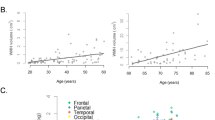
Abstract
White matter hyperintensities (WMHs) on magnetic resonance imaging are commonly found in older adults. The mechanisms underpinning the dose-dependent association between WMH severity and cognition are not well understood. This study aimed to investigate how brain activity changes with WMH severity, and if altered brain activity mediates the relationship between WMH and cognitive function. A total of 35 participants with moderate to severe WMHs (Fazekas grade 2 or 3) and 34 participants with mild WMHs (Fazekas grade 1), who were cognitively normal, were included. Resting-state brain function was analyzed using the amplitude of low-frequency fluctuation (ALFF). A mean fractional anisotropy (FA) value of 20 tract-specific regions of interest was calculated. Mediation analysis was used to assess whether ALFF values mediated the relationship between WMH and cognition. The results showed that compared to those with mild WMHs, participants with confluent WMHs had worse memory and naming ability and also had increased ALFF in the right middle frontal gyrus and decreased ALFF in the left middle occipital gyrus. After controlling for age, gender, education and apolipoprotein E (ApoE) ε4 status, increased ALFF in the right prefrontal cortex was associated with worse immediate recall and recognition, and ALFF values mediated the relationships between both Fazekas scores and FA values and memory. In conclusion, our study suggests that cognitively normal adults with high WMH load exhibit subclinical cognitive dysfunction and altered spontaneous brain activity. The mediating effects of brain activity help to shed light on our understanding of the relationship between WMHs and cognition.


Similar content being viewed by others
References
Baron, R. M., & Kenny, D. A. (1986). The moderator-mediator variable distinction in social psychological research: Conceptual, strategic, and statistical considerations. Journal of Personality and Social Psychology, 51(6), 1173–1182. https://doi.org/10.1037//0022-3514.51.6.1173
Benson, G., Hildebrandt, A., Lange, C., Schwarz, C., Kobe, T., Sommer, W., Floel, A., & Wirth, M. (2018). Functional connectivity in cognitive control networks mitigates the impact of white matter lesions in the elderly. Alzheimer’s Research Theraphy, 10(1), 109. https://doi.org/10.1186/s13195-018-0434-3
Chen, X., Huang, L., Ye, Q., Yang, D., Qin, R., Luo, C., Li, M., Zhang, B., & Xu, Y. (2019). Disrupted functional and structural connectivity within default mode network contribute to WMH-related cognitive impairment. Neuroimage Clinical, 24, 102088. https://doi.org/10.1016/j.nicl.2019.102088
Cheng, R., Qi, H., Liu, Y., Zhao, S., Li, C., Liu, C., & Zheng, J. (2017). Abnormal amplitude of low-frequency fluctuations and functional connectivity of resting-state functional magnetic resonance imaging in patients with leukoaraiosis. Brain and Behavior: A Cognitive Neuroscience Perspective, 7(6), e00714. https://doi.org/10.1002/brb3.714
Cox, S. R., Bastin, M. E., Ferguson, K. J., Allerhand, M., Royle, N. A., Maniega, S. M., Starr, J. M., MacLullich, A. M., Wardlaw, J. M., Deary, I. J., & MacPherson, S. E. (2015). Compensation or inhibitory failure? Testing hypotheses of age-related right frontal lobe involvement in verbal memory ability using structural and diffusion MRI. Cortex, 63, 4–15. https://doi.org/10.1016/j.cortex.2014.08.001
Cui, Z., Zhong, S., Xu, P., He, Y., & Gong, G. (2013). PANDA: A pipeline toolbox for analyzing brain diffusion images. Frontiers in Human Neuroscience, 7, 42. https://doi.org/10.3389/fnhum.2013.00042
