Abstract
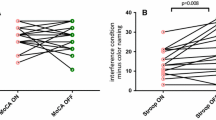
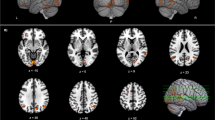
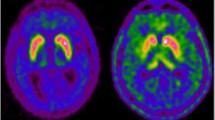
Subtle cognitive and behavioral changes are common in early Parkinson’s disease. The cause of these symptoms is probably multifactorial but may in part be related to extra-striatal dopamine levels. 6-[18 F]-Fluoro-L-dopa (FDOPA) positron emission tomography has been widely used to quantify dopamine metabolism in the brain; the most frequently measured kinetic parameter is the tissue uptake rate constant, Ki. However, estimates of dopamine turnover, which also account for the small rate of FDOPA loss from areas of specific trapping, may be more sensitive than Ki for early disease-related changes in dopamine biosynthesis. The purpose of the present study was to compare effective distribution volume ratio (eDVR), a metric for dopamine turnover, to cognitive and behavioral measures in Parkinson’s patients. We chose to focus the investigation on anterior cingulate cortex, which shows highest FDOPA uptake within frontal regions and has known roles in executive function. Fifteen non-demented early-stage PD patients were pretreated with carbidopa and tolcapone, a central catechol-O-methyl transferase (COMT) inhibitor, and then underwent extended imaging with FDOPA PET. Anterior cingulate eDVR was compared with composite scores for language, memory, and executive function measured by neuropsychological testing, and behavior change measured using two informant-based questionnaires, the Cambridge Behavioral Inventory and the Behavior Rating Inventory of Executive Function-Adult Version. Lower mean eDVR (thus higher dopamine turnover) in anterior cingulate cortex was related to lower (more impaired) behavior scores. We conclude that subtle changes in anterior cingulate dopamine metabolism may contribute to dysexecutive behaviors in Parkinson’s disease.


Similar content being viewed by others
References
Athey, R. J., & Walker, R. W. (2006). Demonstration of cognitive decline in Parkinson's disease using the Cambridge Cognitive Assessment (Revised) (CAMCOG-R). International Journal of Geriatric Psychiatry, 21(10), 977–982.
Benedict, R. H., Schretlen, D., Groninger, L., & Brandt, J. (1998). Hopkins Verbal Learning Test—Revised: Normative data and analysis of inter-form and test–retest reliability. Clinical Neuropsychologist, 12, 43–55.
Benton, A., Hamsher, D., & Sivan, A. (1983). Multilingual Aphasia Examination (3rd ed.). Iowa City: AJA Associates.
Bozeat, S., Gregory, C. A., Ralph, M. A., & Hodges, J. R. (2000). Which neuropsychiatric and behavioural features distinguish frontal and temporal variants of frontotemporal dementia from Alzheimer's disease? Journal of Neurology, Neurosurgery, and Psychiatry, 69(2), 178–186.
Braak, H., Braak, E., Yilmazer, D., Schultz, C., de Vos, R. A., & Jansen, E. N. (1995). Nigral and extranigral pathology in Parkinson's disease. Journal of Neural Transmission, Supplement, 46, 15–31.
Brown, W. D., Taylor, M. D., Roberts, A. D., Oakes, T. R., Schueller, M. J., Holden, J. E., et al. (1999). FluoroDOPA PET shows the nondopaminergic as well as dopaminergic destinations of levodopa. Neurology, 53(6), 1212–1218.
Bruck, A., Aalto, S., Nurmi, E., Bergman, J., & Rinne, J. O. (2005). Cortical 6-[18F]fluoro-L-dopa uptake and frontal cognitive functions in early Parkinson's disease. Neurobiology of Aging, 26(6), 891–898.
Desikan, R. S., Segonne, F., Fischl, B., Quinn, B. T., Dickerson, B. C., Blacker, D., et al. (2006). An automated labeling system for subdividing the human cerebral cortex on MRI scans into gyral based regions of interest. NeuroImage, 31(3), 968–980.
