Abstract
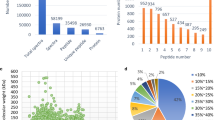
Apple is one of the most important fruit trees in the world. Nevertheless, mainly due to its long juvenile period, its breeding work constantly falls far behind other crops. So the aim of this study is to reveal the mechanism of apple flower bud differentiation, shorten the juvenile period and accelerate its breeding process. Proteomics technology (including two-dimensional gel electrophoresis (2-DE), biomass spectrometry and bioinformatics) was applied to work on the specific protein of flower bud and leaf bud after the brachyblasts of ‘Fuji’ stopped growth for 3–9 weeks. The results showed that the morphodifferentiation of flower bud did not begin until the seventh week after the brachyblast stopped growth. Furthermore, compared with the leaf bud, flower bud had significant changes in the expression of 283 protein spots in quality and quantity on 2-DE maps. Among the 283 protein spots, four protein spots (16.4, 30.2, 40.3 and 65.1 kD) were characteristic of the flower bud in the archae-stage (initial inflorescence appeared) at the beginning of flower-bud differentiation, three (39.3, 60.2 and 66.3 kD) in the post-stage (Lateral-flower appears) and one (77.1 kD) in the sepal stage on 2-DE maps. Analysis by peptide mass fingerprinting and matrix-assisted laser desorption ionization time of flight mass spectrometry also identified and forecasted functionally by blasting different databases. In the four specific proteins, it was found that spots No. 256 (16.4 kD) and 298 (30.2 kD) were unknown proteins, spot Nos. 327 (40.3 kD) was identified as the synthesis enzyme protein and spot No. 367 (40.3 kD) was identified as a RNA-binding protein involved in transcription. When flower bud started to differentiate morphologically, we detected four specific proteins which were 16.4, 30.2, 40.3 and 65.1 kD. Three specific proteins 39.3, 60.2 and 66.3 kD were observed at side flower-appearing stage. When calyx began to emerge, there was one specific protein: 77.1 kD. The proteins 16.4 kD and 30.2 kD were two unknown specific proteins. The 40.3 kD protein was related to methionine synthetase while 65.1 kD protein, a RNA-binding protein, was related to transcription.
Similar content being viewed by others
References
Anderson L, Seilhamer J (1997). A comparison of selected mRNA and protein abundances in human. Electrophoresis, 18: 533
Bevan M, Bancroft I, Bent E, et al. (1998). Analysis of 1.9 Mb of contiguous sequence from chromosome 4 of Arabidopsis thaliana. Nature, 391: 485–488
Cao S Y, Tang Y Z, Jiang A H (2001). Effects of PP333 and GA3 on the mechanism of flower bud induction in apple tree. Acta Horticulturae Sinica, 28: 339–341 (in Chinese)
Cao S Y, Zhang J M, Wei L H (2000). Studies on the changes of endogenous hormones in the differentiation period of flower bud in apple trees. Journal of Fruit Science, 17: 244–248 (in Chinese)
Cao S Y, Zhang Q M, Guo J Y, Chen Y L (2005). Progress in proteomics technology and its application on fruit tree. Journal of Fruit Science, 22: 345–350 (in Chinese)
Cao S Y, Zhang Q M, Wu S (2003). Advances in research on the mechanism of flower-bud differentiation of fruit trees. Journal of Fruit Science, 20: 345–350 (in Chinese)
Chen W, Huang C M, Lu L X (2001). The improved method of twodimensional electrophoresis of protein in recalcitrant plant litchi. Journal of Fujian Agricultural University (Natural Science), 30: 123–126 (in Chinese)
Courchesne P L, Patterson S D (1999). Identification of proteins by matrix-assisted laserdesorption/ionization mass spectrometry using peptide and fragment ion masses. In: Link AJ ed. 2-D Proteome Analysis Protocols. New Jersey, USA: Humana Press, 487–511
Gygi S P, Bochon Y, Franza B R (1999). Correlation between protein and mRNA abundance in yeast. Mol Cell Biol, 19: 1720–1730
Leandro P, Martin T M, Juaraz G (2001). Constitutive expression of Arabidopsis LEAFY or APETALA1, genes in citrus reduces their generation time. Bio Nature, 19(3): 263–267
Meyerowitz E M (1998). Genetic and molecular mechanisms of pattern formation in Arabidopsis flower development. J Plant Res, 111: 233–242
Sung S K, An G (1997). Molecular cloning and characterization of a MADS-box Cdna clone of the Fuji apple. Plant Cell Physiol, 38: 484–489
Sung S K, Yu G H, An G (1999). Characterization of MdMADS2, a member of the SOUAMOSA subfamily of genes in apple. Plant Physiol, 120: 969–978
Sung S K, Yu G H, Nam J, Jeong D H, An G (2000). Developmentally regulated expression of two MADS-box genes, MdMADS3 and MdMADS4, in the morphogenesis of flower buds and fruits in apple. Planta, 210: 519–528
Watson B S, Asirvatham V S, Wang L J (2003). Mapping the proteome of barrel medic (Medicagotruncatula). Plant Physiol, 131: 1104–1123
Weigel D (1995). The genetics of flower development: from induction to ovule morphogenesis. Annu Rer Genet, 29: 19–39
Weigel D, Nilsson O (1995). A developmental switch sufficient for flower initiation in diverse plants. Nature, 377: 495–500
Wilkins V C, Williams S J, Cerpa P A (1995). Progress with geneproduct mapping of the Mollicutes: Mycoplasma genitalium. Electrophoresis, 16: 1090–1094
Yao J L, Dong Y H, Morris B A M (2001). Parthenocarpic apple fruit production conferred by transposon insertion mutations in a MADS-box transcription factor. Proc Natl Acad Sci USA, 98(3): 1306–1311
Author information
Authors and Affiliations
Corresponding author
Additional information
__________
Translated from Scientia Agricultura Sinica, 2007, 40(10): 2281–2288 [译自 : 中国农业科学]
About this article
Cite this article
Cao, S., Zhang, Q., Zhu, Z. et al. Preliminary proteomics analysis of the total proteins of flower bud induction of apple trees. Front. Agric. China 2, 467–473 (2008). https://doi.org/10.1007/s11703-008-0076-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11703-008-0076-5