de Leeuw, F. E., de Groot, J. C., Achten, E., Oudkerk, M., Ramos, L. M., Heijboer, R., Hofman, A., Jolles, J., van Gijn, J., & Breteler, M. M. (2001). Prevalence of cerebral white matter lesions in elderly people: a population based magnetic resonance imaging study. The Rotterdam Scan Study. Journal of Neurology, Neurosurgery & Psychiatry, 70(1), 9–14. https://doi.org/10.1136/jnnp.70.1.9
Debette, S., & Markus, H. S. (2010). The clinical importance of white matter hyperintensities on brain magnetic resonance imaging: Systematic review and meta-analysis. BMJ, 341, c3666. https://doi.org/10.1136/bmj.c3666
Ding, X., Ding, J., Hua, B., Xiong, X., Xiao, L., Peng, F., Chen, L., Pan, X., & Wang, Q. (2017). Abnormal cortical functional activity in patients with ischemic white matter lesions: A resting-state functional magnetic resonance imaging study. Neuroscience Letters, 644, 10–17. https://doi.org/10.1016/j.neulet.2017.02.015
Ding, J. R., Ding, X., Hua, B., Xiong, X., Wen, Y., Ding, Z., Wang, Q., & Thompson, P. (2018). Altered connectivity patterns among resting state networks in patients with ischemic white matter lesions. Brain Imaging and Behavior, 12(5), 1239–1250. https://doi.org/10.1007/s11682-017-9793-9
Disner, S. G., Marquardt, C. A., Mueller, B. A., Burton, P. C., & Sponheim, S. R. (2018). Spontaneous neural activity differences in posttraumatic stress disorder: A quantitative resting-state meta-analysis and fMRI validation. Human Brain Mapping, 39(2), 837–850. https://doi.org/10.1002/hbm.23886
Dupont, P. S., Bocti, C., Joannette, M., Lavallee, M. M., Nikelski, J., Vallet, G. T., Chertkow, H., & Joubert, S. (2019). Amyloid burden and white matter hyperintensities mediate age-related cognitive differences. Neurobiology of Aging. https://doi.org/10.1016/j.neurobiolaging.2019.08.025
Fazekas, F., Chawluk, J. B., Alavi, A., Hurtig, H. I., & Zimmerman, R. A. (1987). MR signal abnormalities at 1.5 T in Alzheimer’s dementia and normal aging. AJR American Journal of Roentgenology, 149(2), 351–356. https://doi.org/10.2214/ajr.149.2.351
Folstein, M. F., Folstein, S. E., & McHugh, P. R. (1975). “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. Journal of Psychiatric Research, 12(3), 189–198.
Griebe, M., Amann, M., Hirsch, J. G., Achtnichts, L., Hennerici, M. G., Gass, A., & Szabo, K. (2014). Reduced functional reserve in patients with age-related white matter changes: A preliminary FMRI study of working memory. PLoS ONE, 9(8), e103359. https://doi.org/10.1371/journal.pone.0103359
Kloppenborg, R. P., Nederkoorn, P. J., Geerlings, M. I., & van den Berg, E. (2014). Presence and progression of white matter hyperintensities and cognition: A meta-analysis. Neurology, 82(23), 2127–2138. https://doi.org/10.1212/WNL.0000000000000505
Lee, M. H., Smyser, C. D., & Shimony, J. S. (2013). Resting-state fMRI: A review of methods and clinical applications. AJNR. American Journal of Neuroradiology, 34(10), 1866–1872. https://doi.org/10.3174/ajnr.A3263
Li, C., Yang, J., Yin, X., Liu, C., Zhang, L., Zhang, X., Gui, L., & Wang, J. (2015). Abnormal intrinsic brain activity patterns in leukoaraiosis with and without cognitive impairment. Behavioural Brain Research, 292, 409–413. https://doi.org/10.1016/j.bbr.2015.06.033
Maj, M., Delia, L., Satz, P., Janssen, R., Zaudig, M., Uchiyama, C., Starace, F., Galderisi, S., Chervinsky, A., & World Health Organization, D. o. M. H. G. P. o. A. (1993). Evaluation of two new neuropsychological tests designed to minimize cultural bias in the assessment of HIV-1 seropositive persons: A WHO study. Archives of Clinical Neuropsychology, 8(2), 123–135.
