Abstract
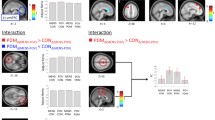
Neuroimaging studies have demonstrated the critical role of the insula in pain pathways and its close relation with the perceived intensity of nociceptive stimuli. We aimed to identify the structural and functional characteristics of the insula during periovulatory phase in women with primary dysmenorrhea (PDM), and further investigate its association with the intensity of perceived pain during menstruation. Optimized voxel-based morphometry and functional connectivity (FC) analyses were applied by using 3-dimensional T1-weighted and resting functional magnetic resonance imaging (fMRI) in 36 patients at the peri-ovulation phase and 29 age-, education-, and gender-matched healthy controls (HC). A visual analogue scale (VAS) was used to examine the intensity of the abdominal pain at periovulation and menstruation. In our results, PDM patients had significant higher VAS-rating during menstruaion than periovulation. Compared with the HC, PDM patients had lower gray matter density in the left anterior insula (aINS). Taken the left aINS as a seed region, we further found hypoconnectivity between aINS and medial prefrontal cortex (mPFC), which showed negative relation with the VAS during menstruation. As the aINS is a key site of the salience network (SN) and the mPFC is a critical region in the default mode network (DMN), it’s implicated a trait-related central-alteration that communications between pain attention and perception networks were disrupted without the ongoing menstrual pain. Moreover, result of correlation analysis, at least in part, suggested a possible role of altered FC (pain-free period) in predicting pain perception (menstruation).



Similar content being viewed by others
References
Amodio, D. M., & Frith, C. D. (2006). Meeting of minds: the medial frontal cortex and social cognition. Nature Reviews. Neuroscience, 7(4), 268–277.
Andersson, J. L., Jenkinson, M. & Smith, S. (2007a) Non-linear optimisation. FMRIB Analysis Group Technical Reports. TR07JA1. Oxford: University of Oxford.
Andersson, J. L. R., Jenkinson, M., & Smith, S. (2007b). Non-linear registration, aka spatial normalisation FMRIB technical report TR07JA2. FMRIB Analysis Group of the University of Oxford.
Andrews-Hanna, J. R., Smallwood, J., & Spreng, R. N. (2014). The default network and self-generated thought: component processes, dynamic control, and clinical relevance. Annals of the New York Academy of Sciences, 1316(1), 29–52.
Augustine, J. R. (1985). The insular lobe in primates including humans. Neurological Research, 7(1), 2–10.
Baliki, M. N., & Apkarian, A. V. (2015). Nociception, pain, negative moods, and behavior selection. Neuron, 87(3), 474–491.
Baliki, M. N., Geha, P. Y., & Apkarian, A. V. (2009). Parsing pain perception between nociceptive representation and magnitude estimation. Journal of Neurophysiology, 101(2), 875–887.
Baliki, M. N., Petre, B., Torbey, S., Herrmann, K. M., Huang, L., Schnitzer, T. J., et al. (2012). Corticostriatal functional connectivity predicts transition to chronic back pain. Nature Neuroscience, 15(8), 1117–1119.
Baliki, M. N., Mansour, A. R., Baria, A. T., & Apkarian, A. V. (2014). Functional reorganization of the default mode network across chronic pain conditions. PloS One, 9(9), e106133.
Biswal, B. B., Mennes, M., Zuo, X. N., Gohel, S., Kelly, C., Smith, S. M., et al. (2010). Toward discovery science of human brain function. Proceedings of the National Academy of Sciences of the United States of America, 107(10), 4734–4739.
Borsook, D., Edwards, R., Elman, I., Becerra, L., & Levine, J. (2013). Pain and analgesia: the value of salience circuits. Progress in Neurobiology, 10493–10105.
Borsook, D., Veggeberg, R., Erpelding, N., Borra, R., Linnman, C., Burstein, R., et al. (2015). The insula: a "hub of activity" in migraine. The Neuroscientist. doi:10.1177/1073858415601369.
Buhle, J. T., Silvers, J. A., Wager, T. D., Lopez, R., Onyemekwu, C., Kober, H., et al. (2014). Cognitive reappraisal of emotion: a meta-analysis of human neuroimaging studies. Cerebral Cortex, 24(11), 2981–2990.
Cauda, F., Palermo, S., Costa, T., Torta, R., Duca, S., Vercelli, U., et al. (2014). Gray matter alterations in chronic pain: a network-oriented meta-analytic approach. Neuroimage Clin, 4, 676–686.
