Abstract
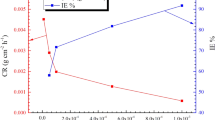
An ionic liquid (IL) comprising imidazolium moiety, specified as 3-(2,5-difluorobenzyl)-1-methyl-1H-imidazol-3-ium bromide [DFBMIm]Br is synthesized and applied, herein, for corrosion mitigation of iron–carbon (Fe–C) steel or mild steel in 0.5 M H2SO4 matrix. The studies were conducted after characterization of the synthesized IL with NMR and IR spectroscopy, for potentiodynamic polarization techniques and impedance spectroscopy for procuring the nature of IL for corrosion control, supplemented by SEM, X-ray (EDX) and AFM for getting knowledge about surface changes. Besides, DFTs, MD, adsorption kinetics and thermodynamical investigations for deep insights of adsorption mechanisms were computed. The IL shows good protection (with 99.39% efficiency through polarisation and 99.49% efficiency using EIS method), each as percent inhibition at 0.01 M and 298 K. Theoretical parameters using Jaguar—quantum mechanical engine for DFT and its comparison with Gaussian 09 program studies for the IL are also presented. Moreover, the interactions of the liquid over the metal substrate using Schrodinger–MS Suite for MD findings are corroborated with the estimable mechanism. Importantly, the electrical double layer-based model is also presented, specifically to support the practical observations for appreciable efficiency.












Similar content being viewed by others
References
Abd El-Lateef HM (2015) Experimental and computational investigation on the corrosion inhibition characteristics of mild steel by some novel synthesized imines in hydrochloric acid solutions. Corros Sci 92:104–117. https://doi.org/10.1016/j.corsci.2014.11.040
Aliaga C, Baldelli S (2006) Sum frequency generation spectroscopy and double-layer capacitance studies of the 1-butyl-3-methylimidazolium dicyanamide-platinum interface. J Phys Chem B 110:18481–18491. https://doi.org/10.1021/jp063476z
Azeez FA, Al-Rashed OA, Nazeer AA (2018) Controlling of mild-steel corrosion in acidic solution using environmentally friendly ionic liquid inhibitors: effect of alkyl chain. J Mol Liq 265:654–663. https://doi.org/10.1016/j.molliq.2018.05.093
Baldelli S (2003) Influence of water on the orientation of cations at the surface of a room-temperature ionic liquid: a sum frequency generation vibrational spectroscopic study. J Phys Chem B 107:6148–6152. https://doi.org/10.1021/jp027753n
Bentiss F, Lebrini M, Vezin H, Lagrenée M (2004) Experimental and theoretical study of 3-pyridyl-substituted 1,2,4-thiadiazole and 1,3,4-thiadiazole as corrosion inhibitors of mild steel in acidic media. Mater Chem Phys 87:18–23. https://doi.org/10.1016/j.matchemphys.2004.05.040
Bhaskaran PPD, Lata S, Singh G (2019) Imidazolium based ionic liquid as an efficient and green corrosion constraint for mild steel at acidic pH levels. J Mol Liq 278:467–476. https://doi.org/10.1016/j.molliq.2019.01.068
Bhrara K, Kim H, Singh G (2008) Inhibiting effects of butyl triphenyl phosphonium bromide on corrosion of mild steel in 0.5 M sulphuric acid solution and its adsorption characteristics. Corros Sci 50:2747–2754. https://doi.org/10.1016/j.corsci.2008.06.054
Bockris JO, Reddy AKN (2007) Modern electrochemistry 2A: fundamentals of electrodics. Springer, New York
Bouklah M, Hammouti B, Lagrenée M, Bentiss F (2006) Thermodynamic properties of 2,5-bis(4-methoxyphenyl)-1,3,4-oxadiazole as a corrosion inhibitor for mild steel in normal sulfuric acid medium. Corros Sci 48:2831–2842. https://doi.org/10.1016/j.corsci.2005.08.019
Chidiebere MA, Oguzie EE, Liu L, Li Y, Wang F (2014) Corrosion inhibition of Q235 mild steel in 0.5 M H2SO4 solution by phytic acid and synergistic iodide additives. Ind Eng Chem Res 53:7670–7679. https://doi.org/10.1021/ie404382v
Dahiya S, Lata S, Kumar P, Kumar R (2016) A descriptive study for corrosion control of low-alloy steel by Aloe vera extract in acidic medium. Corros Rev 34:241–248. https://doi.org/10.1515/corrrev-2016-0015
