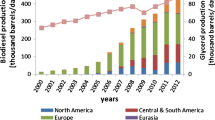
Abstract
Thermodynamic chemical equilibrium analysis of steam reforming of glycerol (SRG) for selective hydrogen production was performed based on the Gibbs free energy minimisation method. The ideal SRG reaction (C3H8O3+ 3H2O → 3CO2+7H2) and a comprehensive set of side reactions during SRG are considered for the formation of a wide range of products. Specifically, this work focused on the analysis of formation of CO2, CO and CH4 in the gas phase and determination of the carbon free region in SRG under the conditions at atmospheric pressure, 600 K–1100 K and 1.013 × 105–1.013 × 106 Pa with the steam-to-glycerol feed ratios (SGFR) of 1:5–10. The reaction conditions which favoured SRG for H2 production with minimum coke formation were identifies as: atmospheric pressure, temperatures of 900 K–1050 K and SGFR of 10:1. The influence of using the inert carrier gas (i.e., N2) in SRG was studied as well at atmospheric pressure. Although the presence of N2 in the stream decreased the partial pressure of reactants, it was beneficial to improve the equilibrium yield of H2. Under both conditions of SRG (with/without inert gas), the CH4 production is minimised, and carbon formation was thermodynamically unfavoured at steam rich conditions of SGFR > 5:1.

Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Papageridis K N, Siakavelas G, Charisiou N D, Avraam D G, Tzounis L, Kousi K, Goula M A. Comparative study of Ni, Co, Cu supported on γ-alumina catalysts for hydrogen production via the glycerol steam reforming reaction. Fuel Processing Technology, 2016, 152: 156–175
Zhou C H, Zhao H, Tong D S, Wu L M, Yu W H. Recent advances in catalytic conversion of glycerol. Catalysis Reviews, 2013, 55(4): 369–453
Buffoni I N, Pompeo F, Santori G F, Nichio N N. Nickel catalysts applied in steam reforming of glycerol for hydrogen production. Catalysis Communications, 2009, 10(13): 1656–1660
Wang W. Thermodynamic analysis of glycerol partial oxidation for hydrogen production. Fuel Processing Technology, 2010, 91(11): 1401–1408
Yang G, Yu H, Peng F, Wang H, Yang J, Xie D. Thermodynamic analysis of hydrogen generation via oxidative steam reforming of glycerol. Renewable Energy, 2011, 36(8): 2120–2127
Avasthi K S, Reddy R N, Patel S. Challenges in the production of hydrogen from glycerol—a biodiesel byproduct via steam reforming process. Procedia Engineering, 2013, 51: 423–429
Schwengber C A, Alves H J, Schaffner R A, da Silva F A, Sequinel R, Bach V R, Ferracin R J. Overview of glycerol reforming for hydrogen production. Renewable & Sustainable Energy Reviews, 2016, 58: 259–266
Dou B, Song Y, Wang C, Chen H, Xu Y. Hydrogen production from catalytic steam reforming of biodiesel byproduct glycerol: issues and challenges. Renewable & Sustainable Energy Reviews, 2014, 30: 950–960
Bagnato G, Iulianelli A, Sanna A, Basile A. Glycerol production and transformation: a critical review with particular emphasis on glycerol reforming reaction for producing hydrogen in conventional and membrane reactors. Membranes, 2017, 7(2): 17
Roslan N A, Abidin S Z, Ideris A, Vo D V N. A review on glycerol reforming processes over Ni-based catalyst for hydrogen and syngas productions. International Journal of Hydrogen Energy, 2020, 45(36): 18466–18489
Chen H, Ding Y, Cong N T, Dou B, Dupont V, Ghadiri M, Williams P T. A comparative study on hydrogen production from steamglycerol reforming: thermodynamics and experimental. Renewable Energy, 2011, 36(2): 779–788
Silva J M, Soria M A, Madeira L M. Thermodynamic analysis of glycerol steam reforming for hydrogen production with in situ hydrogen and carbon dioxide separation. Journal of Power Sources, 2015, 273: 423–430
Lin K H, Lin W H, Hsiao C H, Chang H F, Chang A C C. Hydrogen production in steam reforming of glycerol by conventional and membrane reactors. International Journal of Hydrogen Energy, 2012, 37(18): 13770–13776
