Abstract
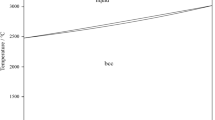
A deep knowledge of phase relationships involving a liquid phase is of critical interest in solidification process. If chemically active elements are involved, the conditions under which phase equilibria will be studied should be carefully defined. An illustration is given by the investigation of the Fe-rich corner of the Fe-Ti-B system between 1200 and 1450 °C, for which Ti and B reactivity, very low solubility and flotation effects of TiB2 should be taken into account. The strategy is to combine both static and dynamic methods. Electromagnetic phase separation technique, developed at our laboratory, allows to determine phase field boundaries in the liquid/solid domains at selected temperatures and to locate univariant lines on a liquidus projection. Differential thermal analysis is supplemented by characterization of resulting microstructures to establish liquidus temperature of selected alloys and to evaluate ternary invariant points. These new experimental data may be used as a basis for a relevant thermodynamic description of the investigated ternary system.










Similar content being viewed by others
References
V. Raghavan, The B-Fe-Ti (Boron-Iron-Titanium) System, Phase Diagrams of Ternary Iron Alloys, Part 6, Indian Institute of Metals, Calcutta, 1992, p 430-440
V. Raghavan, B-Fe-Ti (Boron-Iron-Titanium), J. Phase Equilib., 2003, 24(5), p 455-456
T.F. Federov and Yu.B. Kuzma, The Titanium-Iron-Boron System, Inorg. Mater., 1967, 3(8), p 1307-1308
L. Ottavi, J.M. Chaix, C. Allibert, and H. Pastor, Thermodynamic Guidelines for the Liquid Phase Sintering of TiB2 Cermets, Solid State Phenom., 1992, 25-26, p 543-550
L. Ottavi, C. Saint-Jours, N. Valignat, and C.H. Allibert, Phase Equilibria and Solidification of Fe-Ti-B Alloys in the Region Close to Fe-TiB2, Z. Metall., 1992, 83(2), p 80-83
G.V. Samsonov, Nature of Interaction Between Titanium Boride and the Iron-Group Metals, Metall. Term. Obrabot. Metall., 1958, 1, p 35-38
V.I. Eremenko, Multicomponent Titanium Alloys, Izd. AN USSR Kiev, 1962, cited by [3]
A.K. Shurin and V.E. Panarin, Phase Equilibria and Structure of Alloys Fe-TiB2, Fe-Zr B2 and Fe-HfB2, Russ. Metall., 1974, 5, p 192-195
I. Smid and E. Kny, Evaluation of Binder Phases for Hard Metal Systems Based On TiB2, Int. J. Refract. Hard Met., 1988, 7, p 135-138
C. Allibert, A. Wicker, J. Driole, and E. Bonnier, Détermination de diagrammes de phases à haute température dans les systèmes de métaux réfractaires, Rev. Int. Hautes Temp., 1970, 7, p 45-50
C. Allibert, A. Wicker, J. Driole, and E. Bonnier, Etude d’alliages de métaux réfractaires à base de niobium, Rev. Phys. Appl., 1970, 5, p 449-453
J. Driole, C. Allibert, E. Bonnier, and A. Wicker, Process of Separation, Particularly of a Solid Phase, from a Matrix in Liquid Phase, United States, Agence Nationale de Valorisation de la Recherche (ANVAR) (Neuilly-sur-Seine, France), Patent 4049437, 1977. http://www.freepatentsonline.com/4049437.html
B. Sundman, B. Jansson, and J.O. Andersson, The Thermocalc Databank System, CALPHAD, 1985, 9(2), p 153-190
SGTE Alloy Solutions Database SSOL4 Version 4.10. Thermo-Calc Software AB, Stockholm, Sweden, 2008
B. Hallemans, P. Wollants, and J.R. Roos, Thermodynamic Reassessment and Calculation of the Fe-B Phase Diagram, Z. Metall., 1994, 85(10), p 676-682
K.C. Hari Kumar, L. Dumitrescu, B. Sundman, and P. Wollants, Thermodynamic Assessment of the Fe-Ti System with Special Emphasis on the Modelling of the FeTi (B2) Phase, XXVIII Calphad Meeting, Grenoble, May 2-7 1999, p 95
C. Bätzner, System B-Ti, COST 507, Thermochemical Database for Light Metal Alloys, Eur18499, I. Ansara, A.T. Dinsdale, and M.H. Rand, Eds., Vol. 2, 1998, p 129-134
A.T. Dinsdale, SGTE Data for Pure Elements, CALPHAD, 1991, 15(4), p 317-425
O. Redlich and A.T. Kister, Algebraic Representation of Thermodynamic Properties and the Classification of Solutions, Ind. Eng. Chem., 1948, 40, p 345-348
Y.M. Muggianu, M. Gambino, and J.P. Bros, Enthalpies of Formation of Liquid Alloy Bismuth-Gallium-Tin at 723 K, Choice of An Analytical Representation of Integral and Partial Excess Functions of Mixing, J. Chim. Phys., 1975, 72, p 83-88
B. Sundman and J. Ågren, A Regular Solution Model for Phases with Several Components and Sublattices, Suitable for Computer Applications, J. Comput. Phys. Chem. Solids, 1981, 42, p 297-301
J.O. Anderson, A.F. Guillermet, M. Hillert, B. Jansson, and B. Sundman, A Compound-Energy Model of Ordering in a Phase with Sites of Different Coordination Numbers, Acta Metall., 1986, 34(3), p 437-445
M. Hillert, The Compound Energy Formalism, J. Alloys Compd., 2001, 320(2), p 161-176
G.H. Geigerand and D.R. Poirier, Transport Phenomena in Metallurgy, Addison-Wesley Publishing Company, Reading, 1980, p 461
S.H. Moll and R.E. Ogilvie, Solubility and Diffusion of Titanium in Iron, Trans. TMS AIME, 1959, 215(4), p 613-618
S. Nakamichi, S. Tsurekawa, Y. Morizono, M. Nishiba, and A. Chiba, Diffusion of Carbon and Titanium in γ-Iron in a Magnetic Field and a Magnetic Field Gradient, J. Mater. Sci., 2005, 40, p 3191-3198
P.E. Busby, M.E. Warga, and C. Wells, Diffusion and Solubility of Boron in Iron and Steel, Trans. TMS AIME, 1953, 197(11), p 1463-1468
Acknowledgments
This work is supported by the French National Research Agency (ANR) through the ADRERA project ANR-09-MAPR-0001.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Antoni-Zdziobek, A., Gospodinova, M., Bonnet, F. et al. Experimental Determination of Solid-Liquid Equilibria with Reactive Components: Example of the Fe-Ti-B Ternary System. J. Phase Equilib. Diffus. 35, 701–710 (2014). https://doi.org/10.1007/s11669-014-0355-1
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11669-014-0355-1