Abstract
In this review, we summarize the novel findings from our series of studies on the leading metastasis-related gene, osteopontin (OPN). In our previous gene expression profiling study, OPN was identified as one of the leading genes associated with the metastasis of hepatocellular carcinoma (HCC). We focused on OPN to evaluate its prognostic values and important roles in HCC metastasis. A retrospective study of large cohorts of HCC patients demonstrated that plasma OPN level was one of the leading independent prognostic factors for HCC patients, even in the early stage of HCC, and could serve as a surrogate serologic biomarker for monitoring the treatment response and tumor recurrence after HCC resection. Using both in vitro and in vivo investigations, we found that OPN has an important role in metastasis and tumor growth of HCC and is an attractive potential therapeutic target for combating HCC metastasis. We also found that OPN+ HCC cells have much more amplifications at chromosomal regions, and promoter polymorphisms are important in the regulation of OPN expression and tumor growth and lung metastasis of HCC.
Similar content being viewed by others
References
McAllister SS, Gifford AM, Greiner AL, Kelleher SP, Saelzler MP, Ince TA, Reinhardt F, Harris LN, Hylander BL, Repasky EA, Weinberg RA. Systemic endocrine instigation of indolent tumor growth requires osteopontin. Cell 2008; 133(6): 994–1005
Denhardt DT, Giachelli CM, Rittling SR. Role of osteopontin in cellular signaling and toxicant injury. Annu Rev Pharmacol Toxicol 2001; 41(1): 723–749
Weber GF. The metastasis gene osteopontin: a candidate target for cancer therapy. Biochim Biophys Acta 2001; 1552(2): 61–85
Wu CY, Wu MS, Chiang EP, Wu CC, Chen YJ, Chen CJ, Chi NH, Chen GH, Lin JT. Elevated plasma osteopontin associated with gastric cancer development, invasion and survival. Gut 2007; 56(6): 782–789
Khodavirdi AC, Song Z, Yang S, Zhong C, Wang S, Wu H, Pritchard C, Nelson PS, Roy-Burman P. Increased expression of osteopontin contributes to the progression of prostate cancer. Cancer Res 2006; 66(2): 883–888
Bramwell VH, Doig GS, Tuck AB, Wilson SM, Tonkin KS, Tomiak A, Perera F, Vandenberg TA, Chambers AF. Serial plasma osteopontin levels have prognostic value in metastatic breast cancer. Clin Cancer Res 2006; 12(11): 3337–3343
Medico E, Gentile A, Lo Celso C, Williams TA, Gambarotta G, Trusolino L, Comoglio PM. Osteopontin is an autocrine mediator of hepatocyte growth factor-induced invasive growth. Cancer Res 2001; 61(15): 5861–5868
Tuck AB, Hota C, Wilson SM, Chambers AF. Osteopontin-induced migration of human mammary epithelial cells involves activation of EGF receptor and multiple signal transduction pathways. Oncogene 2003; 22(8): 1198–1205
Ariztia EV, Subbarao V, Solt DB, Rademaker AW, Iyer AP, Oltvai ZN. Osteopontin contributes to hepatocyte growth factor-induced tumor growth and metastasis formation. Exp Cell Res 2003; 288(2): 257–267
Sharp JA, Sung V, Slavin J, Thompson EW, Henderson MA. Tumor cells are the source of osteopontin and bone sialoprotein expression in human breast cancer. Lab Invest 1999; 79(7): 869–877
Urquidi V, Sloan D, Kawai K, Agarwal D, Woodman AC, Tarin D, Goodison S. Contrasting expression of thrombospondin-1 and osteopontin correlates with absence or presence of metastatic phenotype in an isogenic model of spontaneous human breast cancer metastasis. Clin Cancer Res 2002; 8(1): 61–74
Chen H, Ke Y, Oates AJ, Barraclough R, Rudland PS. Isolation of and effector for metastasis-inducing DNAs from a human metastatic carcinoma cell line. Oncogene 1997; 14(13): 1581–1588
Oates AJ, Barraclough R, Rudland PS. The identification of osteopontin as a metastasis-related gene product in a rodent mammary tumour model. Oncogene 1996; 13(1): 97–104
