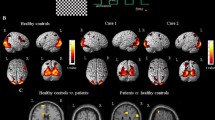
Abstract
Our primary objective was to assess consistent activation and deactivation among healthy participants and patients reporting vertigo. Our secondary aim was to evaluate the influence of the stimulus and the direction of the perception of self-motion We realized a systematic review with an extensive data visualization. We included neuroimaging studies (e.g., functional magnetic resonance imaging [fMRI], positron emission tomography [PET] or near infrared spectroscopy [NIRS]) that have measured functional activity in human adults reporting vertigo and/or dizziness. We included 21 studies (n = 336 participants), ~ 64% male, age ranging from 18 to 80.5 years. The different stimuli used to induce vertigo: caloric stimulation, galvanic stimulation, visual stimulation or vibratory stimulus on neck muscles. We found a consistent activation of the insular cortex, inferior parietal lobule, putamen, cerebellum, anterior cingulate cortex, precentral gyrus, superior temporal gyrus and thalamus. Cortical and subcortical activation seems to have a contralateral pattern to the perception of self-movement. We found a deactivation pattern of structures related to the ventral and third visual pathway. Vertigo is an unpleasant and subjective experience which involves multiple vestibular and non-specific networks with the involvement of a cortico–basal ganglia– cerebellar–thalamic network.
Graphical abstract





Similar content being viewed by others
Data availability
Data are available in Supplementary Information C.
Code availability
R scripts are available contacting with CVR.
References
Azevedo Caldas, M., Freitas Ganança, C., Freitas Ganança, F., Malavasi Ganança, M., & Helena Caovilla, H. (2009). Clinical features of benign paroxysmal positional vertigo. Brazilian Journal of Otorhinolaryngology, 75(4), 502–506. https://doi.org/10.1016/S1808-8694(15)30487-0.
Badre, D., & Nee, D. E. (2018). Frontal Cortex and the Hierarchical Control of Behavior. Trends in Cognitive Sciences, 22(2), 170–188. https://doi.org/10.1016/j.tics.2017.11.005
Baier, B., Bense, S., Birklein, F., Buchholz, H. G., Mischke, A., Schreckenberger, M., & Dieterich, M. (2010). Evidence for modulation of opioidergic activity in central vestibular processing: A [18F] diprenorphine PET study. Human Brain Mapping, 31(4), 550–555. https://doi.org/10.1002/hbm.20886
Barrett, L. F., & Simmons, W. K. (2015). Interoceptive predictions in the brain. Nature Reviews. Neuroscience, 16(7), 419–429. https://doi.org/10.1038/nrn3950
Bisdorff, A., Von Brevern, M., Lempert, T., & Newman-Toker, D. E. (2009). Classification of vestibular symptoms: Towards an international classification of vestibular disorders. Journal of Vestibular Research : Equilibrium & Orientation, 19(1–2), 1–13. https://doi.org/10.3233/VES-2009-0343
Blanke, O. (2012). Multisensory brain mechanisms of bodily self-consciousness. Nature Reviews Neuroscience, 13(8), 556–571. https://doi.org/10.1038/nrn3292
Bottini, G., Karnath, H. O., Vallar, G., Sterzi, R., Frith, C. D., Frackowiak, R. S. J., & Paulesu, E. (2001). Cerebral representations for egocentric space: Functional-anatomical evidence from caloric vestibular stimulation and neck vibration. Brain, 124(6), 1182–1196. https://doi.org/10.1093/brain/124.6.1182
Bottini, G., Sterzi, R., Paulesu, E., Vallar, G., Cappa, S. F., Erminio, F., et al. (1994). Identification of the central vestibular projections in man: A positron emission tomography activation study. Experimental Brain Research, 99(1), 164–169. https://doi.org/10.1007/BF00241421
Cicchetti, D. V., & Allison, T. (1971). A New Procedure for Assessing Reliability of Scoring EEG Sleep Recordings. American Journal of EEG Technology, 11(3), 101–110. https://doi.org/10.1080/00029238.1971.11080840
Corbetta, M., & Shulman, G. L. (2002). Control of goal-directed and stimulus-driven attention in the brain. Nature Reviews Neuroscience, 3(3), 201–215. https://doi.org/10.1038/nrn755
