Amarowicz R., Weidner S. 2001. Content of phenolic acids in rye caryopses determined using DAD-HPLC method. Czech J. Food Sci. 19: 201–203.
Balthazard J. 1969. Températures alternées, longueur des embryons et pouvoir germinatif des graines de Vigne. Comptes Rendus de l’Académie des Sciences de Paris, Serie D 269: 2355–2358.
Baskin C.C., Baskin J.M. 1998. Seeds. Ecology, biogeography, and evolution of dormancy and germination. Acad. Press. New York.
Bewley J.D., Black M. 1994. Seeds-physiology of development and germination. Plenum Press, New York and London.
Bray E.A., Bailey-Serres J., Weretilnyk E. 2000. Responses to Abiotic Stresses. Chapter 22. In: Responses to Abiotic stresses. Buchannan B., Jones R. (Eds). American Society of Plant Physiologists, Rockville, MD: 1158–1249.
Bugnon F., Bessis R. 1968. Biologie de la Vigne. Acquisitions récentes et problémes acutels. Paris: Masson et Cie.
Dixon R.D., Paiva N.L. 1995. Stress-induced phenylpropanoid metabolism. Plant Cell 7: 1085–1097.
Hagerman A., Butler L. 1978. Protein precipitation method for quantitative determination of tannins. J. Agric. Food Chem., 26: 809–811.
Janas K.M., Cvikrova M., Pałagiewicz A., Eder J. 2000. Alternations in phenylpropanoid content in soybean roots during low temperature acclimation. Plant Physiol. Biochem. 38: 587–593.
Janas K.M., Cvikrova M., Pał giewicz A., Szafra-ska K., Posmyk M.M. 2002. Constitutive elevated accumulation of phenylpropanoids in soybean roots at low temperature. Plant Sci. 163: 369–373
Jayaprakasha G.K., Selvi T., Sakariah K.K. 2003. Antibacterial and antioxidant activities of grape (Vitis vinifera) seeds extracts. Food Res. Inter. 36: 117–122.
Jayaprakasha G.K., Singh R.P., Sakariah K.K. 2001. Antioxidant activity of grape seed (Vitis vinifera) extracts on peroxidation models in vitro. Food Chem., 73: 285–290.
Kennedy J.A., Troup G.J., Pilbrow J.R., Hutton D.R., Hewitt D., Hunter C.R., Ristic R., Iland P.G., Jones G.P. 2000. Development of seed polyphenols in berries from Vitis vinifera L. cv. Shiraz. Aust. J. Grape Wine Res. 6: 244–254.
Kratsch H.A., Wise R.R. 2000. The ultrastructure of chilling stress. Plant Cell Envir. 23: 337–350.
Krygier K., Sosulski F.W., Hogge L. 1982. Free, esterified, and insoluble-bound phenolic acids. 1. Extraction and purification procedure. J. Agric. Food Chem. 30: 330–334.
Naczk M., Shahidi F. 1989. The effect of methanol-ammonia-water treatment on the content of phenolic acids of canola. Food Chem. 31: 15–164.
Nikolayeva M.G. 1977. Factors controlling the seeds dormancy pattern. In: The physiology and biochemistry of seed dormancy and germination, Khan A.A. (Ed) Amsterdam: North-Holland: 51–74.
Oszmia ski J., Lamer-Zarawska E. 1993. Wyst - powanie i znaczenie tanin w ro linach leczniczych. Wiad. Zielar., 35: 16–17.
Oszmia ski J., Bourzeix M. 1995. Preparation of catechin and procyanidin standards from hawthorn (Crataegus azarolus L.) and pine (Pine mesogeensis fieschi) barks. Pol. J. Food. Nutr. Sci. 4/45: 89–96.
Peki B., Kova V., Alonso E., Revilla E. 1998. Study of extraction of proanthocyanidins from grape seeds. Food Chem., 61: 201–206.
Price N.J., Van Scoyoc S., Butler L.G. 1978. A critical evaluation of the vanillic reactions an assay for tannin in sorghum grain. J. Agric. Food Chem. 26: 1214–1218.
Rhodes M.J.C., Wooltorton L.S.C. 1977. Changes in tomatoes stored at low temperatures. Trends Plant Sci. 2: 152–159.
Rivero R.M., Ruiz J.M., Garcia P.C., Lopez-Lefebre L.R., Sanchez E., Romero L. 2001. Resistance to cold and heat stress: accumulation of phenolic compounds in tomato and watermelon plants. Plant Sci. 160: 315–321.
Saito M., Hosoyama H., Ariga T., Kataoka S., Yamaji N. 1998. Antiulcer Activity of grape seed extract and procyanidins. J. Agric. Food Chem., 46: 1460–1464.
Shahidi F. 1997. Natural antioxidants: chemistry, health effect, and applications. AOCS Press, Champaign, Illinois.
Sokół-Ł towska A. 1997. Próby opracowania i zastosowania preparatów zwi zków fenolowych wybranych surowców ro linnych jako przeciwutleniaczy. Zeszyty Naukowe Akademii Rolniczej we Wrocławiu. Technologia ywno ci XI, 319: 99–115.
Solecka D. 1997. Role of phenylpropanoid compounds in plant responses to different stress factors. Acta Physiol. Plant 19: 257–268.
Solecka D., Kacperska A. 2003. Phenylpropanoid dificency affects the course of plant acclimation to cold. Physiol. Plant. 119: 253–262.
Soquet J.M., Cheynier V., Brossaud F., Moutounet M. 1996. Polymeric proanthocyanidins from grape skins. Phytochemistry 43: 509–512.
Sun B., Leandro C., da Silva J.M.R., Spranger I. 1998. Separation of grape and wine proanthocyanidins according to their degree of polymerization. J. Agric. Food Chem., 46: 1390–1396.
Weidner S., Amarowicz R., Karama M., Fr czek E. 2000. Changes in endogenous phenolic acids during development of Secale cereale caryopses and after dehydration treatment of unripe rye grains. Plant Physiol. Biochem., 38: 595–602.
Weidner S., Fr czek E., Amarowicz R., Abe S. 2001. Alterations in phenolic acids content in developing rye grains in normal environment and during enforcet dehydration. Acta Physiol. Plant. 23: 475–482.
Weidner S., Krupa U., Amarowicz R., Karama M., Abe S. 2002. Phenolic compounds in embryos of triticale caryopses at different stages of development and maturation in normal environment and after dehydration treatment. Euphytica, 126: 115–112.
Weidner S., Paprocka J., Kamieniecki B., Zadernowski R. 1993. The role of phenolic acids in dormancy of barley caryopses. In Pre-Harvest Sprouting in cereals 1992, ed by M.K. Walker-Simmons and J.L. Ried. American Association of Cereal Chemists. St. Paul, Minnesota, USA.
Zadernowski R., Kozłowska H. 1983. Phenolic acids in soybean and rapeseed flours. Lebensm. Wiss. Technol. 16: 110–114.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wróbel, M., Karama, M., Amarowicz, R. et al. Metabolism of phenolic compounds in Vitis riparia seeds during stratification and during germination under optimal and low temperature stress conditions. Acta Physiol Plant 27, 313–320 (2005). https://doi.org/10.1007/s11738-005-0008-4
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11738-005-0008-4