Cerliani, L., Thomas, R. M., Jbabdi, S., Siero, J. C., Nanetti, L., Crippa, A., et al. (2012). Probabilistic tractography recovers a rostrocaudal trajectory of connectivity variability in the human insular cortex. Human Brain Mapping, 33(9), 2005–2034.
Cloutman, L. L., Binney, R. J., Drakesmith, M., Parker, G. J., & Lambon Ralph, M. A. (2012). The variation of function across the human insula mirrors its patterns of structural connectivity: evidence from in vivo probabilistic tractography. NeuroImage, 59(4), 3514–3521.
Crombez, G., Van Damme, S., & Eccleston, C. (2005). Hypervigilance to pain: an experimental and clinical analysis. Pain, 116(1–2), 4–7.
Dawood, M. Y. (2006). Primary dysmenorrhea: advances in pathogenesis and management. Obstetrics and Gynecology, 108(2), 428–441.
Douaud, G., Smith, S., Jenkinson, M., Behrens, T., Johansen-Berg, H., Vickers, J., et al. (2007). Anatomically related grey and white matter abnormalities in adolescent-onset schizophrenia. Brain, 130(Pt 9), 2375–2386.
Duerden, E. G., & Albanese, M. C. (2013). Localization of pain-related brain activation: a meta-analysis of neuroimaging data. Human Brain Mapping, 34(1), 109–149.
Dun, W., Yang, J., Yang, L., Ma, S., Guo, C., Zhang, X., et al. (2016). Abnormal white matter integrity during pain-free periovulation is associated with pain intensity in primary dysmenorrhea. Brain Imaging and Behavior. doi:10.1007/s11682-016-9582-x.
Elsenbruch, S., Schmid, J., Kullmann, J. S., Kattoor, J., Theysohn, N., Forsting, M., et al. (2014). Visceral sensitivity correlates with decreased regional gray matter volume in healthy volunteers: a voxel-based morphometry study. Pain, 155(2), 244–249.
Fritz, H.-C., McAuley, J. H., Wittfeld, K., Hegenscheid, K., Schmidt, C. O., Langner, S., et al. (2015). Chronic back pain is associated with decreased prefrontal and anterior insular gray matter: results from a population-based cohort study. The Journal of Pain, 17(1), 111–118.
Geha, P., & Waxman, S. G. (2016). Pain perception: multiple matrices or one? JAMA Neurology, 73(6), 628–630.
Geha, P. Y., Baliki, M. N., Harden, R. N., Bauer, W. R., Parrish, T. B., & Apkarian, A. V. (2008). The brain in chronic CRPS pain: abnormal gray-white matter interactions in emotional and autonomic regions. Neuron, 60(4), 570–581.
Good, C. D., Johnsrude, I. S., Ashburner, J., Henson, R. N., Friston, K. J., & Frackowiak, R. S. (2001). A voxel-based morphometric study of ageing in 465 normal adult human brains. NeuroImage, 14(1 Pt 1), 21–36.
Guenot, M., Isnard, J., & Sindou, M. (2004). Surgical anatomy of the insula. Advances and Technical Standards in Neurosurgery, 2, 9265–9288.
Hashmi, J. A., Baliki, M. N., Huang, L., Baria, A. T., Torbey, S., Hermann, K. M., et al. (2013). Shape shifting pain: chronification of back pain shifts brain representation from nociceptive to emotional circuits. Brain, 136(Pt 9), 2751–2768.
Isnard, J., Magnin, M., Jung, J., Mauguiere, F., & Garcia-Larrea, L. (2011). Does the insula tell our brain that we are in pain? Pain, 152(4), 946–951.
Jenkinson, M., & Smith, S. (2001). A global optimisation method for robust affine registration of brain images. Medical Image Analysis, 5(2), 143–156.
Jenkinson, M., Bannister, P., Brady, M., & Smith, S. (2002). Improved optimization for the robust and accurate linear registration and motion correction of brain images. NeuroImage, 17(2), 825–841.
Kuchinad, A., Schweinhardt, P., Seminowicz, D. A., Wood, P. B., Chizh, B. A., & Bushnell, M. C. (2007). Accelerated brain gray matter loss in fibromyalgia patients: premature aging of the brain? The Journal of Neuroscience, 27(15), 4004–4007.
Kucyi, A., & Davis, K. D. (2015). The dynamic pain connectome. Trends in Neurosciences, 38(2), 86–95.
Kucyi, A., Salomons, T. V., & Davis, K. D. (2013). Mind wandering away from pain dynamically engages antinociceptive and default mode brain networks. Proceedings of the National Academy of Sciences of the United States of America, 110(46), 18692–18697.
