Abstract
Background
Intestinal electrical stimulation (IES) has been proposed as a potential treatment for obesity. The aim of this study was to explore the central mechanism underlying the reduction of food intake and body weight by IES by studying the expression of anorexigenic- and orexigenic-peptide-containing neurons in the hypothalamus.
Materials and Methods
Diet-induced obese (DIO) rats were divided into three groups to receive sham, IES, and pair-feeding for 4 weeks. Food intake was measured automatically and presented as daily and body weight measured weekly. The expressions of oxytocin, an anorexigenic neuropeptide, in the paraventricular nucleus of the hypothalamus (PVN) and the supraoptic nuclei of the hypothalamus (SON) and orexin-A, an orexigenic neuropeptide, in the lateral hypothalamic area (LHA) were studied using immunohistochemistry.
Results
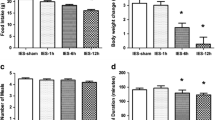
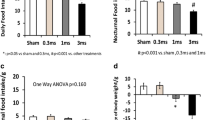
Compared with sham, IES reduced daily food intake by 28.3% at week 1, 35.6% at week 2, 15.6% at week 3, and 27.1% at week 4. Consistently, IES reduced body weight by 6.3%, compared with a weight gain of 7.2% in sham, and a slight weight loss of 0.5% in pair-feeding. Compared with sham, IES increased the expression of oxytocin-immunoreactive neurons in PVN and SON. Compared with sham, IES decreased the expression of orexin-immunoreactive neurons in LHA. Rats with pair-feeding also showed a relative decease in weight without any changes in the central hormones.
Conclusion
IES reduces food intake and body weight and improves glucose tolerance and insulin sensitivity in DIO rats. Its central mechanisms involve enhancement of anorexigenic peptides and suppression of orexigenic peptides in the hypothalamus.





Similar content being viewed by others
References
Ward ZJ, Bleich SN, Cradock AL, et al. Projected U.S. state-level prevalence of adult obesity and severe obesity. N Engl J Med. 2019;381(25):2440–50.
Kim DD, Basu A. Estimating the medical care costs of obesity in the United States: systematic review, meta-analysis, and empirical analysis. Value Health J Int Soc Pharmacoecon Outcomes Res. 2016;19(5):602–13.
De Luca M, Angrisani L, Himpens J, et al. Indications for surgery for obesity and weight-related diseases: position statements from the International Federation for the Surgery of Obesity and Metabolic Disorders (IFSO). Obes Surg. 2016;26(8):1659–96.
Sagar PM. Surgical treatment of morbid obesity. Br J Surg. 1995;82(6):732–9.
Crookes PF. Surgical treatment of morbid obesity. Annu Rev Med. 2006;57:243–64.
Dargent J. Gastric electrical stimulation as therapy of morbid obesity: preliminary results from the French study. Obes Surg. 2002;12(Suppl 1):21S–5S.
Cigaina V. Gastric pacing as therapy for morbid obesity: preliminary results. Obes Surg. 2002;12(Suppl 1):12S–6S.
Greenstein RJ, Belachew M. Implantable gastric stimulation (IGS) as therapy for human morbid obesity: report from the 2001 IFSO symposium in Crete. Obes Surg. 2002;12(Suppl 1):3S–5S.
Favretti F, De Luca M, Segato G, et al. Treatment of morbid obesity with the transcend implantable gastric stimulator (IGS): a prospective survey. Obes Surg. 2004;14(5):666–70.
Horbach T, Thalheimer A, Seyfried F, et al. Abiliti closed-loop gastric electrical stimulation system for treatment of obesity: clinical results with a 27-month follow-up. Obes Surg. 2015;25(10):1779–87.
Xu X, Lei Y, Chen JDZ. Duodenum electrical stimulation delays gastric emptying, reduces food intake and accelerates small bowel transit in pigs. Obes Silver Spring Md. 2011;19(2):442–8.
Li S, Chen JDZ. Pulse width-dependent effects of intestinal electrical stimulation for obesity: role of gastrointestinal motility and hormones. Obes Surg. 2017;27(1):70–7.
Sun Y, Chen J. Intestinal electric stimulation decreases fat absorption in rats: therapeutic potential for obesity. Obes Res. 2004;12(8):1235–42.
Aberle J, Busch P, Veigel J, et al. Duodenal electric stimulation: results of a first-in-man study. Obes Surg. 2016;26(2):369–75.
Yin J, Zhang J, Chen JDZ. Inhibitory effects of intestinal electrical stimulation on food intake, weight loss and gastric emptying in rats. Am J Physiol Regul Integr Comp Physiol. 2007;293(1):R78–82.
Tang M, Zhang J, Xu L, et al. Implantable gastric stimulation alters expression of oxytocin- and orexin-containing neurons in the hypothalamus of rats. Obes Surg. 2006;16(6):762–9.
Allen YS, Adrian TE, Allen JM, et al. Neuropeptide Y distribution in the rat brain. Science. 1983;221(4613):877–9.
Park ES, Yi SJ, Kim JS, et al. Changes in orexin-A and neuropeptide Y expression in the hypothalamus of the fasted and high-fat diet fed rats. J Vet Sci. 2004;5(4):295–302.
Blevins JE, Ho JM. Role of oxytocin signaling in the regulation of body weight. Rev Endocr Metab Disord. 2013;14(4):311–29.
Arletti R, Benelli A, Bertolini A. Oxytocin inhibits food and fluid intake in rats. Physiol Behav. 1990;48(6):825–30.
Rinaman L, Rothe EE. GLP-1 receptor signaling contributes to anorexigenic effect of centrally administered oxytocin in rats. Am J Physiol Regul Integr Comp Physiol. 2002;283(1):R99–106.
