Abstract
Background
Little is known regarding the use of opioid analgesics among patients who have undergone bariatric surgery.
The Roux-en-Y gastric bypass (RYGB) procedure has been shown to significantly increase the rate of absorption of and exposure to morphine, raising concerns regarding the potentially increased risk of side-effects and the development of substance-use disorder.
Objectives
The aim of this study was to describe the pattern of opioid use over time following RYGB and to see if the pattern differs between patients with a high opioid consumption (HOC) prior to surgery and those with a low consumption (LOC).
Setting
University Hospital of Örebro, Sweden.
Methods
The study was a descriptive retrospective population-based cohort study where two registers with complete coverage were cross-matched.
Results
The study population comprised 35,612 persons (1628 HOC, and 33,984 LOC). After surgery, the number of HOC patients increased to 2218.
Mean daily opioid consumption in the total population and the LOC group increased after surgery (p < .0005). In the HOC group, there was no difference between mean daily consumption before and after surgery.
Conclusion
In this nationwide study, we have showed that there is an increase in consumption of opioid analgesics after gastric bypass surgery in Sweden. The increase in the number of individuals with high opioid consumption in the total population was mainly due to an increase in the group of patients with a low consumption prior to surgery.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Morbid obesity is associated with various conditions causing musculoskeletal pain [1] often leading to use of prescription analgesics [2], including opioids. Bariatric surgery has proved to be an efficient intervention in the management of obesity resulting in lasting weight-loss and reduced obesity-related co-morbidity [3], improved physical function [4], and health-related quality-of-life (HRQoL) [5]. As a result, the number of post-bariatric surgery patients in the population is growing. Little is known regarding the use of opioid analgesics among patients who have undergone bariatric surgery. Recent studies has shown an increase in postoperative use of opioid analgesics both regarding the total dose and number of new persistent users [6,7,8,9]. The anatomical changes after Roux-en-Y gastric bypass (RYGB) can lead to changes in the pharmacokinetic properties of opioid analgesics leading to faster and more complete absorption of morphine oral solution [10], which is similar to the changes seen with ethanol [11]. This raises concerns regarding a potentially increased risk of developing substance-use disorder (SUD) [12,13,14]. The increased risk of side effects of non-steroidal anti-inflammatory drugs (NSAID) [15] after RYGB limits the pharmacological options available and likely pushes patients and prescribers toward opioids. It is thus important to gain a deeper understanding of the use of opioids after bariatric surgery.
The aim of this retrospective cohort study, including all patients undergoing gastric bypass surgery in Sweden between May 2007 and November 2013, was to describe the pattern of opioid use over time following surgery, and to see if the pattern differs between patients with a high opioid consumption prior to surgery (HOC) and those with a low consumption (LOC).
Methods
Study Design
The study was a descriptive retrospective population-based cohort study where two registers with complete coverage were cross-matched.
Patient Population
Patient data were obtained from the Scandinavian Obesity Surgery Register (SOReg), a Scandinavian bariatric surgery register used for research and quality improvement. The SOReg has nationwide coverage and is > 98% complete in terms of patient enrolment [16]. All patients undergoing bariatric surgery in Sweden May 2007–Nov 2013 were identified in the SOReg and data cross-matched with the Swedish Prescribed Drug Register (SPDR). This register contains patient data on all prescription medications dispensed in Sweden. It is intended to be used for epidemiological studies, research, and statistics within the health-care system [17]. From the SPDR, we obtained information on all opioid analgesics prescribed and dispensed (identified by ATC-codes N02AA01, N02AA03, N02AA05, N02AA55, N02AA59, N02AB01, N02AB02, N02AB03, N02AE01, N02AF02, N02AG01, N02AG02, N02AX02, and N02AX06). Participants were excluded if they had not been operated with gastric bypass (duodenal switch (n = 222 (0.6%), gastric banding (n = 266 (0.7%), sleeve gastrectomy (n = 644 (1.7%)), if they had died (n = 126 (0.3%)), and if they had had multiple bariatric surgery procedures (n = 22 (0.1%)).
Morphine Equivalents
Each participant’s daily intake of opioid was calculated as milligrams of oral morphine equivalent (OMEQ) using a previously described method [8, 9]. More accurate oral morphine equivalence factors [18] were used for transdermal and transmucosal buprenorphine [19], transdermal fentanyl, and oral tapentadol. Oral and rectal administration of ketobemidone [20] was included since this had not been considered in previous studies. Total opioid consumption was calculated as number of tablets/suppositories/ml/patches multiplied by strength in milligrams (milligrams per hour for patches) multiplied by the opioid conversion factor.
Opioid Use
In this study, dispensed opioids were considered to be the dose taken. The participants were divided between two groups according to total amount of opioid consumed prior to surgery, HOC with a mean daily intake greater than 10 mg OMEQ, and LOC with a mean daily intake of 10 mg OMEQ or less. Definition of HOC was chosen based on classification of chronic users from previous studies [8, 9]. Chronological data on use were presented as mean daily intake in mg OMEQ over a series of 3-month periods starting 24 months prior to surgery ending 24 months after surgery. This interval correlates to the 3-month period traditionally applied when renewing an opioid prescription for chronic pain in Sweden.
