Abstract
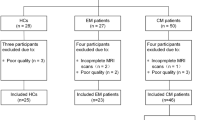
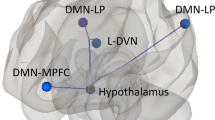
Cluster headache (CH) shows a more severe clinical picture than migraine (Mig). We tested whether brain changes can explain such difference. Multimodal MRI was acquired in attack-free patients with CH (n = 12), Mig (n = 13) and in normal controls (NC, n = 13). We used FSL for MRI data analysis and nonparametric permutation testing for voxelwise analyses (p < 0.01, corrected). CH showed lower grey matter (GM) volume, compared to Mig and NC, in frontal cortex regions (inferior frontal gyrus and frontal pole [FP], respectively) and, only compared to Mig, in lateral occipital cortex (LOC). Functional connectivity (FC) of CH was higher than Mig and NC within working memory and executive control networks and, only compared to Mig, between cerebellar and auditory language comprehension networks. In the attack-free state, the CH brain seems to be characterized by: (i) GM volume decrease, compared to both Mig and NC, in pain modulation regions (FP) and, only with respect to Mig, in a region of visual processing modulation during pain and working memory (LOC); (ii) increased FC at short range compared to both Mig and NC and at long range only with respect to Mig, in key cognitive networks, likely due to maladaptation towards more severe pain experience.



Similar content being viewed by others
References
(IHS) HCCotIHS. (2013). The international classification of headache disorders, 3rd edition (beta version). Cephalalgia, 33(9), 629–808.
Absinta, M., Rocca, M. A., Colombo, B., Falini, A., Comi, G., & Filippi, M. (2012). Selective decreased grey matter volume of the pain-matrix network in cluster headache. Cephalalgia, 32(2), 109–115.
Arkink, E. B., Schmitz, N., Schoonman, G. G., van Vliet, J. A., Haan, J., van Buchem, M. A., Ferrari, M. D., & Kruit, M. C. (2017). The anterior hypothalamus in cluster headache. Cephalalgia, 37(11), 1039–1050.
Baliki, M. N., Mansour, A. R., Baria, A. T., & Apkarian, A. V. (2014). Functional reorganization of the default mode network across chronic pain conditions. PLoS One, 9(9), e106133.
Baumann, O., & Mattingley, J. B. (2010). Scaling of neural responses to visual and auditory motion in the human cerebellum. The Journal of Neuroscience, 30(12), 4489–4495.
Berryman, C., Stanton, T. R., Jane Bowering, K., Tabor, A., McFarlane, A., & Lorimer Moseley, G. (2013). Evidence for working memory deficits in chronic pain: a systematic review and meta-analysis. Pain, 154(8), 1181–1196.
Bingel, U., Rose, M., Glascher, J., & Buchel, C. (2007). fMRI reveals how pain modulates visual object processing in the ventral visual stream. Neuron, 55(1), 157–167.
Chen, Z., Chen, X., Liu, M., Liu, S., Shu, S., Ma, L., & Yu, S. (2016). Altered functional connectivity of the marginal division in migraine: a resting-state fMRI study. The Journal of Headache and Pain, 17(1), 89.
Chou, K. H., Yang, F. C., Fuh, J. L., Huang, C. C., Lirng, J. F., Lin, Y. Y., Lee, P. L., Kao, H. W., Lin, C. P., & Wang, S. J. (2014). Altered white matter microstructural connectivity in cluster headaches: a longitudinal diffusion tensor imaging study. Cephalalgia, 34(13), 1040–1052.
Dobromyslin, V. I., Salat, D. H., Fortier, C. B., Leritz, E. C., Beckmann, C. F., Milberg, W. P., & McGlinchey, R. E. (2012). Distinct functional networks within the cerebellum and their relation to cortical systems assessed with independent component analysis. Neuroimage, 60(4), 2073–2085.
Dresler, T., Lurding, R., Paelecke-Habermann, Y., Gaul, C., Henkel, K., Lindwurm-Spath, A., Leinisch, E., & Jurgens, T. P. (2012). Cluster headache and neuropsychological functioning. Cephalalgia, 32(11), 813–821.
Fazekas, F., Chawluk, J. B., Alavi, A., Hurtig, H. I., & Zimmerman, R. A. (1987). MR signal abnormalities at 1.5 T in Alzheimer's dementia and normal aging. AJR. American Journal of Roentgenology, 149(2), 351–356.