Ye QH, Qin LX, Forgues M, He P, Kim JW, Peng AC, Simon R, Li Y, Robles AI, Chen Y, Ma ZC, Wu ZQ, Ye SL, Liu YK, Tang ZY, Wang XW. Predicting hepatitis B virus-positive metastatic hepatocellular carcinomas using gene expression profiling and supervised machine learning. Nat Med 2003; 9(4): 416–423
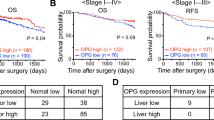
Zhang H, Ye QH, Ren N, Zhao L, Wang YF, Wu X, Sun HC, Wang L, Zhang BH, Liu YK, Tang ZY, Qin LX. The prognostic significance of preoperative plasma levels of osteopontin in patients with hepatocellular carcinoma. J Cancer Res Clin Oncol 2006; 132(11): 709–717
Huang H, Zhang XF, Zhou HJ, Xue YH, Dong QZ, Ye QH, Qin LX. Expression and prognostic significance of osteopontin and caspase-3 in hepatocellular carcinoma patients after curative resection. Cancer Sci 2010; 101(5): 1314–1319
Sun J, Xu HM, Zhou HJ, Dong QZ, Zhao Y, Fu LY, Hei ZY, Ye QH, Ren N, Jia HL, Qin LX. The prognostic significance of preoperative plasma levels of osteopontin in patients with TNM stage-I of hepatocellular carcinoma. J Cancer Res Clin Oncol 2010; 136(1): 1–7
Zhou C, Zhou HJ, Zhang XF, Lou LL, Ye QH, Zheng Y, Wang J, Zhu HT, Dong QZ, Jia HL, Zhu WW, Guo L, Zhao Y, Gao DM, Qin LX. Postoperative serum osteopontin level is a novel monitor for treatment response and tumor recurrence after resection of hepatitis B-related hepatocellular carcinoma. Ann Surg Oncol 2013; 20(3): 929–937
Sun BS, Dong QZ, Ye QH, Sun HJ, Jia HL, Zhu XQ, Liu DY, Chen J, Xue Q, Zhou HJ, Ren N, Qin LX. Lentiviral-mediated miRNA against osteopontin suppresses tumor growth and metastasis of human hepatocellular carcinoma. Hepatology 2008; 48(6): 1834–1842
Xue YH, Zhang XF, Dong QZ, Sun J, Dai C, Zhou HJ, Ren N, Jia HL, Ye QH, Qin LX. Thrombin is a therapeutic target for metastatic osteopontin-positive hepatocellular carcinoma. Hepatology 2010; 52(6): 2012–2022
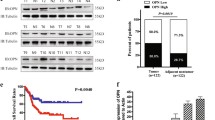
Wu JC, Sun BS, Ren N, Ye QH, Qin LX. Genomic aberrations in hepatocellular carcinoma related to osteopontin expression detected by array-CGH. J Cancer Res Clin Oncol 2010; 136(4): 595–601
Dong QZ, Zhang XF, Zhao Y, Jia HL, Zhou HJ, Dai C, Sun HJ, Qin Y, Zhang WD, Ren N, Ye QH, Qin LX. Osteopontin promoter polymorphisms at locus −443 significantly affect the metastasis and prognosis of human hepatocellular carcinoma. Hepatology 2013; 57(3): 1024–1034
Khodavirdi AC, Song Z, Yang S, Zhong C, Wang S, Wu H, Pritchard C, Nelson PS, Roy-Burman P. Increased expression of osteopontin contributes to the progression of prostate cancer. Cancer Res 2006; 66(2): 883–888
Coppola D, Szabo M, Boulware D, Muraca P, Alsarraj M, Chambers AF, Yeatman TJ. Correlation of osteopontin protein expression and pathological stage across a wide variety of tumor histologies. Clin Cancer Res 2004; 10(1): 184–190
Sedivy R, Peters K, Klöppel G. Osteopontin expression in ductal adenocarcinomas and undifferentiated carcinomas of the pancreas. Virchows Arch 2005; 446(1): 41–45
Mack PC, Redman MW, Chansky K, Williamson SK, Farneth NC, Lara PN Jr, Franklin WA, Le QT, Crowley JJ, Gandara DR; SWOG. Lower osteopontin plasma levels are associated with superior outcomes in advanced non-small-cell lung cancer patients receiving platinum-based chemotherapy: SWOG Study S0003. J Clin Oncol 2008; 26(29): 4771–4776
Anborgh PH, Mutrie JC, Tuck AB, Chambers AF. Role of the metastasis-promoting protein osteopontin in the tumour microenvironment. J Cell Mol Med 2010; 14(8): 2037–2044
Kim J, Ki SS, Lee SD, Han CJ, Kim YC, Park SH, Cho SY, Hong YJ, Park HY, Lee M, Jung HH, Lee KH, Jeong SH. Elevated plasma osteopontin levels in patients with hepatocellular carcinoma. Am J Gastroenterol 2006; 101(9): 2051–2059