Kurth, F., Zilles, K., Fox, P. T., Laird, A. R., & Eickhoff, S. B. (2010). A link between the systems: functional differentiation and integration within the human insula revealed by meta-analysis. Brain Structure & Function, 214(5–6), 519–534.
Lee, L. C., Tu, C. H., Chen, L. F., Shen, H. D., Chao, H. T., Lin, M. W., et al. (2014). Association of brain-derived neurotrophic factor gene Val66Met polymorphism with primary dysmenorrhea. PloS One, 9(11), e112766.
Lisofsky, N., Martensson, J., Eckert, A., Lindenberger, U., Gallinat, J., & Kuhn, S. (2015). Hippocampal volume and functional connectivity changes during the female menstrual cycle. NeuroImage, 118, 154–162.
Liu, P., Yang, J., Wang, G., Liu, Y., Liu, X., Jin, L., et al. (2015). Altered regional cortical thickness and subcortical volume in women with primary dysmenorrhoea. European Journal of Pain, 20(4), 512–520.
Maleki, N., Becerra, L., Brawn, J., Bigal, M., Burstein, R., & Borsook, D. (2012). Concurrent functional and structural cortical alterations in migraine. Cephalalgia, 32(8), 607–620.
Mayer, E. A., Gupta, A., Kilpatrick, L. A. & Hong, J. Y. (2015). Imaging brain mechanisms in chronic visceral pain. Pain, 156 Suppl 1S50–63.
Menon, V., & Uddin, L. Q. (2010). Saliency, switching, attention and control: a network model of insula function. Brain Structure & Function, 214(5–6), 655–667.
Mesulam, M. M., & Mufson, E. J. (1982). Insula of the old world monkey. III: efferent cortical output and comments on function. The Journal of Comparative Neurology, 212(1), 38–52.
Moayedi, M., Weissman-Fogel, I., Crawley, A. P., Goldberg, M. B., Freeman, B. V., Tenenbaum, H. C., et al. (2011). Contribution of chronic pain and neuroticism to abnormal forebrain gray matter in patients with temporomandibular disorder. NeuroImage, 55(1), 277–286.
Morrow, C., & Naumburg, E. H. (2009). Dysmenorrhea. Primary Care, 36(1), 19–32.
Mufson, E. J., & Mesulam, M. M. (1982). Insula of the old world monkey. II: afferent cortical input and comments on the claustrum. The Journal of Comparative Neurology, 212(1), 23–37.
Napadow, V., LaCount, L., Park, K., As-Sanie, S., Clauw, D. J., & Harris, R. E. (2010). Intrinsic brain connectivity in fibromyalgia is associated with chronic pain intensity. Arthritis and Rheumatism, 62(8), 2545–2555.
Nieuwenhuys, R. (2011). The insular cortex: a review. Progress in Brain Research, 195, 123–163.
Ploner, M., Lee, M. C., Wiech, K., Bingel, U., & Tracey, I. (2010). Prestimulus functional connectivity determines pain perception in humans. Proceedings of the National Academy of Sciences of the United States of America, 107(1), 355–360.
Schweinhardt, P., Glynn, C., Brooks, J., McQuay, H., Jack, T., Chessell, I., et al. (2006). An fMRI study of cerebral processing of brush-evoked allodynia in neuropathic pain patients. NeuroImage, 32(1), 256–265.
Seeley, W. W., Menon, V., Schatzberg, A. F., Keller, J., Glover, G. H., Kenna, H., et al. (2007). Dissociable intrinsic connectivity networks for salience processing and executive control. The Journal of Neuroscience, 27(9), 2349–2356.
Smith, S. M. (2002). Fast robust automated brain extraction. Human Brain Mapping, 17(3), 143–155.
Stankewitz, A., & May, A. (2011). Increased limbic and brainstem activity during migraine attacks following olfactory stimulation. Neurology, 77(5), 476–482.
Starr, C. J., Sawaki, L., Wittenberg, G. F., Burdette, J. H., Oshiro, Y., Quevedo, A. S., et al. (2009). Roles of the insular cortex in the modulation of pain: insights from brain lesions. The Journal of Neuroscience, 29(9), 2684–2694.
Tracey, I., & Mantyh, P. W. (2007). The cerebral signature for pain perception and its modulation. Neuron, 55(3), 377–391.
Tu, C. H., Niddam, D. M., Chao, H. T., Liu, R. S., Hwang, R. J., Yeh, T. C., et al. (2009). Abnormal cerebral metabolism during menstrual pain in primary dysmenorrhea. NeuroImage, 47(1), 28–35.
