Abstract
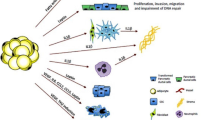
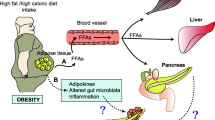
The only universally accepted risk factors for the development of pancreatic cancer are a positive family history or a history of smoking. Although the contribution of pancreatitis to pancreatic carcinogenesis has been debated for decades in the epidemiology literature, the actual mechanism is still unclear. With the rising epidemic of obesity, scientists have begun to focus on the contribution of chronic inflammatory state of morbidly obese patients in an effort to better understand the contribution of inflammation to the comorbidities of obesity. Notably, population studies are beginning to show that one of the most serious potential comorbidities of obesity is an increased lifetime risk of developing cancer. In this article, the current literature that exists supporting this Chronic Inflammatory Hypothesis as it pertains to obesity and pancreatic carcinogenesis is reviewed. To date, studies have focused on interleukin-6, a cytokine known to play a role in obesity, chronic pancreatitis and pancreatic cancer. The anti-inflammatory adipocytokine, adiponectin, has also shown promise as a key player in this mechanism and has recently been found to be more specific than standard tumor markers in differentiating pancreatic cancer from chronic pancreatitis. If the pathogenesis of pancreatic cancer is related to hormone levels associated with obesity, such as adipocytokines, and cytokines associated with chronic inflammation, this could potentially lead to the development of new pancreatic cancer tumor markers and ultimately new therapies and methods of prevention.
Similar content being viewed by others
References
Cottam DR, Mattar SG, Barinas-Mitchell E, et al. The chronic inflammatory hypothesis for the morbidity associated with morbid obesity: implications and effects of weight loss. Obes Surg. 2004;14:589–600.
Nunez NP, Oh WJ, Rozenberg J, et al. Accelerated tumor formation in a fatless mouse with type 2 diabetes and inflammation. Cancer Res. 2006;66:5469–76.
Gonullu G, Ersoy C, Ersoy A, et al. Relation between insulin resistance and serum concentrations of IL-6 and TNF-alpha in overweight or obese women with early stage breast cancer. Cytokine. 2005;31:264–9.
Rose DP, Komninou D, Stephenson GD. Obesity, adipocytokines, and insulin resistance in breast cancer. Obes Rev. 2004;5:153–65.
Nair S, Lee YH, Rousseau E, et al. Increased expression of inflammation-related genes in cultured preadipocytes/stromal vascular cells from obese compared with non-obese Pima Indians. Diabetologia. 2005;48:1784–8.
Adams TD, Gress RE, Smith SC, et al. Long-term mortality after gastric bypass surgery. N Engl J Med. 2007;357:753–61.
Duell EJ, Casella DP, Burk RD, Kelsey KT, Holly EA. Inflammation, genetic polymorphisms in proinflammatory genes TNF-A, RANTES, and CCR5, and risk of pancreatic adenocarcinoma. Cancer Epidemiol Biomarkers Prev. 2006;15:726–31.
Esposito I, Menicagli M, Funel N, et al. Inflammatory cells contribute to the generation of an angiogenic phenotype in pancreatic ductal adenocarcinoma. J Clin Pathol. 2004;57:630–6.
Silha JV, Krsek M, Sucharda P, Murphy LJ. Angiogenic factors are elevated in overweight and obese individuals. Int J Obes (Lond). 2005;29:1308–14.
Gumbs AA, Modlin IM, Ballantyne GH. Changes in insulin resistance following bariatric surgery: role of caloric restriction and weight loss. Obes Surg. 2005;15:462–73.
McDade TP, Perugini RA, Vittimberga FJ Jr, Carrigan RC, Callery MP. Salicylates inhibit NF-kappaB activation and enhance TNF-alpha-induced apoptosis in human pancreatic cancer cells. J Surg Res. 1999;83:56–61.
Somasundar P, Yu AK, Vona-Davis L, McFadden DW. Differential effects of leptin on cancer in vitro. J Surg Res. 2003;113:50–5.
Gumbs AA, Bessler M, Milone L, Schrope B, Chabot J. Contribution of obesity to pancreatic carcinogenesis. Surg Obes Relat Dis. 2008;4:186–93.
Ballantyne GH, Gumbs A, Modlin IM. Changes in insulin resistance following bariatric surgery and the adipoinsular axis: role of the adipocytokines, leptin, adiponectin and resistin. Obes Surg. 2005;15:692–9.