Statistical Analysis
Difference in mean daily consumption (OMEQ) before and after bariatric surgery was analyzed using paired-sample T test, and difference between men and women was analyzed using independent-sample T test. Analysis was performed on the full time-line and excluding the 3-month periods immediately before and after surgery. Data were analyzed using IBM SPSS Statistics version 22.
Results
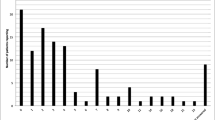
The study population comprised 35,612 persons (Table 1) who had undergone gastric bypass surgery (1628 HOC, and 33,984 LOC). After surgery, the number of HOC patients increased fairly consistently to 2218 at end of follow-up (Fig. 1).
When comparing mean daily opioid consumption in the total population prior to and after surgery, there was a statistically significant increase. In the HOC group, there was no difference between mean daily consumption before and after surgery. In the LOC group, however, there was a statistically significant increase in mean daily consumption after surgery. Furthermore, the amount of opioid consumed daily more than doubled after surgery in this group (Table 2). This increase remained when the 3-month periods immediately before and after surgery were excluded from the analysis.
In the group classed as high opioid consumers prior to surgery, 422 participants (25.9%) used 10 mg or less opioid analgesics after surgery (from a mean daily consumption before surgery of 22.9 to 3.0 mg after surgery) and 1206 participants (74.1%) continued or increased their consumption after surgery (mean daily consumption before surgery 31.5 to 43.8 mg after surgery).
In the LOC group 33,224 participants (97.8%) used 10 mg/day or less after surgery (from mean daily consumption 0.4 mg before surgery to 0.6 mg after surgery), and 760 LOCs (2.2%) became high opioid consumers after surgery (mean daily consumption 2.4 mg before surgery to 40.6 mg after surgery) (Figs. 2 and 3).
Full square indicates mean daily opioid consumption for patients with high opioid consumption before operation that used > 10 mg daily after surgery (n = 1206). Hollow circle indicates mean daily opioid consumption for patients with high opioid consumption before operation that used ≤ 10 mg daily after surgery (n = 422)
Full square indicates mean daily opioid consumption for patients with low opioid consumption before operation that used > 10 mg daily after surgery (n = 760). Hollow circle indicates mean daily opioid consumption for patients with low opioid consumption before operation that used ≤ 10 mg daily after surgery (n = 33,224)
There was no significant difference in mean daily use before and after surgery between women and men (Table 2).
Discussion
This study is one of few looking at the consumption of opioid analgesics in patients undergoing bariatric surgery. Our results show that there is an increase in opioid consumption after gastric bypass surgery, both the number of HOC patients and the amount of opioids consumed.
Previous studies have shown an unexpected increase in opioid analgesics consumption after bariatric surgery [6,7,8,9]. Our results with a larger population are consistent with their findings. However, in our study, the ratio of high opioid consumers before surgery and of low opioid consumers before surgery that became high consumers after surgery is slightly lower compared to previous studies. This can most likely be attributed to differences in classification of HOC. It is worrying that 760 patients in the LOC group became high opioid consumers during the 2 years following surgery, especially since 169 had not taken opioids during the 2 years prior to surgery. The post-surgery increase in consumption of opioids in the LOC group was probably the result of painful postoperative complications but other causes such as substance-use disorder cannot be ruled out. Patients considering gastric bypass surgery in general and opioid naïve patients in particular should be informed of the possible risks regarding postoperative opioid use. Consumption of opioid analgesics in the HOC group as a whole did not change after surgery, but, as can be seen in Fig. 2 approximately 25% of these patients significantly decrease their consumption and the remainder increase it. Mean age among HOCs was significantly lower after surgery (Table 2); this potentially protective effect has been shown previously [8] but not repeatedly [7], and there are many potentially confounding factors that we in this study are unable to adjust for. The number of HOC individuals in the total population appeared to increase fairly consistently, and if the 3-month period after surgery is excluded, bariatric surgery does not seem to have had an impact on this increase (Fig. 1).
Future studies prompted by these results would be to compare opioid use in patients who have undergone gastric bypass surgery with an obese control group and an in-depth analysis of the group of opioid-naïve patients that after surgery became HOC to find out what caused this change.
The strength of this study is that it used cohort data from comprehensive nationwide registers covering all patients undergoing gastric bypass surgery in Sweden, as well as having complete information on dispensed opioid analgesics. Furthermore, we used a previously described and established method to calculate morphine equivalents. Limitation of this study is the lack of a control group with obesity. It should also be noted that we cannot be certain that all dispensed opioids have been used and that our data do not include opioids given during hospital admissions.
