Abstract
Diabetes is associated with macrovascular and microvascular complications and is a major risk factor for neurological and psychiatric disorders, such as dementia and depression. Type 1 diabetes (T1DM) and type 2 diabetes (T2DM) have distinct etiologies and pathophysiological effects while sharing a common endpoint of persistent hyperglycemia. Neuroimaging studies in T1DM have revealed reductions in numerous regions, including the parahippocampal and occipital regions, while in T2DM there have been numerous reports of hippocampal atrophy. This meta-analysis aimed to identify consistent regional abnormalities in cerebral structures in T1DM and T2DM respectively, and also to examine the impact of potential confounds, including age, depression and vascular risk factors. Neuroimaging studies of both voxel-based morphometry (VBM) data and volumetric data were included. Ten T1DM studies (n = 613 patients) and 23 T2DM studies (n = 1364 patients) fulfilled inclusion criteria. The T1DM meta-analysis revealed reduced bilateral thalamus grey matter density in adults. The T2DM meta-analysis revealed reduced global brain volume and regional atrophy in the hippocampi, basal ganglia, and orbitofrontal and occipital lobes. Moreover, hippocampal atrophy in T2DM was not modified by hypertension, although there were more marked reductions in younger patients relative to healthy controls. In conclusion, T1DM and T2DM demonstrated distinct cerebral effects with generalised and specific target areas of grey matter reduction. Thalamic atrophy in T1DM may be a substrate of associated cognitive deficits. In T2DM, global cerebral atrophy may reflect atherosclerotic factors, while hippocampal atrophy was an independent effect providing a potential common neuropathological etiology for the comorbidity of T2DM with dementia and depression.




Similar content being viewed by others
References
Ali, S., Stone, M. A., Peters, J. L., Davies, M. J., & Khunti, K. (2006). The prevalence of co-morbid depression in adults with type 2 diabetes: a systematic review and meta-analysis. Diabetic Medicine, 23, 1165–1173.
Alosco, M., Brickman, A. M., Spitznagel, M. B., Griffith, E. Y., Narkhede, A., Raz, N., et al. (2013). The adverse impact of type 2 diabetes on brain volume in heart failure. Journal of Clinical and Experimental Neuropsychology, 35, 309–318.
Anan, F., Masaki, T., Shimomura, T., Fujiki, M., Umeno, Y., Eshima, N., et al. (2010). Abdominal visceral fat accumulation is associated with hippocampus volume in non-dementia patients with type 2 diabetes mellitus. NeuroImage, 49, 57–62.
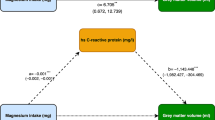
Anan, F., Masaki, T., Shimomura, T., Fujiki, M., Umeno, Y., Eshima, N., et al. (2011). High-sensitivity C-reactive protein is associated with hippocampus volume in nondementia patients with type 2 diabetes mellitus. Metabolism, Clinical and Experimental, 60, 460–66.
Ashburner, J., & Friston, K. J. (2000). Voxel-based morphometry–the methods. NeuroImage, 11(6 Pt 1), 805–21.
Aye, T., Reiss, A. L., Kesler, S., Hoang, S., Drobny, J., Park, Y., et al. (2011). The feasibility of detecting neuropsychological and neuroanatomic effects of type 1 diabetes in young children. Diabetes Care, 34, 1458–1462.
Balakrishnan, S., Mathew, J., & Paulose, C. S. (2010). Cholinergic and glutamergic receptor functional regulation in long-term, low dose somatotropin and insulin treatment to ageing rats: rejuvenation of brain function. Mollecular and Cellular Endocrinology, 314, 23–30.
Ballmaier, M., Toga, A. W., Blanton, R. E., Sowell, E. R., Lavretsky, H., et al. (2004). Anterior cingulate, gyrus rectus, and orbitofrontal abnormalities in elderly depressed patients: an MRI-based parcellation of the prefrontal cortex. American Journal of Psychiatry, 161, 99–108.
