Abstract
Background
Diaphragmatic muscular remodeling is caused by various conditions and was mainly studied in pulmonary pathologies and chronic alterations of intra-thoracic pressure. We investigate the effect of the chronically increased intra-abdominal pressure (IAP) on the diaphragm by morphological and biochemical analysis.
Methods
Thirty rabbits were divided into control and study groups. IAP was increased in group B to 12 mmHg for 2 months. The left hemidiaphragm underwent morphological, while the right underwent biochemical analysis.
Results
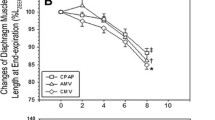
In H&E, all fibers were normal. ATPase analysis demonstrated that type I fibers show no differences between groups. Type ΙΙΑ were decreased (p = 0.016) while type ΙΙΒ/X fibers were increased (p = 0.025) in group B. Fibers with resistance to fatigue were decreased in group B (p = 0.024). In group B, biochemical activity for glutathione reductase (p = 0.004), glutathione peroxidase (p = 0.021), protein carbonylation (0.029), lipid peroxidation (p = 0.005), and balance of preoxidative–antioxidative factors (p = 0.006) was increased.
Conclusions
Chronically increased IAP induces alterations to the rabbit diaphragm. Adaptation, equivalent to strenuous contraction, transforms the diaphragm to be functionally more efficient toward workload but makes it vulnerable against oxidative stress.

Similar content being viewed by others
References
Edwards RHT, Faulkner JA. Structure and function of the respiratory muscles. In: Roussos C, Macklem PT, editors. The thorax, part A. New York: Marcel Dekker; 1986. p. 297–326.
Engel WK. The essentiality of histo and cytochemical studies of skeletal muscles in the investigation of neuromuscular disease. Neurology. 1972;12:778–94.
Leith DE, Bradley M. Ventilatory muscle strength and endurance training. J Appl Physiol. 1976;41:508–16.
Powers SK, Grinton S, Lawler J, et al. High intensity exercise training-induced metabolic alterations in respiratory muscles. Respir Physiol. 1992;89:169–77.
Keens TG, Chen V, Patel P, et al. Cellular adaptations of the ventilatory muscles to a chronic increased respiratory load. J Appl Physiol. 1978;44:905–8.
Papavramidis TS, Kotidis E, Ioannidis K, et al. Diaphragmatic adaptation following intra-abdominal weight charging. Obes Surg. 2010;21:1612–6.
Papavramidis TS, Lazou TP, Cheva A, et al. Chronically increased intra-abdominal pressure: validating a model. Obes Surg. 2010;20(7):900–5.
Karnak I, Aksöz E, Ekinci S, et al. Increased maternal intra-abdominal pressure alters the contractile properties of the fetal rabbit bladder. J Pediatr Surg. 2008;43:1711–7.
Sugerman H, Windsor A, Bessos M, et al. Intra-abdominal pressure, sagittal abdominal diameter and obesity comorbidity. J Intern Med. 1997;241:71–9.
Sugerman HJ. Effects of increased intra-abdominal pressure in severe obesity. Surg Clin North Am. 2001;81:1063–75.
Varela JE, Hinojosa M, Nguyen N. Correlations between intra-abdominal pressure and obesity-related co-morbidities. Surg Obes Relat Dis. 2009;5:524–8.
Bhatia SJ, Narawane NM, Shalia KK, et al. Effect of tense ascites on esophageal body motility and lower esophageal sphincter pressure. Indian J Gastroenterol. 1999;18:63–5.
Twardowski ZJ, Tully RJ, Ersoy FF, et al. Computerized tomography with and without intraperitoneal contrast for determination of intraabdominal fluid distribution and diagnosis of complications in peritoneal dialysis patients. ASAIO Trans. 1990;36:95–103.
Papavramidis TS, Duros V, Michalopoulos A, et al. Intra-abdominal pressure alterations after large pancreatic pseudocyst transcutaneous drainage. BMC Gastroenterol. 2009;9:42.
Van der Laan L, Nagel PH, Lohle PN, et al. Retroperitoneal haematoma seen on CT scan in 2 patients with abdominal symptoms, aneurysm of the abdominal aorta and normal hemodynamics. Ned Tijdschr Geneeskd. 2005;149:2001–4.
Zacchi P, Mearin F, Humbert P, et al. Effect of obesity on gastroesophageal resistance to flow in man. Dig Dis Sci. 1991;36:1473–80.
Sugrue M, Buist MD, Hourihan F, et al. Prospective study of intraabdominal hypertension and renal function after laparotomy. Br J Surg. 1995;82:235–8.
Sugrue M, Jones F, Deane SA, et al. Intraabdominal hypertension is an independent cause of postoperative renal impairment. Arch Surg. 1999;134:1082–5.
Kotidis EV, Papavramidis TS, Ioannidis K, et al. The effect of chronically increased intra-abdominal pressure on rectus abdominis muscle histology an experimental study on rabbits. J Surg Res. doi:10.1016/j.jss.2010.06.034
Dubovitz V, Brooke HM. Muscle biopsy: a modern approach vol. 2. London: Saunders; 1973.
