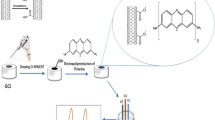
Abstract
The work herein describes the electrochemical detection of heavy metal ions, specifically cadmium and lead. The introduction and modification of functional groups such as Schiff bases leads to an enhanced sensitivity of the electrode to analytes. In this study, a platinum electrode has, for the first time, been modified with poly(3,4-ethylenedioxythiophene) (PEDOT/Schiff base) in CH2Cl2 containing Bu4NPF6 to detect cadmium(II) and lead(II) ions. The structures and morphologies of the polymer coatings were characterised via Fourier transform infrared spectroscopy and scanning electron microscopy, respectively. The electrochemical synthesis and redox state response in monomer-free synthesised films have been studied via cyclic voltammetry. Moreover, the effect of scan rate on the electrochemical behaviour of the modified electrodes was also studied. The voltammetric findings have been used to calculate the surface coverage required for the polymer films and the stability of polymer electrodes in the monomer-free solutions. Square wave voltammetry was applied for the determination of cadmium(II) and lead(II) ion concentrations and to assess the effects of pH on aqueous samples. The limits of detection for the modified electrode for cadmium(II) and lead(II) were found to be 0.95 μg L−1 and 1.84 μg L−1, respectively. These findings revealed that modified films can be considered good candidates for application in electrochemical detection devices.











taken from Fig. 9 using a poly(EDOT/ Schiff) electrode



Similar content being viewed by others
References
Abd Ali LI, Ismail HK, Aboul-Enein HY (2021) A nanocomposite based on polyaniline, nickel and manganese oxides for dye removal from aqueous solutions. Int J Environ Sci Technol 18:2031–2050. https://doi.org/10.1007/s13762-020-02961-0
Alesary HF, Ismail HK, Khudhair AF, Mohammed MQ (2018) Effects of dopant ions on the properties of polyaniline conducting polymer. Orient J Chem 34:2525–2533. https://doi.org/10.13005/ojc/340539
Alesary HF, Ismail HK, Mohammed MQ, Mohammed HN, Abbas ZK, Barton S (2021) A comparative study of the effect of organic dopant ions on the electrochemical and chemical synthesis of the conducting polymers polyaniline, poly(o-toluidine) and poly(o-methoxyaniline). Chem Pap 75:5087–5101. https://doi.org/10.1007/s11696-020-01477-8
Almashal FA, Mohammed MQ, Hassan QMA, Emshary CA, Sultan HA, Dhumad AM (2020) Spectroscopic and thermal nonlinearity study of a Schiff base compound. Opt Mater (amst) 100:109703. https://doi.org/10.1016/j.optmat.2020.109703
Barsan MM, Ghica ME, Brett CMA (2015) Electrochemical sensors and biosensors based on redox polymer/carbon nanotube modified electrodes: a review. Anal Chim Acta 881:1–23. https://doi.org/10.1016/j.aca.2015.02.059
Bidan G (1992) Electroconducting conjugated polymers: new sensitive matrices to build up chemical or electrochemical sensors. A review. Sens Actuators B Chem 6:45–56. https://doi.org/10.1016/0925-4005(92)80029-W
Bobacka J (2006) Conducting polymer-based solid-state ion-selective electrodes. Electroanal Int J Devoted Fundam Pract Asp Electroanal 18:7–18. https://doi.org/10.1002/elan.200503384
Bozzini B, Bocchetta P, Alemán B, Amati M, Gianoncelli A, Gregoratti L, Sezen H, Taurino A, Kiskinova M (2015) Electrodeposition and pyrolysis of Mn/polypyrrole nanocomposites: a study based on soft X ray absorption, fluorescence and photoelectron microspectroscopies. J Mater Chem A 3:19155–19167. https://doi.org/10.1039/C5TA05572E
Brown RM, Hillman AR (2012) Electrochromic enhancement of latent fingerprints by poly(3,4-ethylenedioxythiophene). Phys Chem Chem Phys 14:8653–8661. https://doi.org/10.1039/C2CP40733G
Dang VH, Yen PTH, Giao NQ et al (2018) A versatile carbon fiber cloth-supported Au nanodendrite sensor for simultaneous determination of Cu(II), Pb(II) and Hg(II). Electroanalysis 30:2222–2227. https://doi.org/10.1002/elan.201800332
Darmanin T, Guittard F (2013) Superoleophobic surfaces with short fluorinated chains. Soft Matter 9:5982–5990. https://doi.org/10.1039/C3SM50643F