Iulianelli A, Seelam P K, Liguori S, Longo T, Keiski R, Calabrò V, Basile A. Hydrogen production for PEM fuel cell by gas phase reforming of glycerol as byproduct of bio-diesel. The use of a Pd-Ag membrane reactor at middle reaction temperature. International Journal of Hydrogen Energy, 2011, 36(6): 3827–3834
Silva J M, Soria M A, Madeira L M. Challenges and strategies for optimization of glycerol steam reforming process. Renewable & Sustainable Energy Reviews, 2015, 42: 1187–1213
Adhikari S, Fernando S D, Haryanto A. Hydrogen production from glycerin by steam reforming over nickel catalysts. Renewable Energy, 2008, 33(5): 1097–1100
Adhikari S, Fernando S, Gwaltney S, Filipto S, Markbricka R, Steele P, Haryanto A. A thermodynamic analysis of hydrogen production by steam reforming of glycerol. International Journal of Hydrogen Energy, 2007, 32(14): 2875–2880
Wang X, Li M, Wang M, Wang H, Li S, Wang S, Ma X. Thermodynamic analysis of glycerol dry reforming for hydrogen and synthesis gas production. Fuel, 2009, 88(11): 2148–2153
Wang H, Wang X, Li M, Li S, Wang S, Ma X. Thermodynamic analysis of hydrogen production from glycerol autothermal reforming. International Journal of Hydrogen Energy, 2009, 34(14): 5683–5690
Li J, Yu H, Yang G, Peng F, Xie D, Wang H, Yang J. Steam reforming of oxygenate fuels for hydrogen production: a thermodynamic study. Energy & Fuels, 2011, 25(6): 2643–2650
Fasolini A, Cespi D, Tabanelli T, Cucciniello R, Cavani F. Hydrogen from renewables: a case study of glycerol reforming. Catalysts, 2019, 9(9): 772
Smith J M, Van Ness H C, Abbott M M. Introduction to Chemical Engineering Thermodynamics. 7th ed. New York: McGraw-Hill, 2005, 450–508
Tahir M, Mulewa W, Amin N A S, Zakaria Z Y. Thermodynamic and experimental analysis on ethanol steam reforming for hydrogen production over Ni-modified TiO2/MMT nanoclay catalyst. Energy Conversion and Management, 2017, 154: 25–37
ChemCAD process simulator. Version 6.5.6. Houston, TX: Chemstations, Inc., 2012
Nikoo M K, Amin N A S. Thermodynamic analysis of carbon dioxide reforming of methane in view of solid carbon formation. Fuel Processing Technology, 2011, 92(3): 678–691
Cheng C K, Foo S Y, Adesina A A. Thermodynamic analysis of glycerol-steam reforming in the presence of CO2 or H2 as carbon gasifying agent. International Journal of Hydrogen Energy, 2012, 37(13): 10101–10110
Charisiou N D, Papageridis K N, Siakavelas G, Tzounis L, Kousi K, Baker M A, Hinder S J, Sebastian V, Polychronopoulou K, Goula M A. Glycerol steam reforming for hydrogen production over nickel supported on alumina, zirconia and silica catalysts. Topics in Catalysis, 2017, 60(15–16): 1226–1250
Charisiou N D, Polychronopoulou K, Asif A, Goula M A. The potential of glycerol and phenol towards H2 production using steam reforming reaction: a review. Surface and Coatings Technology, 2018, 352: 92–111
Cantelo R C. The thermal decomposition of methane. Journal of Physical Chemistry, 1924, 28(10): 1036–1048
Gallo A, Pirovano C, Ferrini P, Marelli M, Psaro R, Santangelo S, Faggio G, Dal Santo V. Influence of reaction parameters on the activity of ruthenium based catalysts for glycerol steam reforming. Applied Catalysis B: Environmental, 2012, 121–122: 40–49
Sundari R, Vaidya P D. Reaction kinetics of glycerol steam reforming using a Ru/Al2O3 catalyst. Energy & Fuels, 2012, 26(7): 4195–4204
Acknowledgements
This project has received funding from European Union’s Horizon 2020 research and innovation programme under grant agreement No. 872102. Ammaru Ismaila thanks the financial support by the Petroleum Technology Development Fund (PTDF) Nigeria for his PhD research at The University of Manchester (File No. PTDF/ED/OSS/PHD/IA/1209/17).
Author information
Authors and Affiliations
Corresponding authors
Electronic Supplementary Material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ismaila, A., Chen, X., Gao, X. et al. Thermodynamic analysis of steam reforming of glycerol for hydrogen production at atmospheric pressure. Front. Chem. Sci. Eng. 15, 60–71 (2021). https://doi.org/10.1007/s11705-020-1975-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11705-020-1975-0