Doudet, D. J., Chan, G. L., Holden, J. E., Morrison, K. S., Wyatt, R. J., & Ruth, T. J. (1997). Effects of catechol-O-methyltransferase inhibition on the rates of uptake and reversibility of 6-fluoro-L-Dopa trapping in MPTP-induced parkinsonism in monkeys. Neuropharmacology, 36(3), 363–371.
Doudet, D. J., Chan, G. L., Holden, J. E., McGeer, E. G., Aigner, T. A., Wyatt, R. J., et al. (1998). 6-[18F]Fluoro-L-DOPA PET studies of the turnover of dopamine in MPTP-induced parkinsonism in monkeys. Synapse, 29(3), 225–232.
Dubois, B., Burn, D., Goetz, C., Aarsland, D., Brown, R. G., Broe, G. A., et al. (2007). Diagnostic procedures for Parkinson’s Disease dementia: recommendations for the Movement Disorder Society Task Force. Movement Disorders, 22(16), 2314–2324.
Firnau, G., Sood, S., Chirakal, R., Nahmias, C., & Garnett, E. S. (1988). Metabolites of 6-[18 F]fluoro-L-dopa in human blood. Journal of Nuclear Medicine, 29(3), 363–369.
Folstein, M. F., Folstein, S. E., & McHugh, P. R. (1975). “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. Journal of Psychiatric Research, 12, 189–198.
Gallagher, C., Oakes, T., Johnson, S., Chung, M., Holden, J., Bendlin, B., et al. (2011a). Rate of 6-[18 F]fluoro-L-dopa (FDOPA) uptake decline in striatal subregions in Parkinson’s disease. Movement Disorders, 4(26), 614–620.
Gallagher, C. L., Christian, B. T., Holden, J. E., Dejesus, O. T., Nickles, R. J., Buyan-Dent, L., et al. (2011b). A within-subject comparison of 6-[18 F]fluoro-m-tyrosine and 6-[18 F]fluoro-L-dopa in Parkinson's disease. Movement Disorders, 26(11), 2032–2038.
Gallagher, C. L., Bell, B., Bendlin, B., Palotti, M., Okonkwo, O., Sodhi, A., et al. (2013). White matter microstructural integrity and executive function in Parkinson's disease. Journal of the International Neuropsychological Society, 19(3), 349–354.
Golden, C. J. (1976). Identification of brain disorders by the Stroop Color and Word Test. Journal of Clinical Psychology, 32(3), 654–658.
Hoehn, M. M., & Yahr, M. D. (1967). Parkinsonism: onset, progression and mortality. Neurology, 17(5), 427–442.
Holden, J. E., Doudet, D., Endres, C. J., Chan, G. L., Morrison, K. S., Vingerhoets, F. J., et al. (1997). Graphical analysis of 6-fluoro-L-dopa trapping: effect of inhibition of catechol-O-methyltransferase. Journal of Nuclear Medicine, 38(10), 1568–1574.
Holden, J. E., Gallagher, C. L., Christian, B. T., & Sossi, V. (2010). COMT inhibition allows 6-[18 F]Fluoro-L-DOPA turnover assessment with reference tissue input functions in Parkinson Disease. NeuroImage, 52(Supplement 1), S224–S224.
Isquith, P., Roth, R., & Gioia, G. (2006). Behavior rating inventory of executive function - Adult Version. PAR, Inc.
Jordan, L. L., Zahodne, L. B., Okun, M. S., & Bowers, D. (2013). Hedonic and behavioral deficits associated with apathy in Parkinson's disease: potential treatment implications. Movement Disorders, 28(9), 1301–1304.
Kaasinen, V., Nurmi, E., Bruck, A., Eskola, O., Bergman, J., Solin, O., & Rinne, J. O. (2001). Increased frontal [18 F]fluorordopa uptake in early Parkinson’s disease: sex differences in the prefrontal cortex. Brain, 124, 1125–1130.
Kaplan, E., Goodglass, H., & Weintraub, S. (1983). Boston naming test. Philadelphia: Lee and Febiger.