Pitt HA. Hepato-pancreato-biliary fat: the good, the bad and the ugly. HPB (Oxford). 2007;9:92–7.
Mathur A, Marine M, Lu D, et al. Nonalcoholic fatty pancreas disease. HPB (Oxford). 2007;9:312–8.
Chang MC, Chang YT, Su TC, et al. Adiponectin as a potential differential marker to distinguish pancreatic cancer and chronic pancreatitis. Pancreas. 2007;35:16–21.
Papachristou GI, Papachristou DJ, Avula H, Slivka A, Whitcomb DC. Obesity increases the severity of acute pancreatitis: performance of APACHE-O score and correlation with the inflammatory response. Pancreatology. 2006;6:279–85.
Martinez J, Johnson CD, Sanchez-Paya J, et al. Obesity is a definitive risk factor of severity and mortality in acute pancreatitis: an updated meta-analysis. Pancreatology. 2006;6:206–9.
Algul H, Treiber M, Lesina M, Schmid RM. Mechanisms of disease: chronic inflammation and cancer in the pancreas—a potential role for pancreatic stellate cells? Nat Clin Pract Gastroenterol Hepatol. 2007;4:454–62.
Ebrahimi B, Tucker SL, Li D, Abbruzzese JL, Kurzrock R. Cytokines in pancreatic carcinoma: correlation with phenotypic characteristics and prognosis. Cancer. 2004;101:2727–36.
Farrow B, Sugiyama Y, Chen A, et al. Inflammatory mechanisms contributing to pancreatic cancer development. Ann Surg. 2004;239:763–9; discussion 69–71.
Fenton JI, Hursting SD, Perkins SN, Hord NG. Interleukin-6 production induced by leptin treatment promotes cell proliferation in an Apc (Min/+) colon epithelial cell line. Carcinogenesis. 2006;27:1507–15.
Cozen W, Gebregziabher M, Conti DV, et al. Interleukin-6-related genotypes, body mass index, and risk of multiple myeloma and plasmacytoma. Cancer Epidemiol Biomarkers Prev. 2006;15:2285–91.
Okada S, Okusaka T, Ishii H, et al. Elevated serum interleukin-6 levels in patients with pancreatic cancer. Jpn J Clin Oncol. 1998;28:12–5.
Anderson KE, Johnson TW, Lazovich D, Folsom AR. Association between nonsteroidal anti-inflammatory drug use and the incidence of pancreatic cancer. J Natl Cancer Inst. 2002;94:1168–71.
Falconer JS, Fearon KC, Plester CE, Ross JA, Carter DC. Cytokines, the acute-phase response, and resting energy expenditure in cachectic patients with pancreatic cancer. Ann Surg. 1994;219:325–31.
Barber MD, McMillan DC, Wallace AM, et al. The response of leptin, interleukin-6 and fat oxidation to feeding in weight-losing patients with pancreatic cancer. Br J Cancer. 2004;90:1129–32.
Makino T, Noguchi Y, Yoshikawa T, Doi C, Nomura K. Circulating interleukin 6 concentrations and insulin resistance in patients with cancer. Br J Surg. 1998;85:1658–62.
Wigmore SJ, Fearon KC, Sangster K, et al. Cytokine regulation of constitutive production of interleukin-8 and -6 by human pancreatic cancer cell lines and serum cytokine concentrations in patients with pancreatic cancer. Int J Oncol. 2002;21:881–6.
Martignoni ME, Kunze P, Hildebrandt W, et al. Role of mononuclear cells and inflammatory cytokines in pancreatic cancer-related cachexia. Clin Cancer Res. 2005;11:5802–8.
Amaral ME, Barbuio R, Milanski M, et al. Tumor necrosis factor-alpha activates signal transduction in hypothalamus and modulates the expression of pro-inflammatory proteins and orexigenic/anorexigenic neurotransmitters. J Neurochem. 2006;98:203–12.
Author information
Authors and Affiliations
Corresponding author
Additional information
The author has no conflicts of interest to disclose.
Rights and permissions
About this article
Cite this article
Gumbs, A.A. Obesity, Pancreatitis, and Pancreatic Cancer. OBES SURG 18, 1183–1187 (2008). https://doi.org/10.1007/s11695-008-9599-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-008-9599-3