Alamdari DH, Paletas K, Pegiou T, et al. A novel assay for the evaluation of the prooxidant–antioxidant balance, before and after antioxidant vitamin administration in type II diabetes patients. Clin Biochem. 2007;40(3–4):248–54.
Ohkawa H, Ohishi N, Yagi K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem. 1979;95(2):351–8.
Tatzber F, Griebenow S, Wonisch W, et al. Dual method for the determination of peroxidase activity and total peroxides-iodide leads to a significant increase of peroxidase activity in human sera. Anal Biochem. 2003;316:147–53.
Cribb AE, Leeder JS, Spielberg SP. Use of a microplate reader in assay of glutathione reductase using 5,5′-dithiobis(2-nitrobenzoic acid). Anal Biochem. 1989;183:195–6.
Paglia DE, Valentine WN. Studies on the quantitative and qualitative characterization of erythrocyte glutathione peroxidase. J Lab Clin Med. 1967;70:158–69.
Teare JP, Punchard NA, Powell JJ, et al. Automated spectrophotometric method for determining oxidized and reduced glutathione in liver. Clin Chem. 1993;39:686–9.
Alamdari DH, Kostidou E, Paletas K, et al. High sensitivity enzyme-linked immunosorbent assay (ELISA) method for measuring protein carbonyl in samples with low amounts of protein. Free Radical Biol Med. 2005;39:1362–7.
Oliven A, Carmi N, Coleman R, et al. Age-related changes in upper airway muscles morphological and oxidative properties. Exp Gerontol. 2001;36(10):1673–86.
Rodrigues CJ, Rodrigues AJ, Bohm GM. Effects of aging on muscle fibers and collagen content of the diaphragm: a comparison with the rectus abdominis muscle. Gerontology. 1996;42(4):218–28.
Fox J, Garber P, Hoffman M, et al. Morphological characteristics of skeletal muscles in relation to gender. Aging Clin Exp Res. 2003;15(3):264–9.
Sciote JJ, Horton MJ, Zyman Y, et al. Differential effects of diminished oestrogen and androgen levels on development of skeletal muscle fibers in hypogonadic mice. Acta Physiol Scand. 2001;172(3):179–87.
De Troyer A, Sampson M, Sigrist S, et al. The diaphragm: two muscles. Science. 1981;213(4504):37–8.
Capdevilla X, Lopez S, Bernard N, et al. Effects of controlled mechanical ventilation on respiratory muscle contractile properties in rabbits. Intensive Care Med. 2003;29:103–10.
Moore BJ, Feldman HA, Reid B. Developmental changes in diaphragm contractile properties. J Appl Physiol. 1993;75(2):522–6.
Yang L, Bourdon J, Gottfried SB, et al. Regulation of myosin heavy chain gene expression after short-term diaphragm inactivation. Am J Physiol Lung Cell Mol Physiol. 1998;274:L980–9.
Esposito A, Germinario E, Zanin M, et al. Isoform switching in myofibrillar and excitation–contraction coupling proteins contributes to diminished contractile function in regenerating rat soleus muscle. J Appl Physiol. 2007;102:1640–8.
Hyaes A, Williams DA. Contractile properties of clenbuterol-treated mdx muscle are enhanced by low-intensity swimming. J Appl Physiol. 1997;82(2):435–9.
Dean RT, Fu S, Stoker R, et al. Biochemistry and pathology of radical-mediated protein oxidation. Biochem J. 1997;324:1–18.
Morrissey PA, O’Brien NM. Dietary antioxidants in health and disease. Int Dairy J. 1998;8:463–72.
Diaz PT, She ZW, Davis WB, et al. Hydroxylation of salicylate by the in vitro diaphragm: evidence for hydroxyl radical production during fatigue. J Appl Physiol. 1993;75:540–5.
Reid MB, Shoji T, Moody MR, et al. Reactive oxygen in skeletal muscle. II. Extracellular release of free radicals. J Appl Physiol. 1992;73:1805–9.
Supinski GS, Anzueto A. Oxygen-derived free radicals and the respiratory muscles. In: Roussos C, editor. The thorax. New York: Dekker; 1995. p. 349–86.
Shindoh C, DiMarco A, Thomas A, et al. Effect of N-acetylcysteine on diaphragm fatigue. J Appl Physiol. 1990;68:2107–13.
Martin TP, Vailas AC, Durivage JB, et al. Quantitative histochemical determination of muscle enzymes: biochemical verification. J Histochem Cytochem. 1985;33:1053–9.
Preedy VR, Paice A, Mantle D, et al. Alcoholic myopathy: biochemical mechanisms. Drug Alcohol Depend. 2001;63:199–205.
Acknowledgments
We would like to thank John Martos for taking care of the laboratory animals, Εkaterini Theologou for the technical support concerning the morphologic analysis, and Christina Befani for the assistance concerning the biochemical analysis.
Conflict of Interest
All authors have no conflict of interest to declare.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Papavramidis, T.S., Kotidis, E., Ioannidis, K. et al. The Effects of Chronically Increased Intra-abdominal Pressure on the Rabbit Diaphragm. OBES SURG 22, 487–492 (2012). https://doi.org/10.1007/s11695-012-0587-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-012-0587-2