Abstract
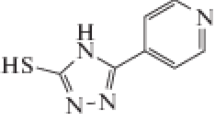
The corrosion inhibition characteristics of rutin a natural flavonoid glycoside has been studied as an eco-friendly green inhibitor for corrosion control of aluminum alloy AA6063 in 0.5 M HCl medium at a temperature range of 30–60 °C by weight loss method, electrochemical impedance spectroscopy (EIS) and potentiodynamic polarization (PDP) techniques. The inhibition efficiency increased with the increase in concentration of the inhibitor and also with increase in temperature. The surface morphologies of the polished, corroded and inhibited surfaces were scanned using the scanning electron microscope images. The activation parameters like energy of activation, enthalpy of activation and entropy of activation were calculated and analyzed. Thermodynamic parameters for the adsorption of the inhibitor on the metal alloy surface were calculated and analyzed. The inhibitor acted as a mixed inhibitor. The mechanism of adsorption deduced from the variation of inhibition efficiency with temperature as well as kinetic and activation parameters suggests significant chemisorption of the inhibitor on the metal surface. The mechanism of inhibition can partly be attributed to the possible coordination of rutin to form stable aluminum–rutin complex. The maximum inhibition efficiency was found to be 95.1 and 94.45% by PDP and EIS studies, respectively, at 60 °C. Density functional theory calculations under the level B3LYP/6-311G(d) is utilized for the calculation of quantum chemical parameters.













Similar content being viewed by others
References
V. Branzoi, F. Golgovici, F. Branzoi, Aluminium corrosion in hydrochloric acid solutions and the effect of some organic inhibitors. Mater. Chem. Phys. 78, 122–131 (2002)
A.M. Abdel-Gaber, E. Khamis, H. Abo-Eldahab, S.H. Adeel, Inhibition of aluminium corrosion in alkaline solutions using natural compound. Mater. Chem. Phys. 109, 297–305 (2008)
E.I. Ating, S.A. Umoren, I.I. Udousoro, E.E. Ebenso, P. Udoha, Leaves extract of ananassativum as green corrosion inhibitor for aluminium in hydrochloric acid solutions. Green Chem. Lett. Rev. 3, 61–68 (2010)
H. Javed, M.M. Khan, A. Ahmad, K. Vaibhav, M.E. Ahmed, A. Khan, M. Ashafaq, F. Islam, M.S. Siddiqui, M.M. Sahi, F. Islam, Rutin prevents cognitive impairments by ameliorating oxidative stress and neuro inflammation in rat model of sporadic dementia of Alzheimer type. J. Neurosci. Res. 17, 340–352 (2012)
M. Nassiri Asl, S.R. Mortazavi, F. Samiee Rad, Z. Amir-Abdollah, F. Safdari, S. Saroukhani, E. Abbasi, Effects of rutin on the development of pentylene tetrazole kindling and memory retrieval in rats. Epilepsy Behav. 18, 50–53 (2010)
U.M. Eduok, S.A. Umoren, A.P. Udoh, Synergistic inhibition effects between leaves and stem extracts of Sidaacuta and iodide ion for mild steel corrosion in 1 M H2SO4 solutions. Arab. J. Chem. 5, 325–337 (2010)
V.O. Njoku, E.E. Oguzie, C. Obi, A.A. Ayuk, Baphianitida leaves extract as a green corrosion inhibitor for the corrosion of mild steel in acidic media. Adv. Chem. 10, 1155 (2014)
A. Popova, S. Raicheva, E. Sokolova, M. Christov, Frequency dispersion of the interfacial impedance at mild steel corrosion in acid media in the presence of benzimidazole derivatives. Langmuir 8, 2083–2089 (1996)