Granziera, C., DaSilva, A. F., Snyder, J., Tuch, D. S., & Hadjikhani, N. (2006). Anatomical alterations of the visual motion processing network in migraine with and without aura. PLoS Medicine, 3(10), e402.
Habas, C., Guillevin, R., & Abanou, A. (2011). Functional connectivity of the superior human temporal sulcus in the brain resting state at 3T. Neuroradiology, 53(2), 129–140.
Hadjipavlou, G., Dunckley, P., Behrens, T. E., & Tracey, I. (2006). Determining anatomical connectivities between cortical and brainstem pain processing regions in humans: a diffusion tensor imaging study in healthy controls. Pain, 123(1–2), 169–178.
Hoffmann, J., & May, A. (2018). Diagnosis, pathophysiology, and management of cluster headache. Lancet Neurology, 17(1), 75–83.
Jenkinson, M., & Smith, S. (2001). A global optimisation method for robust affine registration of brain images. Medical Image Analysis, 5(2), 143–156.
Jin, C., Yuan, K., Zhao, L., Yu, D., von Deneen, K. M., Zhang, M., Qin, W., Sun, W., & Tian, J. (2013). Structural and functional abnormalities in migraine patients without aura. NMR in Biomedicine, 26(1), 58–64.
Kim, J. H., Suh, S. I., Seol, H. Y., Oh, K., Seo, W. K., Yu, S. W., Park, K. W., & Koh, S. B. (2008). Regional grey matter changes in patients with migraine: a voxel-based morphometry study. Cephalalgia, 28(6), 598–604.
Liu, G., Ma, H. J., Hu, P. P., Tian, Y. H., Hu, S., Fan, J., & Wang, K. (2013). Effects of painful stimulation and acupuncture on attention networks in healthy subjects. Behavioral and Brain Functions, 9, 23.
McLachlan, N. M., & Wilson, S. J. (2017). The contribution of brainstem and cerebellar pathways to auditory recognition. Frontiers in Psychology, 8, 265.
Messina, R., Rocca, M. A., Colombo, B., Valsasina, P., Horsfield, M. A., Copetti, M., Falini, A., Comi, G., & Filippi, M. (2013). Cortical abnormalities in patients with migraine: a surface-based analysis. Radiology, 268(1), 170–180.
Moberget, T., & Ivry, R. B. (2016). Cerebellar contributions to motor control and language comprehension: searching for common computational principles. Annals of the New York Academy of Sciences, 1369(1), 154–171.
Naegel, S., Holle, D., Desmarattes, N., Theysohn, N., Diener, H. C., Katsarava, Z., & Obermann, M. (2014a). Cortical plasticity in episodic and chronic cluster headache. NeuroImage: Clinical, 6, 415–423.
Naegel, S., Holle, D., & Obermann, M. (2014b). Structural imaging in cluster headache. Current Pain and Headache Reports, 18(5), 415.
Neeb, L., Bastian, K., Villringer, K., Gits, H. C., Israel, H., Reuter, U., & Fiebach, J. B. (2015). No microstructural white matter alterations in chronic and episodic migraineurs: a case-control diffusion tensor magnetic resonance imaging study. Headache, 55(2), 241–251.
Oguz, I., Farzinfar, M., Matsui, J., Budin, F., Liu, Z., Gerig, G., Johnson, H. J., & Styner, M. (2014). DTIPrep: quality control of diffusion-weighted images. Frontiers in Neuroinformatics, 8, 4.
Pruim, R. H., Mennes, M., van Rooij, D., Llera, A., Buitelaar, J. K., & Beckmann, C. F. (2015). ICA-AROMA: a robust ICA-based strategy for removing motion artifacts from fMRI data. Neuroimage, 112, 267–277.
Qiu, E., Wang, Y., Ma, L., Tian, L., Liu, R., Dong, Z., Xu, X., Zou, Z., & Yu, S. (2013). Abnormal brain functional connectivity of the hypothalamus in cluster headaches. PLoS One, 8(2), e57896.
Rocca, M. A., Ceccarelli, A., Falini, A., Colombo, B., Tortorella, P., Bernasconi, L., Comi, G., Scotti, G., & Filippi, M. (2006). Brain gray matter changes in migraine patients with T2-visible lesions: a 3-T MRI study. Stroke, 37(7), 1765–1770.
Sprenger, T., Ruether, K. V., Boecker, H., Valet, M., Berthele, A., Pfaffenrath, V., Woller, A., & Tolle, T. R. (2007). Altered metabolism in frontal brain circuits in cluster headache. Cephalalgia, 27(9), 1033–1042.