Deshmukh MA, Celiesiute R, Ramanaviciene A, Shirsat MD, Ramanavicius A (2018) EDTA_PANI/SWCNTs nanocomposite modified electrode for electrochemical determination of copper(II), lead(II) and mercury(II) ions. Electrochim Acta 259:930–938. https://doi.org/10.1016/j.electacta.2017.10.131
Duan C, Liu Z, Yuan L, Zhu H, Luo H, Yan K (2020) PEDOT:PSS-metal oxide composite electrode with regulated wettability and work function for high-performance inverted perovskite solar cells. Adv Opt Mater 8:2000216. https://doi.org/10.1002/adom.202000216
Etorki AM, Hillman AR, Ryder KS, Glidle A (2007) Quartz crystal microbalance determination of trace metal ions in solution. J Electroanal Chem 599:275–287. https://doi.org/10.1016/j.jelechem.2006.03.020
Fourou H, Zazoua A, Braiek M, Jaffrezic-Renault N (2016) An enzyme biosensor based on beta-galactosidase inhibition for electrochemical detection of cadium(II) and chromium(VI). Int J Environ Anal Chem 96:872–885. https://doi.org/10.1080/03067319.2016.1209659
Guadalupe AR, Abruna HD (1985) Electroanalysis with chemically modified electrodes. Anal Chem 57:142–149. https://doi.org/10.1021/ac00279a036
Gumpu MB, Sethuraman S, Krishnan UM, Rayappan JBB (2015) A review on detection of heavy metal ions in water–an electrochemical approach. Sens Actuators B Chem 213:515–533. https://doi.org/10.1016/j.snb.2015.02.122
Han J, Yu J, Guo Y, Wang L, Song Y (2020) COFBTLP-1/three-dimensional macroporous carbon electrode for simultaneous electrochemical detection of Cd2+, Pb2+, Cu2+ and Hg2+. Sens Actuators B Chem 321:128498. https://doi.org/10.1016/j.snb.2020.128498
Hassan KM, Gaber SE, Altahan MF, Azzem MA (2018) Novel sensor based on poly(1,2-diaminoanthraquinone) for individual and simultaneous anodic stripping voltammetry of Cd2+, Pb2+, Cu2+ and Hg2+. Electroanalysis 30:1155–1162. https://doi.org/10.1002/elan.201800097
Hassan KM, Elhaddad GM, AbdelAzzem M (2019) Voltammetric determination of cadmium(II), lead(II) and copper(II) with a glassy carbon electrode modified with silver nanoparticles deposited on poly(1,8-diaminonaphthalene). Microchim Acta 186:440. https://doi.org/10.1007/s00604-019-3552-0
He B, Shen XF, Nie J et al (2018) Electrochemical sensor using graphene/Fe3O4 nanosheets functionalized with garlic extract for the detection of lead ion. J Solid State Electrochem 22:3515–3525. https://doi.org/10.1007/s10008-018-4041-9
Hillman AR, Ryder KS, Ismail HK, Unala A, Voorhaara A (2017) Fundamental aspects of electrochemically controlled wetting of nanoscale composite materials. Faraday Discuss 199:75–99. https://doi.org/10.1039/C7FD00060J
Huang H, Zhu W, Gao X, Liu X, Ma H (2016) Synthesis of a novel electrode material containing phytic acid-polyaniline nanofibers for simultaneous determination of cadmium and lead ions. Anal Chim Acta 947:32–41. https://doi.org/10.1016/j.aca.2016.10.012
Intarakamhang S, Schuhmann W, Schulte A (2013) Robotic heavy metal anodic stripping voltammetry: ease and efficacy for trace lead and cadmium electroanalysis. J Solid State Electrochem 17:1535–1542. https://doi.org/10.1007/s10008-013-2018-2
Ismail HK, Alesary HF, Al-Murshedi AY, Kareem JH (2019a) Ion and solvent transfer of polyaniline films electrodeposited from deep eutectic solvents via EQCM. J Solid State Electrochem 23:3107–3121. https://doi.org/10.1007/s10008-019-04415-1
Ismail HK, Alesary HF, Mohammed MQ (2019b) Synthesis and characterisation of polyaniline and/or MoO2/graphite composites from deep eutectic solvents via chemical polymerization. J Polym Res 26:65. https://doi.org/10.1007/s10965-019-1732-6
Lattach Y, Fourati N, Zerrouki C, Fougnion JM, Garnier F, Pernelle C, Remit S (2012) Molecularly imprinted surface acoustic wave sensors: the synergy of electrochemical and gravimetric transductions in chemical recognition processes. Electrochim Acta 73:36–44. https://doi.org/10.1016/j.electacta.2011.11.119
Lei W, Si W, Xu Y, Gu Z, Hao Q (2014) Conducting polymer composites with graphene for use in chemical sensors and biosensors. Microchim Acta 181:707–722. https://doi.org/10.1007/s00604-014-1160-6