Biessels, G. J., Staekenborg, S., Brunner, E., Brayne, C., & Scheltens, P. (2006). Risk of dementia in diabetes mellitus: a systematic review. Lancet Neurology, 5, 64–74.
Bondy, C., Bach, M. A., & Lee, W.-H. (1992). Mapping of brain insulin and insulin-like growth factor receptor gene expression by in situ hybridization. Neuroprotocols, 1, 240–249.
Brands, A. M., Biessels, G. J., de Haan, E. H., Kappelle, L. J., & Kessels, R. P. (2005). The effects of type 1 diabetes on cognitive performance: a meta-analysis. Diabetes Care, 28, 726–735.
Brownlee, M. (1995). Advanced protein glycosylation in diabetes and aging. Annual Review of Medicine, 46, 223–234.
Bruehl, H., Wolf, O. T., & Convit, A. (2009). A blunted cortisol awakening response and hippocampal atrophy in type 2 diabetes mellitus. Psychoneuroendocrinology, 34, 815–821.
Brundel, M., van den Heuvel, M., de Bresser, J., Kappelle, L. J., & Biessels, G. J. (2010). Cerebral cortical thickness in patients with type 2 diabetes. Journal of the Neurological Sciences, 299, 126–130.
Chen, Z., Li, L., Sun, J., & Ma, L. (2012). Mapping the brain in type II diabetes: voxel-based morphometry using DARTEL. European Journal of Radiology, 81, 1870–1876.
Cole, J., Costafreda, S. G., McGuffin, P., & Fu, C. H. Y. (2011). Hippocampal atrophy in first episode depression: a meta-analysis of magnetic resonance imaging studies. Journal of Affective Disorders, 134, 483–487.
Colman, I., Jones, P. B., Kuh, D., Weeks, M., Naicker, K., Richards, M., & Croudace, T. J. (2014). Early development, stress and depression across the life course: pathways to depression in a national British birth cohort. Psychological Medicine, 44(13), 2845–54.
Costafreda, S. G. (2012). Parametric coordinate-based meta-analysis: valid effect size meta-analysis of studies with differing statistical thresholds. Journal of Neuroscience Methods, 210, 291–300.
de Bresser, J., Tiehuis, A. M., van den Berg, E., Reijmer, Y. D., Jongen, C., Kappelle, L. J., et al. (2010). Progression of cerebral atrophy and white matter hypersensitivites in patients with type 2 diabetes. Diabetes Care, 33, 1309–1314.
de Jong, L. W., van der Hiele, K., Veer, I. M., Houwing, J. J., Westendorp, R. G., Bollen, E. L., et al. (2008). Strongly reduced volumes of putamen and thalamus in Alzheimer’s disease: an MRI study. Brain, 131, 3277–3285.
den Heijer, T., Vermeer, S. E., van Dijk, E. J., Prins, N. D., Koudstaal, P. J., Hofman, A., et al. (2003). Type 2 diabetes and atrophy of medial temporal lobe structures on brain MRI. Diabetologia, 46, 1604–1610.
Deng, Y., Li, B., Liu, Y., Iqbal, K., Grundke-Iqbal, I., & Gong, C. X. (2009). Dysregulation of insulin signalling, glucose transporters, O-GlcNAcylation, and phosphorylation of tau and neurofilaments in the brain: implication for Alzheimer’s disease. American Journal of Pathology, 175, 2089–2098.
DerSimonian, R., & Laird, N. (1986). Meta-analysis in clinical trials. Controlled Clinical Trials, 7(3), 177–188.
Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications Study Research Group, Jacobson, A. M., Musen, G., Ryan, C. M., Silvers, N., Cleary, P., et al. (2007). Long-term effect of diabetes and its treatment on cognitive function. New England Journal of Medicine, 356, 1842–1852.
Dowlati, Y., Herrmann, N., Swardfager, W., Liu, H., Sham, L., Reim, E. K., et al. (2010). A meta-analysis of cytokines in major depression. Biological Psychiatry, 67, 446–457.
Egan, M. F., Kojima, M., Callicott, J. H., Goldberg, T. E., & Kolachana, B. S. (2003). The BDNF val66met polymorphism affects activity-dependent secretion of BDNF and human memory and hippocampal function. Cell, 2003(112), 257–69.