Morris, J. C. (1993). The clinical dementia rating (CDR): Current version and scoring rules. Neurology, 43(11), 2412–2414. https://doi.org/10.1212/wnl.43.11.2412-a
Pan, P., Zhu, L., Yu, T., Shi, H., Zhang, B., Qin, R., Zhu, X., Qian, L., Zhao, H., Zhou, H., & Xu, Y. (2017). Aberrant spontaneous low-frequency brain activity in amnestic mild cognitive impairment: A meta-analysis of resting-state fMRI studies. Ageing Research Reviews, 35, 12–21. https://doi.org/10.1016/j.arr.2016.12.001
Prins, N. D., & Scheltens, P. (2015). White matter hyperintensities, cognitive impairment and dementia: An update. Nature Reviews. Neurology, 11(3), 157–165. https://doi.org/10.1038/nrneurol.2015.10
Rabin, J. S., Perea, R. D., Buckley, R. F., Neal, T. E., Buckner, R. L., Johnson, K. A., Sperling, R. A., & Hedden, T. (2019). Global white matter diffusion characteristics predict longitudinal cognitive change independently of amyloid status in clinically normal older adults. Cerebral Cortex, 29(3), 1251–1262. https://doi.org/10.1093/cercor/bhy031
Scheltens, P., Leys, D., Barkhof, F., Huglo, D., Weinstein, H. C., Vermersch, P., Kuiper, M., Steinling, M., Wolters, E. C., & Valk, J. (1992). Atrophy of medial temporal lobes on MRI in “probable” Alzheimer’s disease and normal ageing: Diagnostic value and neuropsychological correlates. Journal of Neurology, Neurosurgery and Psychiatry, 55(10), 967–972. https://doi.org/10.1136/jnnp.55.10.967
Tsai, M. S., Tangalos, E. G., Petersen, R. C., Smith, G. E., Schaid, D. J., Kokmen, E., Ivnik, R. J., & Thibodeau, S. N. (1994). Apolipoprotein E: Risk factor for Alzheimer disease. American Journal of Human Genetics, 54(4), 643–649.
Verdelho, A., Madureira, S., Moleiro, C., Ferro, J. M., Santos, C. O., Erkinjuntti, T., Pantoni, L., Fazekas, F., Visser, M., Waldemar, G., Wallin, A., Hennerici, M., Inzitari, D., & Study, L. (2010). White matter changes and diabetes predict cognitive decline in the elderly: The LADIS study. Neurology, 75(2), 160–167. https://doi.org/10.1212/WNL.0b013e3181e7ca05
Xing, Y., Yang, J., Zhou, A., Wang, F., Wei, C., Tang, Y., & Jia, J. (2021). White matter fractional anisotropy is a superior predictor for cognitive impairment than brain volumes in older adults with confluent white matter hyperintensities. Frontiers in Psychiatry, 12, 633811. https://doi.org/10.3389/fpsyt.2021.633811
Yan, C. G., Wang, X. D., Zuo, X. N., & Zang, Y. F. (2016). DPABI: data processing & analysis for (resting-state) brain imaging. Neuroinformatics, 14(3), 339–351. https://doi.org/10.1007/s12021-016-9299-4
Yesavage, J. A., Brink, T. L., Rose, T. L., Lum, O., Huang, V., Adey, M., & Leirer, V. O. (1982). Development and validation of a geriatric depression screening scale: A preliminary report. Journal of Psychiatric Research, 17(1), 37–49.
Zang, Y. F., He, Y., Zhu, C. Z., Cao, Q. J., Sui, M. Q., Liang, M., Tian, L. X., Jiang, T. Z., & Wang, Y. F. (2007). Altered baseline brain activity in children with ADHD revealed by resting-state functional MRI. Brain & Development, 29(2), 83–91. https://doi.org/10.1016/j.braindev.2006.07.002
Zeng, W., Chen, Y., Zhu, Z., Gao, S., Xia, J., Chen, X., Jia, J., & Zhang, Z. (2019). Severity of white matter hyperintensities: Lesion patterns, cognition, and microstructural changes. Journal of Cerebral Blood Flow & Metabolism. https://doi.org/10.1177/0271678X19893600
Acknowledgements
We thank Dan Yao for operating the MRI scanner and Yunxia Wang for MRI data analysis.
Funding
This work was supported by National Natural Science Foundation of China (81701044, 81671040 and 81970996) and National Key R&D Program of China (2016YFC1306305, 2017YFC1310102 and 2019YFC0118200).
Author information
Authors and Affiliations
Contributions
YX, YT and JJ designed this study. JY was responsible for data collection. AZ and FW enrolled participants and evaluated cognitive function. YX drafted the manuscript, and all authors revised it and agreed the final version to be published.
Corresponding authors
Ethics declarations
Informed consent
Written informed consent was obtained from all participants. This study was approved by the Institutional Review Board of Xuanwu Hospital.
Conflict of interest
The authors have no conflict of interest to report.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Xing, Y., Yang, J., Zhou, A. et al. Altered brain activity mediates the relationship between white matter hyperintensity severity and cognition in older adults. Brain Imaging and Behavior 16, 899–908 (2022). https://doi.org/10.1007/s11682-021-00564-y
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11682-021-00564-y