Kehagia, A. A., Barker, R. A., & Robbins, T. W. (2010). Neuropsychological and clinical heterogeneity of cognitive impairment and dementia in patients with Parkinson’s disease. Lancet Neurology, 9, 1200–1213.
Li, C. T., Palotti, M., Holden, J. E., Oh, J., Okonkwo, O., Christian, B. T., et al. (2014). A dual-tracer study of extrastriatal 6-[18 F]fluoro-m-tyrosine and 6-[18 F]-fluoro-L-dopa uptake in Parkinson's disease. Synapse, 68(8), 325–331.
MacDonald, A. W., 3rd, Cohen, J. D., Stenger, V. A., & Carter, C. S. (2000). Dissociating the role of the dorsolateral prefrontal and anterior cingulate cortex in cognitive control. Science, 288(5472), 1835–1838.
Meyer-Lindenberg, A., Kohn, P. D., Kolachana, B., Kippenhan, S., McInerney-Leo, A., Nussbaum, R., et al. (2005). Midbrain dopamine and prefrontal function in humans: interaction and modulation by COMT genotype. Nature Neuroscience, 8(5), 594–596.
Moore, R. Y., Whone, A. L., McGowan, S., & Brooks, D. J. (2003). Monoamine neuron innervation of the normal human brain: an 18 F-DOPA PET study. Brain Research, 982(2), 137–145.
Moore, R. Y., Whone, A. L., & Brooks, D. J. (2008). Extrastriatal monoamine neuron function in Parkinson's disease: an 18 F-dopa PET study. Neurobiology of Disease, 29(3), 381–390.
Morrison, C. E., Borod, J. C., Brin, M. F., Raskin, S. A., Germano, I. M., Weisz, D. J., et al. (2000). A program for neuropsychological investigation of deep brain stimulation (PNIDBS) in movement disorder patients: development, feasibility, and preliminary data. Neuropsychiatry, Neuropsychology, and Behavioral Neurology, 13(3), 204–219.
Muslimovic, D., Post, B., Speelman, J. D., & Schmand, B. (2005). Cognitive profile of patients with newly diagnosed Parkinson disease. Neurology, 65(8), 1239–1245.
Paolo, A. M., Axelrod, B. N., Troster, A. I., Blackwell, K. T., & Koller, W. C. (1996). Utility of a Wisconsin card sorting test short form in persons with Alzheimer's and Parkinson's disease. Journal of Clinical and Experimental Neuropsychology, 18(6), 892–897.
Patlak, C. S., & Blasberg, R. G. (1985). Graphical evaluation of blood-to-brain transfer constants from multiple-time uptake data. Generalizations. Journal of Cerebral Blood Flow and Metabolism, 5(4), 584–590.
Patlak, C. S., Blasberg, R. G., & Fenstermacher, J. D. (1983). Graphical evaluation of blood-to-brain transfer constants from multiple-time uptake data. Journal of Cerebral Blood Flow and Metabolism, 3(1), 1–7.
Paus, T. (2001). Primate anterior cingulate cortex: where motor control, drive and cognition interface. Nature Reviews Neuroscience, 2(6), 417–424.
Rabin, L. A., Roth, R. M., Isquith, P. K., Wishart, H. A., Nutter-Upham, K. E., Pare, N., et al. (2006). Self- and informant reports of executive function on the BRIEF-A in MCI and older adults with cognitive complaints. Archives of Clinical Neuropsychology, 21(7), 721–32.
Rakshi, J. S., Uema, T., Ito, K., Bailey, D. L., Morrish, P. K., Ashburner, J., et al. (1999). Frontal, midbrain and striatal dopaminergic function in early and advanced Parkinson's disease A 3D [(18)F]dopa-PET study. Brain, 122(Pt 9), 1637–1650.
Reitan, R. (1992). Trail Making Test. Manual for administration and scoring. Tucson, AZ: Reitan Neuropsychological Laboratory.