Olson BR, Drutarosky MD, Chow MS, et al. Oxytocin and an oxytocin agonist administered centrally decrease food intake in rats. Peptides. 1991;12(1):113–8.
Sakurai T, Amemiya A, Ishii M, et al. Orexins and orexin receptors: a family of hypothalamic neuropeptides and G protein-coupled receptors that regulate feeding behavior. Cell. 1998;92(4):573–85.
Farley C, Cook JA, Spar BD, et al. Meal pattern analysis of diet-induced obesity in susceptible and resistant rats. Obes Res. 2003;11(7):845–51.
Zhang J, Maude-Griffin R, Zhu H, et al. Gastric electrical stimulation parameter dependently alters ventral medial hypothalamic activity and feeding in obese rats. Am J Physiol Gastrointest Liver Physiol. 2011;301(5):G912–8.
Tai MM. A mathematical model for the determination of total area under glucose tolerance and other metabolic curves. Diabetes Care. 1994;17(2):152–4.
Zhang G, Bai H, Zhang H, et al. Neuropeptide exocytosis involving synaptotagmin-4 and oxytocin in hypothalamic programming of body weight and energy balance. Neuron. 2011;69(3):523–35.
Shikora SA, Bergenstal R, Bessler M, et al. Implantable gastric stimulation for the treatment of clinically severe obesity: results of the SHAPE trial. Surg Obes Relat Dis Off J Am Soc Bariatr Surg. 2009;5(1):31–7.
Madsen AN, Hansen G, Paulsen SJ, et al. Long-term characterization of the diet-induced obese and diet-resistant rat model: a polygenetic rat model mimicking the human obesity syndrome. J Endocrinol. 2010;206(3):287–96.
Maejima Y, Yokota S, Nishimori K, et al. The anorexigenic neural pathways of oxytocin and their clinical implication. Neuroendocrinology. 2018;107(1):91–104.
Hill JW. PVN pathways controlling energy homeostasis. Indian J Endocrinol Metab. 2012;16(Suppl 3):S627–36.
Altirriba J, Poher A-L, Rohner-Jeanrenaud F. Chronic oxytocin administration as a treatment against impaired leptin signaling or leptin resistance in obesity. Front Endocrinol. 2015;6:119.
Morton GJ, Thatcher BS, Reidelberger RD, et al. Peripheral oxytocin suppresses food intake and causes weight loss in diet-induced obese rats. Am J Physiol Endocrinol Metab. 2012;302(1):E134–44.
Wu Z, Xu Y, Zhu Y, et al. An obligate role of oxytocin neurons in diet induced energy expenditure. PLoS One. 2012;7(9):e45167.
Deblon N, Veyrat-Durebex C, Bourgoin L, et al. Mechanisms of the anti-obesity effects of oxytocin in diet-induced obese rats. PLoS One. 2011;6(9):e25565.
Blais A, Drouin G, Chaumontet C, et al. Impact of orexin-A treatment on food intake, energy metabolism and body weight in mice. PLoS One. 2017;12(1):e0169908.
Van den Pol AN. Lateral hypothalamic damage and body weight regulation: role of gender, diet, and lesion placement. Am J Phys. 1982;242(3):R265–74.
Kirchgessner AL, Liu M. Orexin synthesis and response in the gut. Neuron. 1999;24(4):941–51.
Kim GW, Lin JE, Valentino MA, et al. Regulation of appetite to treat obesity. Expert Rev Clin Pharmacol. 2011;4(2):243–59.
Liu J, Qiao X, Hou X, et al. Effect of intestinal pacing on small bowel transit and nutrient absorption in healthy volunteers. Obes Surg. 2009;19(2):196–201.
Sun Y, Chen JDZ. Intestinal electric stimulation accelerates whole gut transit and promotes fat excrement in conscious rats. Int J Obes 2005. 2009;33(8):817–23.
Li S, Zhu W, Zhang S, et al. Chronic intestinal electrical stimulation improves glucose intolerance and insulin resistance in diet-induced obesity rats. Obes Silver Spring Md. 2017;25(6):1061–8.
Näslund E, Gutniak M, Skogar S, et al. Glucagon-like peptide 1 increases the period of postprandial satiety and slows gastric emptying in obese men. Am J Clin Nutr. 1998;68(3):525–30.
Roslin MS, Cripps CN. Bariatric surgery in managing diabetes mellitus. Curr Opin Gastroenterol. 2016;32(6):481–6.
Maisiyiti A, Chen JD. Systematic review on gastric electrical stimulation in obesity treatment. Expert Rev Med Devices. 2019;16(10):855–61.
Funding
This work was supported by a Merit Review Award #I01BX002010 (to M.M.) from the US Department of Veterans Affairs Biomedical Laboratory Research and Development Program and partly by grant R01DK107754 (to J. C.) from the National Institutes of Health.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
All authors declare that they have no conflict of interest.
Ethical Approval Statement
All applicable institutional and/or national guidelines for the care and use of animals were followed. The study was performed according to the National Institutes of Health Guidelines on the use of laboratory animals and approved by the Animal Care and Use Committee of the Oklahoma City Veterans Health Care System (Oklahoma City, OK).
Informed Consent Statement
Informed consent does not apply.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Li, S., Kim, Y., Chen, J.D.Z. et al. Intestinal Electrical Stimulation Alters Hypothalamic Expression of Oxytocin and Orexin and Ameliorates Diet-Induced Obesity in Rats. OBES SURG 31, 1664–1672 (2021). https://doi.org/10.1007/s11695-020-05177-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-020-05177-w