Elder, G. A., De Gasperi, R., & Gama Sosa, M. A. (2006). Research update: neurogenesis in adult brain and neuropsychiatric disorders. Mount Sinai Journal of Medicine, 73, 931–40.
Espeland, M. A., Bryan, R. N., Goveas, J. S., Robinson, J. G., Siddiqui, M. S., Liu, S., et al. (2013). Influence of type 2 diabetes on brain volumes and changes in brain volumes. Diabetes Care, 36, 90–97.
Frodl, T., Carballedo, A., Hughes, M. M., Saleh, K., Fagan, A., Skokauskas, N., et al. (2012). Reduced expression of glucocorticoid-inducible genes GILZ and SGK-1: high IL-6 levels are associated with reduced hippocampal volumes in major depressive disorder. Translational Psychiatry, 2, e88.
Fu, C. H. Y., Steiner, H., & Costafreda, S. G. (2013). Predictive neural biomarkers of clinical response in depression: a meta-analysis of functional and structural neuroimaging studies of pharmacological and psychological therapies. Neurobiology of Disease, 52, 75–83.
Fujinami, A., Ohta, K., Obayashi, H., Fukui, M., Hasegawa, G., & Nakamura, N. (2008). Serum brain-derived neurotrophic factor in patients with type 2 diabetes mellitus: Relationship to glucose metabolism and biomarkers of insulin resistance. Clinical Biochemistry, 41(10–11), 812–7.
Gaudieri, P. A., Chen, R., Greer, T. F., & Holmes, C. S. (2008). Cognitive function in children with type 1 diabetes: a meta-analysis. Diabetes Care, 31, 1892–1897.
Gold, S. M., Dziobek, I., Sweat, V., Tirsi, A., Rogers, K., Bruehl, H., et al. (2007). Hippocampal damage and memory impairments as possible early brain complications of type 2 diabetes. Diabetologia, 50, 711–719.
Hayashi, K., Kurioka, S., Yamaguchi, T., Morita, M., Kanazawa, I., Takase, H., et al. (2011). Association of cognitive dysfunction with hippocampal atrophy in elderly Japanese people with type 2 diabetes. Diabetes Research and Clinical Practice, 94, 180–185.
Hempel, R., Onopa, R., & Convit, A. (2012). Type 2 diabetes affects hippocampus volume differentially in men and women. Diabetes/Metabolism Research and Reviews, 28, 76–83.
Herrero, M. T., Barcia, C., & Navarro, J. M. (2002). Functional anatomy of thalamus and basal ganglia. Child’s Nervous System, 18, 386–404.
Hershey, T., Perantie, D. C., Wu, J., Weaver, P. M., Black, K. J., et al. (2010). Hippocampal volumes in youth with type 1 diabetes. Diabetes, 59, 236–241.
Higgins, J., Thompson, S., Deeks, J., & Altman, D. (2003). Measuring inconsistency in meta-analysis. BMJ, 327, 557–560.
Jongen, C., van der Grond, J., Kappelle, L. J., Biessels, G. J., Viergever, M. A., & Pluim, J. P. (2007). Automated measurement of brain and white matter lesion volume in type 2 diabetes mellitus. Diabetologia, 50, 1509–1516.
Kamiyama, K., Wada, A., Sugihara, M., Kurioka, S., Hayashi, K., Hayashi, T., et al. (2010). Potential hippocampal region atrophy in diabetes mellitus type 2: a voxel-based morphometry VSRAD study. Japanese Journal of Radiology, 28, 266–272.
Karege, F., Perret, G., Bondolfi, G., Schwald, M., & Bertschy, G. (2002). Decreased serum brain-derived neurotrophic factor levels in major depressed patients. Psychiatry Research, 109, 143–8.
Katon, W., Lyles, C. R., Parker, M. M., Karter, A. J., Huang, E. S., & Whitmer, R. A. (2012). Association of depression with increased risk of dementia in patients with type 2 diabetes. Archives of General Psychiatry, 69, 410–417.