Tu, C. H., Niddam, D. M., Chao, H. T., Chen, L. F., Chen, Y. S., Wu, Y. T., et al. (2010). Brain morphological changes associated with cyclic menstrual pain. Pain, 150(3), 462–468.
Tu, C. H., Niddam, D. M., Yeh, T. C., Lirng, J. F., Cheng, C. M., Chou, C. C., et al. (2013). Menstrual pain is associated with rapid structural alterations in the brain. Pain, 154(9), 1718–1724.
Valfre, W., Rainero, I., Bergui, M., & Pinessi, L. (2008). Voxel-based morphometry reveals gray matter abnormalities in migraine. Headache, 48(1), 109–117.
Vincent, K., Warnaby, C., Stagg, C. J., Moore, J., Kennedy, S., & Tracey, I. (2011). Dysmenorrhoea is associated with central changes in otherwise healthy women. Pain, 152(9), 1966–1975.
Wager, T. D., Atlas, L. Y., Lindquist, M. A., Roy, M., Woo, C. W., & Kross, E. (2013). An fMRI-based neurologic signature of physical pain. The New England Journal of Medicine, 368(15), 1388–1397.
Wei, S. Y., Chao, H. T., Tu, C. H., Li, W. C., Low, I., Chuang, C. Y., et al. (2016). Changes in functional connectivity of pain modulatory systems in women with primary dysmenorrhea. Pain, 157(1), 92–102.
Wiech, K., Lin, C. S., Brodersen, K. H., Bingel, U., Ploner, M., & Tracey, I. (2010). Anterior insula integrates information about salience into perceptual decisions about pain. The Journal of Neuroscience, 30(48), 16324–16331.
Willis, W. D., Al-Chaer, E. D., Quast, M. J., & Westlund, K. N. (1999). A visceral pain pathway in the dorsal column of the spinal cord. Proceedings of the National Academy of Sciences of the United States of America, 96(14), 7675–7679.
Woo, C. W., Roy, M., Buhle, J. T., & Wager, T. D. (2015). Distinct brain systems mediate the effects of nociceptive input and self-regulation on pain. PLoS Biology, 13(1), e1002036.
Wu, T. H., Tu, C. H., Chao, H. T., Li, W. C., Low, I., Chuang, C. Y., et al. (2016). Dynamic changes of functional pain connectome in women with primary dysmenorrhea. Scientific Reports, 6, 624543.
Xue, T., Yuan, K., Zhao, L., Yu, D., Zhao, L., Dong, T., et al. (2012). Intrinsic brain network abnormalities in migraines without aura revealed in resting-state fMRI. PloS One, 7(12), e52927.
Yuan, K., Zhao, L., Cheng, P., Yu, D., Zhao, L., Dong, T., et al. (2013). Altered structure and resting-state functional connectivity of the basal ganglia in migraine patients without aura. The Journal of Pain, 14(8), 836–844.
Zahradnik, H. P., Hanjalic-Beck, A., & Groth, K. (2010). Nonsteroidal anti-inflammatory drugs and hormonal contraceptives for pain relief from dysmenorrhea: a review. Contraception, 81(3), 185–196.
Zhang, Y., Brady, M., & Smith, S. (2001). Segmentation of brain MR images through a hidden Markov random field model and the expectation-maximization algorithm. Medical Imaging, IEEE Transactions on, 20(1), 45–57.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
Wanghuan Dun, Jing Yang, Ling Yang, Dun Ding, Shaohui Ma, Feng-Li Liang, Karen M. von Deneen, and Xiao-ling Xu critically reviewed the content and approved the final version for publication. They declared no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Ethical statements
Informed consent was obtained from all individual participants included in the study.
Funding sources
This work was supported by the National Natural Science Foundation of China (Grant No. 81,371,530, 81,571,640 and 81,471,737) and the Research Funds of the First Affiliated Hospital of Xi’an Jiaotong University, College of Medicine (Grant No. 2014YK1).
Additional information
Wanghuan Dun and Jing Yang contributed equally to this work and should be considered co-first authors.
Rights and permissions
About this article
Cite this article
Dun, Wh., Yang, J., Yang, L. et al. Abnormal structure and functional connectivity of the anterior insula at pain-free periovulation is associated with perceived pain during menstruation. Brain Imaging and Behavior 11, 1787–1795 (2017). https://doi.org/10.1007/s11682-016-9646-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11682-016-9646-y