P.D. Reena, J. Nayak, A. Nityananda Shetty, 3-Methyl-4-amino-5-mercapto-1,2,4-triazole as corrosion inhibitor for 6061/Al-15 (vol%) SiC(p) composite in 0.5 M sodium hydroxide solution. J. Mater. Environ. Sci. 4, 387–402 (2011)
E.E. Oguzie, B.N. Okolue, E.E. Ebenso, N. Onuoha, A.I. Onuchukwu, Evaluation of the inhibitory effect of methylene blue dye on the corrosion of aluminuium in hydrochloric acid. Mater. Chem. Phys. 87, 394–401 (2004)
X. Li, S. Deng, X. Xie, Experimental and theoretical study on corrosion inhibition of o-phenanthroline for aluminum in HCl solution. J. Taiwan Inst. Chem. Eng. 45, 1865–1875 (2014)
A.K. Singh, M. Quraishi, Effect of cafazolin on the corrosion of mild steel in HCl solution. Corros. Sci. 52, 152–160 (2010)
E.A. Noor, Potential of aqueous extract of Hibiscus sabdariffa leaves for inhibiting the corrosion of aluminum in alkaline solutions. J. Appl. Electrochem. 39, 1465–1475 (2014)
A. Popova, M. Christov, S. Raicheva, E. Sokolov, Adsorption and inhibitive properties of benzimidazole derivatives in acid mild steel corrosion. Corros. Sci. 46, 1333–1350 (2004)
A. Hamdy, N.S. El-Gendy, Thermodynamic, adsorption and electrochemical studies for corrosion inhibition of carbon steel by henna extract in acid medium. Egypt J Pet. 22, 17–25 (2013)
A.A.H. Kadhum, A.B. Mohamad, L.A. Hammed, A.A. AI-Amiery, N.H. San, A.Y. Musa, Inhibition of mild steel corrosion in hydrochloric acid solution by New Coumarin. Materials 7, 4335–4348 (2014)
R.K. Hart, The formation of films on aluminium immersed in water. Trans. Faraday Soc. 53, 1020–1027 (1957)
G. Gece, The use of quantum chemical methods in corrosion inhibitor studies. Corros. Sci. 50, 2981–2992 (2008)
L.O. Olasunkanmi, I.B. Obot, M.M. Kabanda, E.E. Ebenso, Some quinoxalin-6-yl derivatives as corrosion inhibitors for mild steel in hydrochloric acid: experimental and theoretical studies. J. Phys. Chem. 119, 16004–16019 (2015)
T. Koopmans, Uber die Zuordnung von Wellenfunktionenund. Eigenwertenzu den Einzelnen Elektronen Eines Atoms. Physica 1, 104–113 (1934)
R.G. Pearson, Absolute electronegativity and hardness correlated with molecular orbital theory. Proc. Natl. Acad. Sci. 83, 8440–8441 (1986)
I.B. Obot, N.O. Obi-Egbedi, An interesting and efficient green corrosion inhibitor for aluminium from extracts of Chlomolaenaodorata L. in acidic solution. J. Appl. Electrochem. 40, 1977–1984 (2010)
S.V. Jovanovic, S. Steenken, M.G. Simic, Y. Hara, Antioxidant properties of flavonoids, in Reduction Potentials and Electron Transfer Reactions of Flavonoid Radicals, in Flavonoids in Health and Disease, ed. by C.A. Rice-Evans, L. Packer (Marcel Dekker, Inc, New York, 1998), pp. 137–161
S.V. Kuntic, L.D. Malesev, V.Z. Radovic, M.M. Kosanic, B.U. Mioc, B.V. Vukojevic, Spectrophotometric investigation of uranil (II)-rutin complex in 70% ethanol. J. Agric. Food Chem. 46, 5139–5142 (1998)
Acknowledgments
The authors would like to acknowledge the department of chemistry NITK, Surathkal, and department of physics SMVITM for providing Instrumentation and basic computational facilities.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Rights and permissions
About this article
Cite this article
Reena Kumari, P.D., Kumari, D. Experimental and Theoretical Evaluation of Rutin as Eco-Friendly Corrosion Inhibitor for Aluminum 6063 Alloy in Acidic Medium. J Fail. Anal. and Preven. 18, 856–867 (2018). https://doi.org/10.1007/s11668-018-0473-x
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11668-018-0473-x