Klunk, W. E., Price, J. C., Mathis, C. A., Tsopelas, N. D., Lopresti, B. J., Ziolko, S. K., et al. (2007). Amyloid deposition begins in the striatum of presenilin-1 mutation carriers from two unrelated pedigrees. The Journal of Neuroscience, 27, 6174–84.
Korczak, D. J., Pereira, S., Koulajian, K., Matejcek, A., & Giacca, A. (2011). Type 1 diabetes mellitus and major depressive disorder: evidence for a biological link. Diabetologia, 54(10), 2483–93.
Korf, E. S., White, L. R., Scheltens, P., & Launer, L. J. (2006). Brain aging in very old men with type 2 diabetes. Diabetes Care, 29, 2268–2274.
Kumar, A., Haroon, E., Darwin, C., Pham, D., Ajilore, O., Rodriguez, G., & Mintz, J. (2008). Gray matter prefrontal changes in type 2 diabetes detected using MRI. Journal of Magnetic Resonance Imaging, 27, 14–19.
Laake, J.P., Stahl, D., Amiel, S.A., Petrak, F., Sherwood, R.A., Pickup, J.C., & Ismail, K. (2014). The association between depressive symptoms and systemic inflammation in people with type 2 diabetes: findings from the south london diabetes study. Diabetes Care, May 19. pii: DC_132522. (Epub ahead of print).
Last, D., Alsop, D. C., Abduljalil, A. M., Marquis, R. P., de Bazelaire, C., Hu, K., et al. (2007). Global and regional effects of type 2 diabetes on brain tissue volumes and cerebral vasoreactivity. Diabetes Care, 30(5), 1193–9.
Liu, F., Shi, J., Tanimukai, H., Gu, J., Gu, J., Grundke-Iqbal, I., et al. (2009). Reduced O-GlcNAcylation links lower brain glucose metabolism and tau phosphorylation in Alzheimer’s disease. Brain, 132, 1820–1832.
Lobnig, B. M., Krömeke, O., Optenhostert-Porst, C., & Wolf, O. T. (2005). Hippocampal volume and cognitive performance in long-standing type 1 diabetic patients without macrovascular complications. Diabetic Medicine, 23, 32–39.
Lu, F. P., Lin, K. P., & Kuo, H. K. (2009). Diabetes and the risk of multi-system aging phenotypes: a systematic review and meta-analysis. PLoS One, 4(1), e4144.
Mangia, S., Tesfaye, N., De Martino, F., Kumar, A. F., Kollasch, P., Moheet, A. A., et al. (2012). Hypoglycemia-induced increases in thalamic cerebral blood flow are blunted in subjects with type 1 diabetes and hypoglycemia unawareness. Journal of Cerebral Blood Flow & Metabolism, 32, 2084–2090.
Manor, B., Newton, E., Abduljalil, A., & Novak, V. (2012). The relationship between brain volume and walking outcomes in older adults with and without diabetic peripheral neuropathy. Diabetes Care, 35, 1907–1912.
Mezuk, B., Eaton, W. W., Albrecht, S., & Golden, S. H. (2008). Depression and type 2 diabetes over the lifespan: a meta-analysis. Diabetes Care, 31, 2383–2390.
Musen, G., Lyoo, I. K., Sparks, C. R., Weinger, K., Hwang, J., Ryan, C. M., et al. (2006). Effects of type 1 diabetes on gray matter density as measured by voxel-based morphometry. Diabetes, 55, 326–333.
Musen, G., Jacobson, A. M., Bolo, N. R., Simonson, D. C., Shenton, M. E., McCartney, R. L., et al. (2012). Resting-state brain functional connectivity is altered in type 2 diabetes. Diabetes, 62, 2375–2379.
Naguib, J. M., Kulinskaya, E., Lomax, C. L., & Garralda, M. E. (2009). Neuro-cognitive performance in children with type 1 diabetes: a meta analysis. Journal of Pediatric Psychology, 34, 271–282.