Pan HW, Ou YH, Peng SY, Liu SH, Lai PL, Lee PH, Sheu JC, Chen CL, Hsu HC. Overexpression of osteopontin is associated with intrahepatic metastasis, early recurrence, and poorer prognosis of surgically resected hepatocellular carcinoma. Cancer 2003; 98(1): 119–127
El-Tanani MK. Role of osteopontin in cellular signaling and metastatic phenotype. Front Biosci 2008; 13: 4276–4284
Ho PK, Hawkins CJ. Mammalian initiator apoptotic caspases. FEBS J 2005; 272(21): 5436–5453
Gu T, Ohashi R, Cui R, Tajima K, Yoshioka M, Iwakami S, Sasaki S, Shinohara A, Matsukawa T, Kobayashi J, Inaba Y, Takahashi K. Osteopontin is involved in the development of acquired chemoresistance of cisplatin in small cell lung cancer. Lung Cancer 2009; 66(2): 176–183
Graessmann M, Berg B, Fuchs B, Klein A, Graessmann A. Chemotherapy resistance of mouse WAP-SVT/t breast cancer cells is mediated by osteopontin, inhibiting apoptosis downstream of caspase-3. Oncogene 2007; 26(20): 2840–2850
Senger DR, Perruzzi CA, Gracey CF, Papadopoulos A, Tenen DG. Secreted phosphoproteins associated with neoplastic transformation: close homology with plasma proteins cleaved during blood coagulation. Cancer Res 1988; 48(20): 5770–5774
Yokosaki Y, Matsuura N, Sasaki T, Murakami I, Schneider H, Higashiyama S, Saitoh Y, Yamakido M, Taooka Y, Sheppard D. The integrin alpha(9)beta(1) binds to a novel recognition sequence (SVVYGLR) in the thrombin-cleaved amino-terminal fragment of osteopontin. J Biol Chem 1999; 274(51): 36328–36334
Helluin O, Chan C, Vilaire G, Mousa S, DeGrado WF, Bennett JS. The activation state of αvβ3 regulates platelet and lymphocyte adhesion to intact and thrombin-cleaved osteopontin. J Biol Chem 2000; 275(24): 18337–18343
Senger DR, Perruzzi CA, Papadopoulos-Sergiou A, Van de Water L. Adhesive properties of osteopontin: regulation by a naturally occurring thrombin-cleavage in close proximity to the GRGDS cell-binding domain. Mol Biol Cell 1994; 5(5): 565–574
Mi Z, Oliver T, Guo H, Gao C, Kuo PC. Thrombin-cleaved COOH (−) terminal osteopontin peptide binds with cyclophilin C to CD147 in murine breast cancer cells. Cancer Res 2007; 67(9): 4088–4097
Nierodzik ML, Karpatkin S. Thrombin induces tumor growth, metastasis, and angiogenesis: evidence for a thrombin-regulated dormant tumor phenotype. Cancer Cell 2006; 10(5): 355–362
Hu L, Lee M, Campbell W, Perez-Soler R, Karpatkin S. Role of endogenous thrombin in tumor implantation, seeding, and spontaneous metastasis. Blood 2004; 104(9): 2746–2751
Singhal H, Bautista DS, Tonkin KS, O’Malley FP, Tuck AB, Chambers AF, Harris JF. Elevated plasma osteopontin in metastatic breast cancer associated with increased tumor burden and decreased survival. Clin Cancer Res 1997; 3(4): 605–611
Blasberg JD, Pass HI, Goparaju CM, Flores RM, Lee S, Donington JS. Reduction of elevated plasma osteopontin levels with resection of non-small-cell lung cancer. J Clin Oncol 2010; 28(6): 936–941
Zhao J, Dong L, Lu B, Wu G, Xu D, Chen J, Li K, Tong X, Dai J, Yao S, Wu M, Guo Y. Down-regulation of osteopontin suppresses growth and metastasis of hepatocellular carcinoma via induction of apoptosis. Gastroenterology 2008; 135(3): 956–968
Smith LL, Cheung HK, Ling LE, Chen J, Sheppard D, Pytela R, Giachelli CM. Osteopontin N-terminal domain contains a cryptic adhesive sequence recognized by α9β1 integrin. J Biol Chem 1996; 271(45): 28485–28491
Senger DR, Perruzzi CA. Cell migration promoted by a potent GRGDS-containing thrombin-cleavage fragment of osteopontin. Biochim Biophys Acta 1996; 1314(1–2): 13–24
Hasegawa M, Nakoshi Y, Iino T, Sudo A, Segawa T, Maeda M, Yoshida T, Uchida A. Thrombin-cleaved osteopontin in synovial fluid of subjects with rheumatoid arthritis. J Rheumatol 2009; 36(2): 240–245
Radjabi AR, Sawada K, Jagadeeswaran S, Eichbichler A, Kenny HA, Montag A, Bruno K, Lengyel E. Thrombin induces tumor invasion through the induction and association of matrix metalloproteinase-9 andβ1-integrin on the cell surface. J Biol Chem 2008; 283(5): 2822–2834