Li Y, Ran G, Lu G, Ni X, Liu D, Sun J, Xie C, Yao D, Bai W (2019) Highly sensitive label-free electrochemical aptasensor based on screen-printed electrode for detection of cadmium(II) ions. J Electrochem Soc 166:B449. https://doi.org/10.1149/2.0991906jes
Lu Y, Liang X, Niyungeko C, Zhou J, Xu J, Tian G (2018) A review of the identification and detection of heavy metal ions in the environment by voltammetry. Talanta 178:324–338. https://doi.org/10.1016/j.talanta.2017.08.033
Manikandan R, Narayanan SS (2019) Simultaneous anodic stripping voltammetric determination of Pb(II) and Cd(II) using poly methyl thymol blue film–modified electrode. Ionics 25:6083–6092. https://doi.org/10.1007/s11581-019-03143-w
Michalska A, Gałuszkiewicz A, Ogonowska M, Ocypa M, Maksymiuk K (2004) PEDOT films: multifunctional membranes for electrochemical ion sensing. J Solid State Electrochem 8:381–389. https://doi.org/10.1007/s10008-003-0459-8
Migliorini FL, Sanfelice RC, Pavinatto A, Steffens J, Steffens C, Correa DS (2017) Voltammetric cadmium(II) sensor based on a fluorine doped tin oxide electrode modified with polyamide 6/chitosan electrospun nanofibers and gold nanoparticles. Microchim Acta 184:1077–1084. https://doi.org/10.1007/s00604-017-2082-x
Mohammed MQ (2018) Patterned functionalisation of conducting polymer films. Thesis, University of Leicester. https://hdl.handle.net/2381/42227
Mohammed MQ, Jassem AM, Al-Shawi JMS, Alesary HF (2020) Comparative electrochemical behavior of poly(3-aminobenzoic acid) films in conventional and non-conventional solvents. In: AIP Conference Proceedings, vol 2290. AIP Publishing LLC, p 030029. https://doi.org/10.1063/5.0027520
Morante-Zarcero S, Pérez-Quintanilla D, Sierra I (2015) A disposable electrochemical sensor based on bifunctional periodic mesoporous organosilica for the determination of lead in drinking waters. J Solid State Electrochem 19:2117–2127. https://doi.org/10.1007/s10008-015-2889-5
Muralikrishna S, Nagaraju DH, Balakrishna RG, Surareungchai W, Ramakrishnappa T, Shivanandareddye AB (2017) Hydrogels of polyaniline with graphene oxide for highly sensitive electrochemical determination of lead ions. Anal Chim Acta 990:67–77. https://doi.org/10.1016/j.aca.2017.09.008
Niu L, Kvarnström C, Fröberg K, Ivaska A (2001) Electrochemically controlled surface morphology and crystallinity in poly(3,4-ethylenedioxythiophene) films. Synth Met 122:425–429. https://doi.org/10.1016/S0379-6779(00)00562-2
Ouyang L, Wei B, Kuo CC, Pathak S, Farrell B, Martin DC (2017) Enhanced PEDOT adhesion on solid substrates with electrografted P (EDOT-NH2). Sci Adv 3:e1600448. https://doi.org/10.1126/sciadv.1600448
Oztekin Y, Ramanaviciene A, Ryskevic N, Yazicigil Z, Üstündağ Z, Solak AO, Ramanavicius A (2011) 1,10-Phenanthroline modified glassy carbon electrode for voltammetric determination of cadmium(II) ions. Sens Actuators B Chem 157:146–153. https://doi.org/10.1016/j.snb.2011.03.041
Pandey SK, Sachan S, Singh SK (2019) Ultra-trace sensing of cadmium and lead by square wave anodic stripping voltammetry using ionic liquid modified graphene oxide. Mater Sci Energy Technol 2:667–675. https://doi.org/10.1016/j.mset.2019.09.004
Pillalamarri SK, Blum FB, Tokuhiro AT, Bertino MF (2005) One-pot synthesis of polyaniline-metal nanocomposites. Chem Mater 17:5941–5944. https://doi.org/10.1021/cm050827y
Promphet N, Rattanarat P, Rangkupan R, Chailapakul O, Rodthongkum N (2015) An electrochemical sensor based on graphene/polyaniline/polystyrene nanoporous fibers modified electrode for simultaneous determination of lead and cadmium. Sens Actuators B Chem 207:526–534. https://doi.org/10.1016/j.snb.2014.10.126
Qin D, Gao S, Wang L et al (2017) Three-dimensional carbon nanofiber derived from bacterial cellulose for use in a Nafion matrix on a glassy carbon electrode for simultaneous voltammetric determination of trace levels of Cd(II) and Pb(II). Microchim Acta 184:2759–2766. https://doi.org/10.1007/s00604-017-2260-x
Raghu GK, Sampath S, Pandurangappa M (2012) Chemically functionalized glassy carbon spheres: a new covalent bulk modified composite electrode for the simultaneous determination of lead and cadmium. J Solid State Electrochem 16:1953–1963. https://doi.org/10.1007/s10008-011-1595-1