Necula, M., & Kuret, J. (2004). Pseudophosphorylation and glycation of tau protein enhance but do not trigger fibrillization in vitro. The Journal of Biological Chemistry, 279, 49694–49703.
Northam, E. A., Rankins, D., Lin, A., Wellard, R. M., Pell, G. S., Finch, S. J., et al. (2009). Central nervous system function in youth with type 1 diabetes 12 years after disease onset. Diabetes Care, 32, 445–450.
Novak, V., Zhao, P., Manor, B., Sejdic, E., Alsop, D., Abduljalil, A., et al. (2011). Adhesion molecules, altered vasoreactivity, and brain atrophy in type 2 diabetes. Diabetes Care, 34, 2438–2411.
Ott, A., Stolk, R. P., van Harskamp, F., Pols, H. A., Hofman, A., Breteler, M. M. (1999). Diabetes mellitus and the risk of dementia: The Rotterdam Study. Neurology, 53(9), 1937–42.
Oxford centre for functional magnetic resonance imaging of the brain. (2003). Oxford thalamic connectivity atlas: www2.fmrib.ox.ac.uk/connect.
Perantie, D. C., Wu, J., Koller, J. M., Lim, A., Warren, S. L., Black, K. J., et al. (2007). Regional brain volume differences associated with hyperglycemia and severe hypoglycemia in youth with type 1 diabetes. Diabetes Care, 30, 2331–2337.
Perantie, D. C., Koller, J. M., Weaver, P. M., Lugar, H. M., Black, K. J., White, N. H., et al. (2011). Prospectively determined impact of type 1 diabetes on brain volume during development. Diabetes, 60, 3006–3014.
Pickup, J. C., & Crook, M. A. (1998). Is type II diabetes a disease of the innate immune system? Diabetologia, 41, 1241–1248.
Sarac, K., Akinci, A., Alkan, A., Aslan, M., Baysal, T., & Ozcan, C. (2007). Brain metabolite changes on proton magnetic resonance spectroscopy in children with poorly controlled type 1 diabetes. Neuroradiology, 47, 562–565.
Scott, K. M., Von Korff, M., Angermeyer, M. C., Benjet, C., Bruffaerts, R., de Girolamo, G., et al. (2011). Association of childhood adversities and early-onset mental disorders with adult-onset chronic physical conditions. Archives of General Psychiatry, 68(8), 838–44.
Sima, A. A., Zhang, W., Kreipke, C. W., Rafols, J. A., & Hoffman, W. H. (2009). Inflammation in diabetic encephalopathy is prevented by C-peptide. The Review of Diabetic Studies, 6, 37–42.
Stamler, J., Vaccaro, O., Neaton, J. D., & Wentworth, D. (1993). Diabetes, other risk factors, and 12-yr cardiovascular mortality for men screened in the Multiple Risk Factor Intervention Trial. Diabetes Care, 16(2), 434–44.
Stetler, C., & Miller, G. E. (2011). Depression and hypothalamic-pituitary-adrenal activation: a quantitative summary of four decades of research. Psychosomatic Medicine, 73, 114–26.
Teicher, M. H., Andersen, S. L., Polcari, A., Anderson, C. M., Navalta, C. P., & Kim, D. M. (2003). The neurobiological consequences of early stress and childhood maltreatment. Neuroscience and Biobehavioral Reviews, 27(1–2), 33–44.
Tesfomariam, B., Brown, M. L., & Cohen, R. A. (1991). Elevated glucose impairs endothelium-dependent relaxation by activating protein kinase C. The Journal of Clinical Investigation, 87, 1643–1648.
Tiehuis, A. M., van der Graaf, Y., Visseren, F. L., Vincken, K. L., Biessels, G. J., Appelman, A. P., et al. (2008). Diabetes increases atrophy and vascular lesions on brain MRI in patients with symptomatic arterial disease. Stroke, 39, 1600–1603.
Twist, K., Stahl, D., Amiel, S. A., Thomas, S., Winkley, K., & Ismail, K. (2013). Comparison of depressive symptoms in type 2 diabetes using a two-stage survey design. Psychosomatic Medicine, 75(8), 791–7.
