Abstract
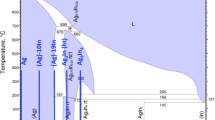
The reaction of indium (In) and silver (Ag) during the electroplating process of indium over a thick silver layer was investigated. It was found that the plated In atoms react with Ag to form AgIn2 intermetallic compounds at room temperature. Indium is commonly used in the electronics industry to bond delicate devices due to its low yield strength and low melting temperature. In this study, copper (Cu) substrates were electroplated with a 60-μm-thick Ag layer, followed by electroplating an In layer with a thickness of 5 μm or 10 μm, at room temperature. To investigate the chemical reaction between In and Ag, the microstructure and composition on the surface and the cross section of samples were observed by scanning electron microscopy (SEM) with energy-dispersive x-ray spectroscopy (EDX). The x-ray diffraction method (XRD) was also employed for phase identification. It was clear that indium atoms reacted with underlying Ag to form AgIn2 during the plating process. After the sample was stored at room temperature in air for 1 day, AgIn2 grew to 5 μm in thickness. With longer storage time, AgIn2 continued to grow until all indium atoms were consumed. The indium layer, thus, disappeared and could barely be detected by XRD.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
S. Choe, W.W. So, and C.C. Lee, Proc. IEEE Electron. Compon. Technol. Conf. (2000), p. 114.
C. C. Lee, C. Y. Wang, and G. Matijasevic, IEEE Trans. Compon. Hybr. Manuf. Tech. 16, 311 (1993). doi:10.1109/33.232058.
Y.-C. Chen, W. W. So, and C. C. Lee, IEEE Trans. Compon. Packag. Manuf. Technol. 20, 46 (1997). doi:10.1109/95.558543.
C. C. Lee and W. W. So, Thin Solid Films 336, 196 (2000). doi:10.1016/S0040-6090(00)00727-6.
K.-M. Chu, J.-H. Choi, J.-S. Lee, H. S. Cho, S.-O. Park, H.-H. Park, and D. Y. Jeon, IEEE Trans. Adv. Packag. 29, 409 (2006). doi:10.1109/TADVP.2006.875417.
P. T. Vianco, J. A. Rejent, A. F. Fossum, and M. K. Neilsen, J. Mater Sci: Mater. Electron. 18, 93 (2007). doi:10.1007/s10854-006-9013-7.
H. Okamoto and T.B. Massalski, Binary Alloy Phase Diagrams (Metal Park, OH: ASM International, 1990), pp. 381–383.
B.D. Cullity, Elements of X-ray Diffraction, 2nd ed. (Addison Wesley, 1978), p. 513.
R. Roy and S. K. Ken, Thin Solid Films 197, 303 (1991). doi:10.1016/0040-6090(91)90241-O.
V. Simic and Z. Marinkovic, Thin Solid Films 61, 149 (1979). doi:10.1016/0040-6090(79)90457-7.
J. S. Kim, P. J. Wang, and C. C. Lee, IEEE Trans. Compon. Packag. Technol 31, 782 (2008). doi:10.1109/TCAPT.2008.2001195.
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
Author information
Authors and Affiliations
Corresponding author
Additional information
Jong S. Kim now with Applied Materials.
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Wang, P.J., Kim, J.S. & Lee, C.C. Intermetallic Reaction of Indium and Silver in an Electroplating Process. J. Electron. Mater. 38, 1860–1865 (2009). https://doi.org/10.1007/s11664-009-0845-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11664-009-0845-9