Eaborn C (1988) Compendium of chemical terminology: IUPAC recommendations. J Organomet Chem 356:C76–C77. https://doi.org/10.1016/0022-328x(88)83113-9
Ebenso EE (2003) Synergistic effect of halide ions on the corrosion inhibition of aluminium in H2SO4 using 2-acetylphenothiazine. Mater Chem Phys 79:58–70. https://doi.org/10.1016/S0254-0584(02)00446-7
Egorova KS, Ananikov VP (2018) Fundamental importance of ionic interactions in the liquid phase: a review of recent studies of ionic liquids in biomedical and pharmaceutical applications. J Mol Liq 272:271–300
El Ashry ESH, El Nemr A, Esawy SA, Ragab S (2006) Corrosion inhibitors. Part II: Quantum chemical studies on the corrosion inhibitions of steel in acidic medium by some triazole, oxadiazole and thiadiazole derivatives. Electrochim Acta 51:3957–3968. https://doi.org/10.1016/j.electacta.2005.11.010
Ezzat AO, Atta AM, Al-Lohedan HA, Hashem AI (2018) Synthesis and application of new surface active poly (ionic liquids) based on 1,3-dialkylimidazolium as demulsifiers for heavy petroleum crude oil emulsions. J Mol Liq 251:201–211. https://doi.org/10.1016/j.molliq.2017.12.081
Fedorov MV, Kornyshev AA (2008) Towards understanding the structure and capacitance of electrical double layer in ionic liquids. Electrochim Acta 53:6835–6840. https://doi.org/10.1016/j.electacta.2008.02.065
Fedorov MV, Georgi N, Kornyshev AA (2010) Double layer in ionic liquids: the nature of the camel shape of capacitance. Electrochem commun 12:296–299. https://doi.org/10.1016/j.elecom.2009.12.019
Gao X, Liu S, Lu H, Gao F, Ma H (2015) Corrosion inhibition of iron in acidic solutions by monoalkyl phosphate esters with different chain lengths. Ind Eng Chem Res 54:1941–1952. https://doi.org/10.1021/ie503508h
Gonçalves RS, Azambuja DS, Serpa Lucho AM (2002) Electrochemical studies of propargyl alcohol as corrosion inhibitor for. Corros Sci 44:467–479. https://doi.org/10.1016/S0010-938X(01)00069-5
Gupta NK, Verma C, Salghi R, Lgaz H, Mukherjee AK, Quraishi MA (2017) New phosphonate based corrosion inhibitors for mild steel in hydrochloric acid useful for industrial pickling processes: experimental and theoretical approach. New J Chem 41:13114–13129. https://doi.org/10.1039/c7nj01431g
Guzmán-Lucero D, Olivares-Xometl O, Martínez-Palou R, Likhanova NV, Domínguez-Aguilar MA, Garibay-Febles V (2011) Synthesis of selected vinylimidazolium ionic liquids and their effectiveness as corrosion inhibitors for carbon steel in aqueous sulfuric acid. Ind Eng Chem Res 50:7129–7140. https://doi.org/10.1021/ie1024744
Haque J, Srivastava V, Verma C, Lgaz H, Salghi R, Quraishi MA (2017) N-Methyl-N,N,N-trioctylammonium chloride as a novel and green corrosion inhibitor for mild steel in an acid chloride medium: electrochemical, DFT and MD studies. New J Chem 41:13647–13662. https://doi.org/10.1039/c7nj02254a
Haque J, Verma C, Srivastava V, Quraishi MA, Ebenso EE (2018) Experimental and quantum chemical studies of functionalized tetrahydropyridines as corrosion inhibitors for mild steel in 1 M hydrochloric acid. Results Phys 9:1481–1493. https://doi.org/10.1016/j.rinp.2018.04.069
Hoai NT, Van Hien P, Vu NSH, Son DL, Van Man T, Tri MD, Nam ND (2019) An improved corrosion resistance of steel in hydrochloric acid solution using Hibiscus sabdariffa leaf extract. Chem Pap 73:909–925. https://doi.org/10.1007/s11696-018-0649-6
Kannan P, Karthikeyan J, Murugan P, Rao TS, Rajendran N (2016) Corrosion Inhibition effect of novel methyl benzimidazolium Ionic liquid for carbon steel in HCl medium. J Mol Liq 221:368–380. https://doi.org/10.1016/j.molliq.2016.04.130
Kansal K, Chopra R, Kumar R, Kumar A, Yadav B, Sharma RK, Singh G (2017) Anti-corrosive properties of 2,3-dihydroxyquinoxaline on mild steel corrosion in sulphuric acid. Indian J Chem Technol 24:169–177