Rahman MD, Kumar P, Park DS, Shim YB (2008) Electrochemical sensors based on organic conjugated polymers. Sensors 8:118–141. https://doi.org/10.3390/s8010118
Salmanipour A, Taher MA (2011) An electrochemical sensor for stripping analysis of Pb(II) based on multiwalled carbon nanotube functionalized with 5-Br-PADAP. J Solid State Electrochem 15:2695–2702. https://doi.org/10.1007/s10008-010-1197-3
Sapstead RM, Corden N, Hillman AR (2015) Latent fingerprint enhancement via conducting electrochromic copolymer films of pyrrole and 3,4-ethylenedioxythiophene on stainless steel. Electrochim Acta 162:119–128. https://doi.org/10.1016/j.electacta.2014.11.061
Schultheiss A, Carella A, Pouget S et al (2020) Water content control during solution-based polymerization: a key to reach extremely high conductivity in PEDOT thin films. J Mater Chem C 8:17254–17260. https://doi.org/10.1039/D0TC04899B
Shervedani RK, Akrami Z (2013) Gold–deferrioxamine nanometric interface for selective recognition of Fe(III) using square wave voltammetry and electrochemical impedance spectroscopy methods. Biosens Bioelectron 39:31–36. https://doi.org/10.1016/j.bios.2012.06.025
Sun Y, Du H, Deng Y, Lan Y, Feng C (2016) Preparation of polyacrylamide via surface-initiated electrochemical-mediated atom transfer radical polymerization (SI-eATRP) for Pb2+ sensing. J Solid State Electrochem 20:105–113. https://doi.org/10.1007/s10008-015-3008-3
Vilas-Boas M, Freire C, De Castro B, Hillman AR (1998) Electrochemical characterization of a novel salen-type modified electrode. J Phys Chem B 102:8533–8540. https://doi.org/10.1021/jp982160r
Vu HD, Nguyen LH, Nguyen TD, Nguyen HB, Nguyen TL, Tran DL (2015) Anodic stripping voltammetric determination of Cd2+ and Pb2+ using interpenetrated MWCNT/P1, 5-DAN as an enhanced sensing interface. Ionics 21:571–578. https://doi.org/10.1007/s11581-014-1199-8
Wang J, Bian C, Tong J, Sun J, Xia S (2012) Simultaneous detection of copper, lead and zinc on tin film/gold nanoparticles/gold microelectrode by square wave stripping voltammetry. Electroanalysis 24:1783–1790. https://doi.org/10.1002/elan.201200131
Xu Z, Fan X, Ma Q et al (2019) A sensitive electrochemical sensor for simultaneous voltammetric sensing of cadmium and lead based on Fe3O4/multiwalled carbon nanotube/laser scribed graphene composites functionalized with chitosan modified electrode. Mater Chem Phys 238:121877. https://doi.org/10.1016/j.matchemphys.2019.121877
Yao Y, Wu H, Ping J (2019) Simultaneous determination of Cd(II) and Pb(II) ions in honey and milk samples using a single-walled carbon nanohorns modified screen-printed electrochemical sensor. Food Chem 274:8–15. https://doi.org/10.1016/j.foodchem.2018.08.110
Yin T, Pan D, Qin W (2012) A solid-contact Pb2+-selective polymeric membrane electrode with Nafion-doped poly (pyrrole) as ion-to-electron transducer. J Solid State Electrochem 16:499–504. https://doi.org/10.1007/s10008-011-1358-z
Zhai Z, Huang N, Zhuang H et al (2018) A diamond/graphite nanoplatelets electrode for anodic stripping voltammetric trace determination of Zn(II), Cd(II), Pb(II) and Cu(II). Appl Surf Sci 457:1192–1201. https://doi.org/10.1016/j.apsusc.2018.06.266
Zhang Y, Li C, Su Y, Mu W, Han X (2020) Simultaneous detection of trace Cd(II) and Pb(II) by differential pulse anodic stripping voltammetry using a bismuth oxycarbide/nafion electrode. Inorg Chem Commun 111:107672. https://doi.org/10.1016/j.inoche.2019.107672
Acknowledgements
The authors would like to thank the Universities of Basrah, Koya, and Kerbala for providing the required materials and instruments for this work.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that might have influenced the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Mohammed, M.Q., Ismail, H.K., Alesary, H.F. et al. Use of a Schiff base-modified conducting polymer electrode for electrochemical assay of Cd(II) and Pb(II) ions by square wave voltammetry. Chem. Pap. 76, 715–729 (2022). https://doi.org/10.1007/s11696-021-01882-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-021-01882-7