Craig, A. D. (2009). How do you feel — now? The anterior insula and human awareness. Nature Reviews Neuroscience, 10(1), 59–70. https://doi.org/10.1038/nrn2555
Della-Justina, H. M., Gamba, H. R., Lukasova, K., Nucci-da-Silva, M. P., Winkler, A. M., & Amaro, E. (2015). Interaction of brain areas of visual and vestibular simultaneous activity with fMRI. Experimental Brain Research, 233(1), 237–252. https://doi.org/10.1007/s00221-014-4107-6
Della-Morte, D., & Rundek, T. (2012). Dizziness and vertigo. Frontiers of Neurology and Neuroscience, 30, 22–25. https://doi.org/10.1159/000333379
Dieterich, M., Bense, S., Lutz, S., Drzezga, A., Stephan, T., Bartenstein, P., & Brandt, T. (2003). Dominance for vestibular cortical function in the non-dominant hemisphere. Cerebral Cortex, 13(9), 994–1007. https://doi.org/10.1093/cercor/13.9.994
Eickhoff, S. B., Weiss, P. H., Amunts, K., Fink, G. R., & Zilles, K. (2006). Identifying human parieto-insular vestibular cortex using fMRI and cytoarchitectonic mapping. Human Brain Mapping, 27(7), 611–621. https://doi.org/10.1002/hbm.20205
Evans, K. C., Banzett, R. B., Adams, L., McKay, L., Frackowiak, R. S. J., & Corfield, D. R. (2002). BOLD fMRI identifies limbic, paralimbic, and cerebellar activation during air hunger. Journal of Neurophysiology, 88(3), 1500–1511. https://doi.org/10.1152/jn.2002.88.3.1500
Fasold, O., Von Brevern, M., Kuhberg, M., Ploner, C. J., Villringer, A., Lempert, T., & Wenzel, R. (2002). Human vestibular cortex as identified with caloric stimulation in functional magnetic resonance imaging. NeuroImage, 17(3), 1384–1393. https://doi.org/10.1006/nimg.2002.1241
Fink, G. R., Marshall, J. C., Weiss, P. H., Stephan, T., Grefkes, C., Shah, N. J., et al. (2003). Performing allocentric visuospatial judgments with induced distortion of the egocentric reference frame: An fMRI study with clinical implications. NeuroImage, 20(3), 1505–1517. https://doi.org/10.1016/j.neuroimage.2003.07.006
Frank, S. M., Baumann, O., Mattingley, J. B., & Greenlee, M. W. (2014). Vestibular and visual responses in human posterior insular cortex. Journal of Neurophysiology, 112(10), 2481–2491. https://doi.org/10.1152/jn.00078.2014
Frank, S. M., & Greenlee, M. W. (2014). An MRI-compatible caloric stimulation device for the investigation of human vestibular cortex. Journal of Neuroscience Methods, 235, 208–218. https://doi.org/10.1016/j.jneumeth.2014.07.008
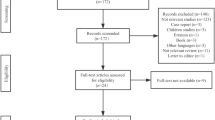
Furlan, A. D., Pennick, V., Bombardier, C., & van Tulder, M. (2009). 2009 updated method guidelines for systematic reviews in the Cochrane Back Review Group. Spine, 34(18), 1929–1941. https://doi.org/10.1097/BRS.0b013e3181b1c99f
Garcia-Larrea, L., & Bastuji, H. (2018). Pain and consciousness. Progress in Neuro-Psychopharmacology & Biological Psychiatry, 87(Pt B), 193–199. https://doi.org/10.1016/j.pnpbp.2017.10.007
Garcia-Larrea, L., & Peyron, R. (2013). Pain matrices and neuropathic pain matrices: A review. Pain, 154(SUPPL. 1), S29–S43. https://doi.org/10.1016/j.pain.2013.09.001
Gavgani, A. M., Wong, R. H. X., Howe, P. R. C., Hodgson, D. M., Walker, F. R., & Nalivaiko, E. (2018). Cybersickness-related changes in brain hemodynamics: A pilot study comparing transcranial Doppler and near-infrared spectroscopy assessments during a virtual ride on a roller coaster. Physiology & Behavior, 191(March), 56–64. https://doi.org/10.1016/j.physbeh.2018.04.007
Haddaway, N. R., Collins, A. M., Coughlin, D., & Kirk, S. (2015). The Role of Google Scholar in Evidence Reviews and Its Applicability to Grey Literature Searching. PLoS ONE, 10(9), e0138237. https://doi.org/10.1371/journal.pone.0138237
Harrison, N. A., Gray, M. A., Gianaros, P. J., & Critchley, H. D. (2010). The embodiment of emotional feelings in the brain. Journal of Neuroscience, 30(38), 12878–12884. https://doi.org/10.1523/JNEUROSCI.1725-10.2010
Higgins, J., & Green, S. (2008). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0[M]. Wiley-Blackwell.