Kowsari E, Payami M, Amini R, Ramezanzadeh B, Javanbakht M (2014) Task-specific ionic liquid as a new green inhibitor of mild steel corrosion. Appl Surf Sci 289:478–486. https://doi.org/10.1016/j.apsusc.2013.11.017
Krauskopf E (1992) A. Wieckowski in J. Lipkowski and PN Ross (Eds.), Molecular adsorption at metal electrodes. VCH, New York
Kumar A, Trivedi M, Sharma RK, Singh G (2017a) Synthetic, spectral and structural studies of a Schiff base and its anticorrosive activity on mild steel in H2SO4. New J Chem 41:8459–8468. https://doi.org/10.1039/c7nj00896a
Kumar R, Chahal S, Dahiya S, Dahiya N, Kumar S, Lata S (2017b) Experimental and theoretical approach to exploit the corrosion inhibition activity of 3-formyl chromone derivatives on mild steel in 1 M H2SO4. Corros Rev 35:95–110. https://doi.org/10.1515/corrrev-2017-0029
Lai C, Xie B, Zou L, Zheng X, Ma X, Zhu S (2017) Adsorption and corrosion inhibition of mild steel in hydrochloric acid solution by S-allyl-O, O′-dialkyldithiophosphates. Results Phys 7:3434–3443. https://doi.org/10.1016/j.rinp.2017.09.012
Likhanova NV, Domínguez-Aguilar MA, Olivares-Xometl O, Nava-Entzana N, Arce E, Dorantes H (2010) The effect of ionic liquids with imidazolium and pyridinium cations on the corrosion inhibition of mild steel in acidic environment. Corros Sci 52:2088–2097. https://doi.org/10.1016/j.corsci.2010.02.030
Lockett V, Horne M, Sedev R, Rodopoulos T, Ralston J (2010) Differential capacitance of the double layer at the electrode/ionic liquids interface. Phys Chem Chem Phys 12:12499–12512. https://doi.org/10.1039/c0cp00170h
Mashuga ME, Olasunkanmi LO, Adekunle AS, Yesudass S, Kabanda MM, Ebenso EE, Chakraborty Banerjee P (2015) Adsorption, thermodynamic and quantum chemical studies of 1-hexyl-3-methylimidazolium based ionic liquids as corrosion inhibitors for mild steel in HCl. Materials (Basel) 8:3607–3632. https://doi.org/10.3390/ma8063607
Morales-Gil P, Walczak MS, Camargo CR, Cottis RA, Romero JM, Lindsay R (2015) Corrosion inhibition of carbon-steel with 2-mercaptobenzimidazole in hydrochloric acid. Corros Sci 101:47–55. https://doi.org/10.1016/j.corsci.2015.08.032
Nanbu N, Sasaki Y, Kitamura F (2003) In situ FT-IR spectroscopic observation of a room-temperature molten salt|gold electrode interphase. Electrochem Commun 5:383–387. https://doi.org/10.1016/S1388-2481(03)00073-0
Nessim MI, Zaky MT, Deyab MA (2018) Three new gemini ionic liquids: synthesis, characterizations and anticorrosion applications. J Mol Liq 266:703–710. https://doi.org/10.1016/j.molliq.2018.07.001
Obot IB, Macdonald DD, Gasem ZM (2015a) Density functional theory (DFT) as a powerful tool for designing new organic corrosion inhibitors: part 1: an overview. Corros Sci 99:1–30
Obot IB, Umoren SA, Gasem ZM, Suleiman R, El AB (2015b) Theoretical prediction and electrochemical evaluation of vinylimidazole and allylimidazole as corrosion inhibitors for mild steel in 1 M HCl. J Ind Eng Chem 21:1328–1339. https://doi.org/10.1016/j.jiec.2014.05.049
Parsons R (1990) Electrical double layer: recent experimental and theoretical developments. Chem Rev 90:813–826. https://doi.org/10.1021/cr00103a008
Qiang Y, Zhang S, Guo L, Zheng X, Xiang B, Chen S (2017) Experimental and theoretical studies of four allyl imidazolium-based ionic liquids as green inhibitors for copper corrosion in sulfuric acid. Corros Sci 119:68–78. https://doi.org/10.1016/j.corsci.2017.02.021
Rivera-Rubero S, Baldelli S (2004) Surface spectroscopy of room-temperature ionic liquids on a platinum electrode: a sum frequency generation study. J Phys Chem B 108:15133–15140. https://doi.org/10.1021/jp048260g
Sadanandhan AM, Khatri PK, Jain SL (2019) A novel series of cyclophosphazene derivatives containing imidazolium ionic liquids with variable alkyl groups and their physicochemical properties. J Mol Liq 295:111722. https://doi.org/10.1016/j.molliq.2019.111722