Szabo, N., Kincses, Z. T., Pardutz, A., Toth, E., Szok, D., Csete, G., & Vecsei, L. (2013). White matter disintegration in cluster headache. The Journal of Headache and Pain, 14, 64.
Teepker, M., Menzler, K., Belke, M., Heverhagen, J. T., Voelker, M., Mylius, V., Oertel, W. H., Rosenow, F., & Knake, S. (2012). Diffusion tensor imaging in episodic cluster headache. Headache, 52(2), 274–282.
Torkamani, M., Ernst, L., Cheung, L. S., Lambru, G., Matharu, M., & Jahanshahi, M. (2015). The neuropsychology of cluster headache: cognition, mood, disability, and quality of life of patients with chronic and episodic cluster headache. Headache, 55(2), 287–300.
Valfre, W., Rainero, I., Bergui, M., & Pinessi, L. (2008). Voxel-based morphometry reveals gray matter abnormalities in migraine. Headache, 48(1), 109–117.
Yang, F. C., Chou, K. H., Fuh, J. L., Huang, C. C., Lirng, J. F., Lin, Y. Y., Lin, C. P., & Wang, S. J. (2013). Altered gray matter volume in the frontal pain modulation network in patients with cluster headache. Pain, 154(6), 801–807.
Yang, F. C., Chou, K. H., Fuh, J. L., Lee, P. L., Lirng, J. F., Lin, Y. Y., Lin, C. P., & Wang, S. J. (2014). Altered hypothalamic functional connectivity in cluster headache: a longitudinal resting-state functional MRI study. Journal of Neurology, Neurosurgery, and Psychiatry, 86(4), 437–445.
Yu, D., Yuan, K., Qin, W., Zhao, L., Dong, M., Liu, P., Yang, X., Liu, J., Sun, J., Zhou, G., von Deneen, K. M., & Tian, J. (2013). Axonal loss of white matter in migraine without aura: a tract-based spatial statistics study. Cephalalgia, 33(1), 34–42.
Yu, Z. B., Lv, Y. B., Song, L. H., Liu, D. H., Huang, X. L., Hu, X. Y., Zuo, Z. W., Wang, Y., Yang, Q., Peng, J., Zhou, Z. H., & Li, H. T. (2017). Functional connectivity differences in the insular sub-regions in migraine without Aura: a resting-state functional magnetic resonance imaging study. Frontiers in Behavioral Neuroscience, 11, 124.
Yuan, K., Qin, W., Liu, P., Zhao, L., Yu, D., Dong, M., Liu, J., Yang, X., von Deneen, K. M., Liang, F., & Tian, J. (2012). Reduced fractional anisotropy of corpus callosum modulates inter-hemispheric resting state functional connectivity in migraine patients without aura. PLoS One, 7(9), e45476.
Acknowledgements
Riccardo Tappa Brocci (University of Siena) helped with MRI data acquisition; Mary Lokken revised the manuscript for English language editing.
Author information
Authors and Affiliations
Contributions
Antonio Giorgio, study concept, acquisition, analysis and interpretation of data, manuscript writing. Chiara Lupi, study concept and design, acquisition of data, manuscript writing. Jian Zhang, acquisition, analysis of data. Francesco De Cesaris, acquisition of data. Mario Alessandri, acquisition of data. Marzia Mortilla, acquisition of data. Antonio Federico, critical revision of manuscript for intellectual content. Pierangelo Geppetti, critical revision of manuscript for intellectual content. Nicola De Stefano, critical revision of manuscript for intellectual content. Silvia Benemei, study concept and design, study supervision, critical revision of manuscript for intellectual content.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest specific for this study.
Ethical approval
All procedures performed in this study were in accordance with the ethical standards of the local Institutional Ethics Committee on Clinical Research and with the Helsinki Declaration (version amended during the 64th WMA General Assembly, Fortaleza, Brazil, October 2013). Written informed consent was obtained from all participants before study entry.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Co-investigators Alberto Chiarugi (Headache Centre, Careggi University Hospital, Department of Health Sciences, University of Florence, Florence, Italy)
Rights and permissions
About this article
Cite this article
Giorgio, A., Lupi, C., Zhang, J. et al. Changes in grey matter volume and functional connectivity in cluster headache versus migraine. Brain Imaging and Behavior 14, 496–504 (2020). https://doi.org/10.1007/s11682-019-00046-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11682-019-00046-2