Rinne, J. O., Portin, R., Ruottinen, H., Nurmi, E., Bergman, J., Haaparanta, M., & Solin, O. (2000). Cognitive impairment and the brain dopaminergic system in Parkinson disease: [18F]fluorodopa positron emission tomographic study. Archives of Neurology, 57(4), 470–475.
Robert, G., Le Jeune, F., Lozachmeur, C., Drapier, S., Dondaine, T., Peron, J., et al. (2012). Apathy in patients with Parkinson disease without dementia or depression. Neurology, 79, 1155–1160.
Sager, M. A., Hermann, B., & La Rue, A. (2005). Middle-aged children of persons with Alzheimer's disease: APOE genotypes and cognitive function in the Wisconsin Registry for Alzheimer's Prevention. Journal of Geriatric Psychiatry and Neurology, 18(4), 245–249.
Scatton, B., Javoy-Agid, F., Rouquier, L., Dubois, B., & Agid, Y. (1983). Reduction of Cortical Dopamine, Noradrenaline, Serotonin, and Their Metabolites in Parkinson’s Disease. Brain Research, 275, 321–328.
Sossi, V., Doudet, D. J., & Holden, J. E. (2001). A reversible tracer analysis approach to the study of effective dopamine turnover. Journal of Cerebral Blood Flow and Metabolism, 21(4), 469–476.
Sossi, V., de La Fuente-Fernandez, R., Holden, J. E., Doudet, D. J., McKenzie, J., Stoessl, A. J., et al. (2002). Increase in dopamine turnover occurs early in Parkinson's disease: evidence from a new modeling approach to PET 18 F-fluorodopa data. Journal of Cerebral Blood Flow and Metabolism, 22(2), 232–239.
Tierney, M. C., Szalai, J. P., Snow, W. G., & Fisher, R. H. (1996). The prediction of Alzheimer disease. The role of patient and informant perceptions of cognitive deficits. Archives of Neurology, 53(5), 423–427.
Wedderburn, C., Wear, H., Brown, J., Mason, S. J., Barker, R. A., Hodges, J., et al. (2008). The utility of the Cambridge Behavioural Inventory in neurodegenerative disease. Journal of Neurology, Neurosurgery, and Psychiatry, 79(5), 500–503.
Wu, K., O’Keeffe, D., Politis, M., O’Keeffe, G. C., Robbins, T. W., Bose, S. K., et al. (2012). The catechol-O-methyltransferase Val158Met polymorphism modulates fronto-cortical dopamine turnover in early Parkinson’s disease: a PET study. Brain, 135, 2449–2457.
Acknowledgments
This work was supported by the Department of Veterans Affairs, Veterans Health Administration, Office of Research and Development, Clinical Science Research and Development Service. It was also funded by the University of Wisconsin Institute for Clinical and Translational Research, through a National Institutes of Health Clinical and Translational Science Award, [grant number 1UL1RR025011]. This work was supported with use of facilities at the William S. Middleton Memorial Veterans Hospital Geriatric Research Education and Clinical Center and the Waisman Laboratory for Brain Imaging and Behavior, Madison, WI, USA.
Disclosures
Authors Gallagher, Bell, Palotti, Oh, Christian, Okonkwo, Sojkova, Buyan-Dent, Nickles, Harding, Stone, Johnson, and Holden declare no conflicts of interest.
Informed Consent
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, and the applicable revisions at the time of this investigation. Informed consent was obtained from all study participants.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gallagher, C.L., Bell, B., Palotti, M. et al. Anterior cingulate dopamine turnover and behavior change in Parkinson’s disease. Brain Imaging and Behavior 9, 821–827 (2015). https://doi.org/10.1007/s11682-014-9338-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11682-014-9338-4
Keywords
- Adult
- Aged
- Humans
- Brain mapping
- Cerebral cortex/metabolism/radionuclide imaging
- Dopamine agents/*diagnostic use/pharmacokinetics
- Dihydroxyphenylalanine/*analogs & derivatives/drug effects/pharmacokinetics
- Positron emission tomography
- Parkinson disease/*physiopathology/*radionuclide imaging
- Research support/U.S. Gov’t/P.H.S.