Saini N, Kumar R, Lgaz H, Salghi R, Chung IM, Kumar S, Lata S (2018) Minified dose of urispas drug as better corrosion constraint for soft steel in sulphuric acid solution. J Mol Liq 269:371–380. https://doi.org/10.1016/j.molliq.2018.08.070
Santos VO, Alves MB, Carvalho MS, Suarez PAZ, Rubim JC (2006) Surface-enhanced Raman scattering at the silver electrode/ionic liquid (BMIPF6) interface. J Phys Chem B 110:20379–20385. https://doi.org/10.1021/jp0643348
Sastri V (1998) Corrosion inhibitors: principles and applications. Wiley, New York
Singh T, Kumar A (2007) Aggregation behavior of ionic liquids in aqueous solutions: effect of alkyl chain length, cations, and anions. J Phys Chem B 111:7843–7851. https://doi.org/10.1021/jp0726889
Tan B, Zhang S, Liu H, Guo Y, Qiang Y, Li W, Guo L, Xu C, Chen S (2019) Corrosion inhibition of X65 steel in sulfuric acid by two food flavorants 2-isobutylthiazole and 1-(1,3-thiazol-2-yl) ethanone as the green environmental corrosion inhibitors: combination of experimental and theoretical researches. J Colloid Interface Sci 538:519–529. https://doi.org/10.1016/j.jcis.2018.12.020
Tao Z, Zhang S, Li W, Hou B (2009) Corrosion inhibition of mild steel in acidic solution by some oxo-triazole derivatives. Corros Sci 51:2588–2595. https://doi.org/10.1016/j.corsci.2009.06.042
Tawfik SM (2016) Ionic liquids based gemini cationic surfactants as corrosion inhibitors for carbon steel in hydrochloric acid solution. J Mol Liq 216:624–635. https://doi.org/10.1016/j.molliq.2016.01.066
Ullah S, Bustam MA, Shariff AM, Gonfa G, Izzat K (2016) Experimental and quantum study of corrosion of A36 mild steel towards 1-butyl-3-methylimidazolium tetrachloroferrate ionic liquid. Appl Surf Sci 365:76–83. https://doi.org/10.1016/j.apsusc.2015.12.232
Vashisht H, Kumar S, Bahadur I, Singh G (2013) Evaluation of (2-hydroxyethyl) triphenyl phosphonium bromide as corrosion inhibitor for mild steel in sulphuric acid. Int J Electrochem Sci 8:684
Vashisht H, Bahadur I, Kumar S, Goyal MS, Kaur G, Singh G, Katata-Seru L, Ebenso EE (2016) Synergistic interactions between tetra butyl phosphonium hydroxide and iodide ions on the mild steel surface for corrosion inhibition in acidic medium. J Mol Liq 224:19–29. https://doi.org/10.1016/j.molliq.2016.09.056
Vastag G, Shaban A, Vraneš M, Tot A, Belić S, Gadžurić S (2018) Influence of the N-3 alkyl chain length on improving inhibition properties of imidazolium-based ionic liquids on copper corrosion. J Mol Liq 264:526–533. https://doi.org/10.1016/j.molliq.2018.05.086
Weingärtner H (2008) Understanding ionic liquids at the molecular level: facts, problems, and controversies. Angew Chem Int Ed 47:654–670
Xhanari K, Grah N, Finšgar M, Fuchs-Godec R, Maver U (2017) Corrosion inhibition and surface analysis of amines on mild steel in chloride medium. Chem Pap 71:81–89. https://doi.org/10.1007/s11696-016-0046-y
Zhao Y, Sun Q, Luo J, Chen H, Cai W, Su X (2018) Hydrothermal fabrication of TiO2–MoO3 nanocomposites with superior performance for water treatment. Nano Struct Nano Objects 13:93–99. https://doi.org/10.1016/j.nanoso.2017.12.003
Acknowledgements
The authors of the manuscript acknowledge the University of Delhi and CSIR, New Delhi for Research and Development (R&D) Grant as well as the Director of USIC, Delhi. Authors are also grateful to Dr. Sudharsan Pandiyan for his training on Schrodinger simulation Suites and to Schrodinger INC for facilitating the evaluation license to run computational simulations.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
There are no conflicts to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bhaskaran, Y., Pancharatana, P.D., Sharma, R.K. et al. To evaluate an ionic liquid for anticorrosive impact on iron–carbon steel: synthesis, computational and experimental mechanism. Chem. Pap. 75, 789–803 (2021). https://doi.org/10.1007/s11696-020-01341-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-020-01341-9