Conclusion
In this nationwide study, we have showed that there is an increase in consumption of opioid analgesics after gastric bypass surgery in Sweden. There was no difference in use of opioid analgesics between women and men. The increase in the number of individuals with high opioid consumption in the total population was mainly due to an increase in the group of patients with a low consumption prior to surgery. Mean daily opioid consumption remained unchanged in the group with high consumption prior to surgery.
References
Hooper MM, Stellato TA, Hallowell PT, et al. Musculoskeletal findings in obese subjects before and after weight loss following bariatric surgery. Int J Obes. 2007;31:114–20.
Gribsholt SB, Thomsen RW, Farkas DK, et al. Changes in prescription drug use after gastric bypass surgery: a nationwide cohort study. Ann Surg. 2017;265:757–65.
Colquitt JL, Pickett K, Loveman E, Frampton GK. Surgery for weight loss in adults. Cochrane Database Syst Rev. 2014:Cd003641.
King WC, Chen JY, Belle SH, et al. Change in pain and physical function following bariatric surgery for severe obesity. JAMA. 2016;315:1362–71.
Raoof M, Näslund I, Rask E, et al. Health-related quality-of-life (HRQoL) on an average of 12 years after gastric bypass surgery. Obes Surg. 2015;25:1119–27.
Jakobsen GS, Smastuen MC, Sandbu R, et al. Association of bariatric surgery vs medical obesity treatment with long-term medical complications and obesity-related comorbidities. JAMA. 2018;319:291–301.
King WC, Chen JY, Belle SH, et al. Use of prescribed opioids before and after bariatric surgery: prospective evidence from a U.S. multicenter cohort study. Surg Obes Relat Dis. 2017.
Raebel MA, Newcomer SR, Bayliss EA, et al. Chronic opioid use emerging after bariatric surgery. Pharmacoepidemiol Drug Saf. 2014;23:1247–57.
Raebel MA, Newcomer SR, Reifler LM, et al. Chronic use of opioid medications before and after bariatric surgery. JAMA. 2013;310:1369–76.
Lloret-Linares C, Hirt D, Bardin C, et al. Effect of a Roux-en-Y gastric bypass on the pharmacokinetics of oral morphine using a population approach. Clin Pharmacokinet. 2014;53:919–30.
Klockhoff H, Naslund I, Jones AW. Faster absorption of ethanol and higher peak concentration in women after gastric bypass surgery. Br J Clin Pharmacol. 2002;54:587–91.
Ivezaj V, Saules KK, Schuh LM. New-onset substance use disorder after gastric bypass surgery: rates and associated characteristics. Obes Surg. 2014;24:1975–80.
Reslan S, Saules KK, Greenwald MK, et al. Substance misuse following Roux-en-Y gastric bypass surgery. Subst Use Misuse. 2014;49:405–17.
Saules KK, Wiedemann A, Ivezaj V, et al. Bariatric surgery history among substance abuse treatment patients: prevalence and associated features. Surg Obes Relat Dis. 2010;6:615–21.
Coblijn UK, Lagarde SM, de Castro SMM, et al. Symptomatic marginal ulcer disease after Roux-en-Y gastric bypass: incidence, risk factors and management. Obes Surg. 2015;25:805–11.
Hedenbro JL, Naslund E, Boman L, et al. Formation of the Scandinavian Obesity Surgery Registry, SOReg. Obes Surg. 2015;25:1893–900.
Wallerstedt SM, Wettermark B, Hoffmann M. The first decade with the Swedish prescribed drug register—a systematic review of the output in the scientific literature. Basic Clin Pharmacol Toxicol. 2016;119:464–9.
Svendsen K, Borchgrevink P, Fredheim O, et al. Choosing the unit of measurement counts: the use of oral morphine equivalents in studies of opioid consumption is a useful addition to defined daily doses. Palliat Med. 2011;25:725–32.
Mercadante S, Caraceni A. Conversion ratios for opioid switching in the treatment of cancer pain: a systematic review. Palliat Med. 2011;25:504–15.
Jylli L, Lundeberg S, Langius-Eklof A, et al. Comparison of the analgesic efficacy of ketobemidone and morphine for management of postoperative pain in children: a randomized, controlled study. Acta Anaesthesiol Scand. 2004;48:1256–9.
Funding
Grants Office at Region Örebro County, Grant OLL-710351.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Author 1 has no conflict of interest; author 2, no conflict of interest; author 3, no conflict of interest; author 4 has received fees for consulting and speeches from Baricol Bariatrics AB, Sweden, outside the submitted work.
Ethical Approval
The trial design was approved by the Regional Ethics Review Board in Uppsala, Sweden; dnr2015/219.
Informed Consent
Informed consent was obtained from all individuals participating in the study.
Electronic supplementary material
ESM 1
(DOCX 15 kb)
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Wallén, S., Szabo, E., Palmetun-Ekbäck, M. et al. Use of Opioid Analgesics Before and After Gastric Bypass Surgery in Sweden: a Population-Based Study. OBES SURG 28, 3518–3523 (2018). https://doi.org/10.1007/s11695-018-3377-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-018-3377-7