Huntgeburth, S. C., & Petrides, M. (2012). Morphological patterns of the collateral sulcus in the human brain. The European Journal of Neuroscience, 35(8), 1295–1311. https://doi.org/10.1111/j.1460-9568.2012.08031.x
Iida, M., Haida, M., & Igarashi, M. (2009). Vertigo and cerebral hemoglobin changes during unilateral caloric stimulation: A near-infrared spectroscopy study. Annals of the New York Academy of Sciences, 1164, 386–389. https://doi.org/10.1111/j.1749-6632.2009.03869.x
Klingner, C. M., Axer, H., Brodoehl, S., & Witte, O. W. (2016). Vertigo and the processing of vestibular information: A review in the context of predictive coding. Neuroscience and Biobehavioral Reviews, 71, 379–387. https://doi.org/10.1016/j.neubiorev.2016.09.009
Lacadie, C. M., Fulbright, R. K., Rajeevan, N., Constable, R. T., & Papademetris, X. (2008). More accurate Talairach coordinates for neuroimaging using non-linear registration. NeuroImage, 42(2), 717–725. https://doi.org/10.1016/j.neuroimage.2008.04.240
Lobel, E., Kleine, J. F., Le Bihan, D., Leroy-Willig, A., & Berthoz, A. (1998). Functional MRI of galvanic vestibular stimulation. Journal of Neurophysiology, 80(5), 2699–2709. https://doi.org/10.1152/jn.1998.80.5.2699
Lopez-Escamez, J. A., Carey, J., Chung, W.-H., Goebel, J. A., Magnusson, M., Mandalà, M., et al. (2015). Diagnostic criteria for Menière’s disease. Journal of Vestibular Research : Equilibrium & Orientation, 25(1), 1–7. https://doi.org/10.3233/VES-150549
Lopez, C., Blanke, O., & Mast, F. W. (2012). The human vestibular cortex revealed by coordinate-based activation likelihood estimation meta-analysis. Neuroscience, 212, 159–179. https://doi.org/10.1016/j.neuroscience.2012.03.028
Luck, S. J. (2014). An Introduction to the Event-Related Potential Techniqu (2nd ed.). MIT Press.
Lutz, A. (2002). Toward a neurophenomenology as an account of generative passages: A first empirical case study. Phenomenology and the Cognitive Sciences, 1(2), 133–167. https://doi.org/10.1023/A:1020320221083
Marcelli, V., Esposito, F., Aragri, A., Furia, T., Riccardi, P., Tosetti, M., et al. (2009). Spatio-temporal pattern of vestibular information processing after brief caloric stimulation. European Journal of Radiology, 70(2), 312–316. https://doi.org/10.1016/j.ejrad.2008.01.042
McHugh, M. L. (2012). Interrater reliability: The kappa statistic. Biochemia Medica, 22(3), 276–282. https://doi.org/10.11613/bm.2012.031.
Müller, V. I., Cieslik, E. C., Laird, A. R., Fox, P. T., Radua, J., Mataix-Cols, D., et al. (2018). Ten simple rules for neuroimaging meta-analysis. Neuroscience and Biobehavioral Reviews, 84, 151–161. https://doi.org/10.1016/j.neubiorev.2017.11.012
Newman-Toker, D. E., Cannon, L. M., Stofferahn, M. E., Rothman, R. E., Hsieh, Y.-H., & Zee, D. S. (2007). Imprecision in patient reports of dizziness symptom quality: A cross-sectional study conducted in an acute care setting. Mayo Clinic Proceedings, 82(11), 1329–1340. https://doi.org/10.4065/82.11.1329
Oosterwijk, S., Lindquist, K. A., Anderson, E., Dautoff, R., Moriguchi, Y., & Barrett, L. F. (2012). States of mind: Emotions, body feelings, and thoughts share distributed neural networks. NeuroImage, 62(3), 2110–2128. https://doi.org/10.1016/j.neuroimage.2012.05.079.
Ouzzani, M., Hammady, H., Fedorowicz, Z., & Elmagarmid, A. (2016). Rayyan—a web and mobile app for systematic reviews. Systematic Reviews, 5(1), 210. https://doi.org/10.1186/s13643-016-0384-4
Page, M. J., McKenzie, J. E., Bossuyt, P. M., Boutron, I., Hoffmann, T. C., Mulrow, C. D., et al. (2021). The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ, 372, n71. https://doi.org/10.1136/bmj.n71
Pierce, J. E., & Péron, J. (2020). The basal ganglia and the cerebellum in human emotion. Social Cognitive and Affective Neuroscience, 15(5), 599–613. https://doi.org/10.1093/scan/nsaa076
Pitcher, D., & Ungerleider, L. G. (2021). Evidence for a Third Visual Pathway Specialized for Social Perception. Trends in Cognitive Sciences, 25(2), 100–110. https://doi.org/10.1016/j.tics.2020.11.006
Purves, D., Augustine, G. J., Fitzpatrick, D., Hall, W. C., LaMantia, A.-S., Mooney, R. D., et al. (Eds.). (2018a). The Vestibular System. In Neuroscience (6th ed., pp. 305–321). Oxford University Press.