Tzourio-Mazoyer, N., Landeau, B., Papathanassiou, D., Crivello, F., Etard, O., Delcroix, N., Mazoyer, B., & Joliot, M. (2002). Automated anatomical labeling of activations in SPM using a macroscopic anatomical parcellation of the MNI MRI single-subject brain. NeuroImage, 15(1), 273–89.
van Elderen, S. G., Brandts, A., van der Grond, J., Westenberg, J. J., Kroft, L. J., van Buchem, M. A., et al. (2011). Cerebral perfusion and aortic stiffness are independent predictors of white matter brain atrophy in type 1 diabetic patients assessed with magnetic resonance imaging. Diabetes Care, 34, 459–463.
Videbech, P., & Ravnkilde, B. (2004). Hippocampal volume and depression: a meta-analysis of MRI studies. American Journal of Psychiatry, 161(11), 1957–1966.
Viechtbauer, W. (2010). Conducting meta-analyses in R with the metafor package. Journal of Statistical Software, 36(3), 1–48.
Watari, K., Elderkin-Thompson, V., Ajilore, O., Haroon, E., Darwin, C., et al. (2008). Neuroanatomical correlates of executive functioning in depressed adults with type 2 diabetes. Journal of Clinical and Experimental Neuropsychology, 30(4), 389–397.
Wessels, A. M., Simsek, S., Remijnse, P. L., Veltman, D. J., Biessels, G. J., Barkhof, F., et al. (2006). Voxel-based morphometry demonstrates reduced grey matter density on brain MRI in patients with diabetic retinopathy. Diabetologia, 49, 2474–2480.
Wessels, A. M., Rombouts, S. A., Remijnse, P. L., Boom, Y., Scheltens, P., Barkhof, F., et al. (2007). Cognitive performance in type 1 diabetes patients is associated with cerebral white matter volume. Diabetologia, 50, 1763–1769.
Williams, S. B., Goldfine, A. B., Timimi, F. K., Ting, H. H., Roddy, M. A., Simonson, D. C., & Creager, M. A. (1998). Acute hyperglycemia attenuates endothelium-dependent vasodilation in humans in vivo. Circulation, 97, 1695–1701.
Yau, P. L., Javier, D. C., Ryan, C. M., Tsui, W. H., Ardekani, B. A., Ten, S., et al. (2010). Preliminary evidence for brain complications in obese adolescents with type 2 diabetes mellitus. Diabetologia, 58, 2298–2306.
Conflict of interest
Calum D. Moulton, Sergi G. Costafreda, Paul Horton, Khalida Ismail, and Cynthia H. Y. Fu declare that they have no conflicts of interest.
Informed Consent
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, and the applicable revisions at the time of the investigation. Informed consent was obtained from all patients for being included in the study.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Fig. 1
Forest plot of hippocampal volume in T1DM studies. Non-significant reduction found in T1DM compared to controls (DOCX 54 kb)
Supplementary Fig. 2
Forest plots of total brain volume. Non-significant reduction found in T1DM compared to controls (left) and significant reduction in T2DM compared to controls (right) (DOCX 108 kb)
Supplementary Fig. 3
Forest plots of total grey matter volume. Non-significant reduction found in T1DM compared to controls (left) and significant reduction in T2DM compared to controls (right) (DOCX 114 kb)
Supplementary Fig. 4
Forest plot of total frontal volume. Non-significant reduction found in T2DM compared to controls (DOCX 65 kb)
Supplementary Table 1
Potential confounding variables for T1DM studies (DOCX 15 kb)
Supplementary Table 2
Potential confounding variables for T2DM studies (DOCX 17 kb)
Supplementary Table 3
Volumetric meta-analysis results in T1DM (DOCX 13 kb)
Rights and permissions
About this article
Cite this article
Moulton, C.D., Costafreda, S.G., Horton, P. et al. Meta-analyses of structural regional cerebral effects in type 1 and type 2 diabetes. Brain Imaging and Behavior 9, 651–662 (2015). https://doi.org/10.1007/s11682-014-9348-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11682-014-9348-2