Young MF, Kerr JM, Termine JD, Wewer UM, Wang MG, McBride OW, Fisher LW. cDNA cloning, mRNA distribution and heterogeneity, chromosomal location, and RFLP analysis of human osteopontin (OPN). Genomics 1990; 7(4): 491–502
Naito M, Matsui A, Inao M, Nagoshi S, Nagano M, Ito N, Egashira T, Hashimoto M, Mishiro S, Mochida S, Fujiwara K. SNPs in the promoter region of the osteopontin gene as a marker predicting the efficacy of interferon-based therapies in patients with chronic hepatitis C. J Gastroenterol 2005; 40(4): 381–388
Giacopelli F, Marciano R, Pistorio A, Catarsi P, Canini S, Karsenty G, Ravazzolo R. Polymorphisms in the osteopontin promoter affect its transcriptional activity. Physiol Genomics 2004; 20(1): 87–96
Hendig D, Arndt M, Szliska C, Kleesiek K, Götting C. SPP1 promoter polymorphisms: identification of the first modifier gene for pseudoxanthoma elasticum. Clin Chem 2007; 53(5): 829–836
Liu CC, Huang SP, Tsai LY, Wu WJ, Juo SH, Chou YH, Huang CH, Wu MT. The impact of osteopontin promoter polymorphisms on the risk of calcium urolithiasis. Clin Chim Acta 2010; 411(9–10): 739–743
D’Alfonso S, Barizzone N, Giordano M, Chiocchetti A, Magnani C, Castelli L, Indelicato M, Giacopelli F, Marchini M, Scorza R, Danieli MG, Cappelli M, Migliaresi S, Bigliardo B, Sabbadini MG, Baldissera E, Galeazzi M, Sebastiani GD, Minisola G, Ravazzolo R, Dianzani U, Momigliano-Richiardi P. Two single-nucleotide polymorphisms in the 5′ and 3′ ends of the osteopontin gene contribute to susceptibility to systemic lupus erythematosus. Arthritis Rheum 2005; 52(2): 539–547
Chiu YW, Tu HF, Wang IK, Wu CH, Chang KW, Liu TY, Kao SY. The implication of osteopontin (OPN) expression and genetic polymorphisms of OPN promoter in oral carcinogenesis. Oral Oncol 2010; 46(4): 302–306
El-Tanani MK, Campbell FC, Kurisetty V, Jin D, McCann M, Rudland PS. The regulation and role of osteopontin in malignant transformation and cancer. Cytokine Growth Factor Rev 2006; 17(6): 463–474
Mochida S, Hashimoto M, Matsui A, Naito M, Inao M, Nagoshi S, Nagano M, Egashira T, Mishiro S, Fujiwara K. Genetic polymorphims in promoter region of osteopontin gene may be a marker reflecting hepatitis activity in chronic hepatitis C patients. Biochem Biophys Res Commun 2004; 313(4): 1079–1085
Sakaki M, Makino R, Hiroishi K, Ueda K, Eguchi J, Hiraide A, Doi H, Omori R, Imawari M. Cyclooxygenase-2 gene promoter polymorphisms affect susceptibility to hepatitis C virus infection and disease progression. Hepatol Res 2010; 40(12): 1219–1226
Shaker OG, Sadik NA, El-Dessouki A. Single-nucleotide polymorphism in the promoter region of the osteopontin gene at nucleotide −443 as a marker predicting the efficacy of pegylated interferon/ribavirin-therapy in Egyptians patients with chronic hepatitis C. Hum Immunol 2012; 73(10): 1039–1045
Yamamoto S, Hijiya N, Setoguchi M, Matsuura K, Ishida T, Higuchi Y, Akizuki S. Structure of the osteopontin gene and its promoter. Ann N Y Acad Sci 1995; 760: 44–58
Schultz J, Lorenz P, Ibrahim SM, Kundt G, Gross G, Kunz M. The functional −443T/C osteopontin promoter polymorphism influences osteopontin gene expression in melanoma cells via binding of c-Myb transcription factor. Mol Carcinog 2009; 48(1): 14–23
Shang S, Plymoth A, Ge S, Feng Z, Rosen HR, Sangrajrang S, Hainaut P, Marrero JA, Beretta L. Identification of osteopontin as a novel marker for early hepatocellular carcinoma. Hepatology 2012; 55(2): 483–490
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Qin, L. Osteopontin is a promoter for hepatocellular carcinoma metastasis: a summary of 10 years of studies. Front. Med. 8, 24–32 (2014). https://doi.org/10.1007/s11684-014-0312-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11684-014-0312-8