Purves, D., Augustine, G. J., Fitzpatrick, D., Hall, W. C., LaMantia, A.-S., Mooney, R. D., et al. (Eds.). (2018b). Central Visual Pathways. In Neuroscience (6th ed., pp. 261–280). Oxford University Press.
R Core Team. (2021). R: A language and environment for statistical computing. Vienna, Austria: R Foundation for Statistical Computing. https://www.r-project.org/.
Roberts, R. E., Ahmad, H., Arshad, Q., Patel, M., Dima, D., Leech, R., et al. (2017). Functional neuroimaging of visuo-vestibular interaction. Brain Structure and Function, 222(5), 2329–2343. https://doi.org/10.1007/s00429-016-1344-4
Roberts, R. E., Ahmad, H., Patel, M., Dima, D., Ibitoye, R., Sharif, M., et al. (2018). An fMRI study of visuo-vestibular interactions following vestibular neuritis. NeuroImage: Clinical, 20(February), 1010–1017. https://doi.org/10.1016/j.nicl.2018.10.007.
RStudio Team. (2021). RStudio: Integrated Development Environment for R. Boston, MA. http://www.rstudio.com/.
Russo, A., Marcelli, V., Esposito, F., Corvino, V., Marcuccio, L., Giannone, A., et al. (2014). Abnormal thalamic function in patients with vestibular migraine. Neurology, 82(23), 2120 LP–2126. https://doi.org/10.1212/WNL.0000000000000496.
Seeley, W. W., Menon, V., Schatzberg, A. F., Keller, J., Glover, G. H., Kenna, H., et al. (2007). Dissociable intrinsic connectivity networks for salience processing and executive control. The Journal of Neuroscience : The Official Journal of the Society for Neuroscience, 27(9), 2349–2356. https://doi.org/10.1523/JNEUROSCI.5587-06.2007
Seth, A. (2016). The real problem. Aeon. https://aeon.co/essays/the-hard-problem-of-consciousness-is-a-distraction-from-the-real-one. Accessed 11 January 2022.
Seth, A. (2021). Being you: A new science of consciousness. Faber & Faber.
Shariff, S. Z., Bejaimal, S. A., Sontrop, J. M., Iansavichus, A. V., Haynes, R. B., Weir, M. A., & Garg, A. X. (2013). Retrieving clinical evidence: A comparison of PubMed and Google Scholar for quick clinical searches. Journal of Medical Internet Research, 15(8), e164. https://doi.org/10.2196/jmir.2624
Slim, K., Nini, E., Forestier, D., Kwiatkowski, F., Panis, Y., & Chipponi, J. (2003). Methodological index for non-randomized studies (minors): Development and validation of a new instrument. ANZ Journal of Surgery, 73(9), 712–716. https://doi.org/10.1046/j.1445-2197.2003.02748.x
Starr, C. J., Sawaki, L., Wittenberg, G. F., Burdette, J. H., Oshiro, Y., Quevedo, A. S., et al. (2011). The contribution of the putamen to sensory aspects of pain: Insights from structural connectivity and brain lesions. Brain : A Journal of Neurology, 134(Pt 7), 1987–2004. https://doi.org/10.1093/brain/awr117
Sterne, J. A. C., Hernán, M. A., Reeves, B. C., Savović, J., Berkman, N. D., Viswanathan, M., et al. (2016). ROBINS-I: A tool for assessing risk of bias in non-randomised studies of interventions. BMJ, 355, i4919. https://doi.org/10.1136/bmj.i4919
Stilwell, P., & Harman, K. (2019). An enactive approach to pain: Beyond the biopsychosocial model. Phenomenology and the Cognitive Sciences, 18(4), 637–665. https://doi.org/10.1007/s11097-019-09624-7
Suzuki, M., Kitano, H., Ito, R., Kitanishi, T., Yazawa, Y., Ogawa, T., et al. (2001). Cortical and subcortical vestibular response to caloric stimulation detected by functional magnetic resonance imaging. Brain Research. Cognitive Brain Research, 12(3), 441–449. https://doi.org/10.1016/s0926-6410(01)00080-5
Takeda, N., Hashikawa, K., Moriwaki, H., Oku, N., Koizuka, I., Kitahara, T., et al. (1996). Effects of caloric vestibular stimulation on parietal and temporal blood flow in human brain: A consecutive technetium-99m-HMPAO spect study. Journal of Vestibular Research : Equilibrium & Orientation, 6(2), 127–134.
