Abstract
Objective
To investigate the expression of beta-catenin and VEGFs (VEGF-A, VEGF-C) and VEGF receptor-2 (VEGFR-2) protein in medulloblastoma.
Methods
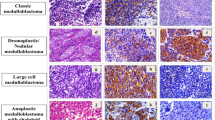
Immunohistochemical staining with SP method was conducted to determine the expression of beta-catenin and VEGFs (VEGF-A, VEGF-C) and VEGFR-2 in 33 cases of medulloblastoma and 10 normal cerebellar tissues.
Results
The expression rate of beta-catenin, and VEGFs (VEGF-A, VEGF-C) and VEGFR-2 in medulloblastoma were significantly higher than that in normal tissue. A significant positive correlation was found between beta-catenin and VEGFs (VEGF-A, VEGF-C) and VEGFR-2 protein in medulloblastoma.
Conclusion
There was a correlation between beta-catenin and VEGFs (VEGF-A, VEGF-C) and VEGFR-2 in medulloblastoma, which may play a role in the pathogenesis and development of medulloblastoma.
Similar content being viewed by others
References
Moon RT, Kohn AD, De Ferrari GV, et al. WNT and beta-catenin signaling: diseases and therapies [J]. Nat Rev Genet 2004; 5:691–701.
Massod R, Cai J, Zheng T, et al. Vascular endothelial growth factor (VEGF) is an autocrine growth factor for VEGF receptor-positive human tumors [J]. Blood 2001;98:1904–1913.
Slongo ML, Molena B, Brunati Am, et al. Functional VEGF and VEGF receptors are expressed in human medulloblastomas [J]. Neuro Oncol 2007; 9:384–392.
Yokota N, Nishizawa S, Ohta S, et al. Role of Wnt pathway in medulloblastoma oncogenesis [J]. Int J Cancer 2002; 101:198–201.
Takanami I, Tanaka F, Hashizume T, et al. Vascular endothelial growth factor and its receptor correlate with angiogenesis and survival in pulmonary adenocarcinoma [J]. Anticancer Res 1997; 17:2811–2814.
Padró T, Bieker R, Ruiz S, et al. Overexpression of vascular endothelial growth factor (VEGF) and its cellular receptor KDR (VEGFR-2) in the bone marrow of patients with acute myeloid leukemia [J]. Leukemia 2002; 16:1302–1310.
Krieghoff E, Behrens J, Mayr B, et al. Nucleo-cytoplasmic distribution of beta-catenin is regulated by retention [J]. J Cell Sci 2006; 119:1453–1463.
Polakis P. Wnt signaling and cancer [J]. Genes Dev 2000;14:1837–1851.
Skurk C, Maatz H, Rocnik E, et al. Glycogen-Synthase kinase3beta/beta-catenin axis promotes angiogenesis through activation of vascular endothelial growth factor signaling in endothelial cells [J]. Circ Res 2005; 96:308–318.
RA Goodlad, AJ Ryan, SR Wedge, et al. Inhibiting vascular endothelial growth factor receptor-2 signaling reduces tumor burden in the ApcMin/+ mouse model of early intestinal cancer [J]. Carcinogenesis 2006; 27:2133–2139.
Easwaran V, Lee SH, Inge L, et al. beta-catenin regulates vascular endothelial growth factor expression in colon cancer [J]. Cancer Res 2003; 63:3145–3153.
Monkley SJ, Delaney SJ, Pennisi DJ, et al. Targeted disruption of the Ent2 gene results in placentation defects [J]. Development 1996; 122:3343–3353.
Ishikawa T, Tamai Y, Zorn AM, et al. Mouse Wnt receptor gene Fzd5 is essential for yolk sac and placental angiogenesis [J]. Development 2001; 128:25–33.
Kim KI, Cho HJ, Hahn JY, et al. beta-catenin overexpression augments angiogenesis and skeletal muscle regeneration through dual mechanism of vascular endothelial growth factor-mediated endothelial cell proliferation and progenitor cell mobilization [J]. Arterioscler Thromb Vasc Biol 2006; 26:91–98.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, Hm., Zhang, X., Li, Y. et al. Relationship between expression of beta-catenin and VEGFs (VEGFA, VEGF-C), VEGF receptors-2 (VEGFR-2) in medulloblastoma. Chin. J. Cancer Res. 20, 44–48 (2008). https://doi.org/10.1007/s11670-008-0044-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11670-008-0044-x