The Jamovi Project. (2021). jamovi (Version 1.6). https://www.jamovi.org.
Ungerleider, L. G., & Mishkin, M. (1982). Two cortical visual systems. In D. J. Ingle, M. A. Goodale, & R. J. W. Mansfield (Eds.), Analysis of visual behavior (pp. 549–586). MIT Press.
van den Heuvel, M. P., & Sporns, O. (2011). Rich-club organization of the human connectome. The Journal of Neuroscience : The Official Journal of the Society for Neuroscience, 31(44), 15775–15786. https://doi.org/10.1523/JNEUROSCI.3539-11.2011
Varela, F. (1996). Neurophenomenology: A Methodological Remedy for the Hard Problem. Journal of Consciousness Studies, 3(4), 330–349.
Vitte, E., Derosier, C., Caritu, Y., Berthoz, A., Hasboun, D., & Soulié, D. (1996). Activation of the hippocampal formation by vestibular stimulation: A functional magnetic resonance imaging study. Experimental Brain Research, 112(3), 523–526. https://doi.org/10.1007/BF00227958
Wang, Z., Chen, M., Gong, X., Huang, W., Xu, L., & Zhou, C. (2008). Why cold water delays the onset of vestibular vertigo-An functional MRI study. European Journal of Radiology, 67(3), 459–465. https://doi.org/10.1016/j.ejrad.2007.08.021
Woolf, C. J. (2011). Central sensitization: Implications for the diagnosis and treatment of pain. Pain, 152(3 Suppl), S2-15. https://doi.org/10.1016/j.pain.2010.09.030
Wu, W. (2018). The Neuroscience of Consciousness. (E. N. Zalta, Ed.)The Stanford Encyclopedia of Philosophy. Metaphysics Research Lab, Stanford University. https://plato.stanford.edu/archives/win2018/entries/consciousness-neuroscience. Accessed 1 March 2022.
Xu, A., Larsen, B., Baller, E. B., Scott, J. C., Sharma, V., Adebimpe, A., et al. (2020). Convergent neural representations of experimentally-induced acute pain in healthy volunteers: A large-scale fMRI meta-analysis. Neuroscience & Biobehavioral Reviews, 112, 300–323. https://doi.org/10.1016/j.neubiorev.2020.01.004.
Yale BioImage Suite. (2020). MNI2TAL v. 1.2.0. https://bioimagesuiteweb.github.io/webapp/mni2tal.html.
Yeo, B. T. T., Krienen, F. M., Sepulcre, J., Sabuncu, M. R., Lashkari, D., Hollinshead, M., et al. (2011). The organization of the human cerebral cortex estimated by intrinsic functional connectivity. Journal of Neurophysiology, 106(3), 1125–1165. https://doi.org/10.1152/jn.00338.2011
Zhu, Q., Chen, W., Cui, Y., Wu, J., Shu, L., Sun, X., et al. (2021). Structural and Functional Changes in the Cerebellum and Brainstem in Patients with Benign Paroxysmal Positional Vertigo. Cerebellum, 20(5), 804–809. https://doi.org/10.1007/s12311-021-01237-8
zu Eulenburg, P., Caspers, S., Roski, C., & Eickhoff, S. B. (2012). Meta-analytical definition and functional connectivity of the human vestibular cortex. NeuroImage, 60(1), 162–169https://doi.org/10.1016/j.neuroimage.2011.12.032.
Author information
Authors and Affiliations
Contributions
CVR designed the systematic review, designed the search strategy, realized the inclusion phase, analyzed the methodological quality and risk of bias, realized the data extraction, analyzed the data, realized the data visualization and wrote the draft. AHG realized the inclusion phase, review the data extraction and critically appraised the draft. JDN analyzed the methodological quality and risk of bias, review the data extraction and critically appraised the manuscript. LSM helped in the management of the project, helped to reach consensus in the analysis of the methodological quality and risk of bias, and critically appraised the manuscript. FCM helped in the management of the project, helped to reach consensus in the inclusion phase, and critically appraised the manuscript.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare no conflicts of interest.
Ethics approval; Consent to participate; and Consent for publication
Not applicable.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Varangot-Reille, C., Herranz-Gomez, A., de la Nava, J. et al. The experience of vertigo: A systematic review of neuroimaging studies. Brain Imaging and Behavior 16, 2797–2808 (2022). https://doi.org/10.1007/s11682-022-00729-3
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11682-022-00729-3