Abstract
Objective
To evaluate the effect of Pien Tze Huang (片仔癀, PZH) on breast cancer chemoresistance and related epithelial-mesenchymal transition (EMT) and investigate the underlying mechanisms.
Methods
3-(4,5-Dimethyl-2-thiazolyl)-2,5-diphenyl-2-H-tetrazolium bromide (MTT) assay was used to determine the cell viability. Adriamycin (ADR) staining observed by fluorescence microscope was performed to detect the accumulation of ADR. Transwell assay was used to analyze the cell migration and invasion. Western-blot was performed to detect the protein expression of related genes.
Results
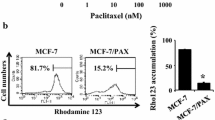
MCF-7/ADR cells were resistant to ADR treatment, and PZH treatment inhibited the viability of MCF-7/ADR cells in a dose-dependent manner. PZH treatment also increased the intercellular accumulation of ADR and down-regulated the expression of ABCG2 and ABCB1 in MCF-7/ADR cells (P<0.05). In addition, PZH treatment inhibited EMT, migration and invasion of MCF-7/ADR cells (P<0.05). Moreover, PZH suppressed activation of transforming growth factor β1 (TGF-β) signaling in MCF-7/ADR cells (P<0.05).
Conclusion
PZH treatment can effectively overcome chemoresistance via down regulating ABCG2, ABCB1 and inhibit EMT in ADR resistant human breast cancer cells via suppression of the TGF-β1 pathway.
Similar content being viewed by others
References
Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. CA Cancer J Clin 2015;65:87–108.
Del Mastro L, De Placido S, Bruzzi P, De Laurentiis M, Boni C, Cavazzini G, et al. Fluorouracil and dose-dense chemotherapy in adjuvant treatment of patients with early-stage breast cancer: an open-label, 2×2 factorial, randomised phase 3 trial. Lancet 2015;385:1863–1872.
Martín M, Ruiz A, Ruiz Borrego M, Barnadas A, González S, Calvo L, et al. Fluorouracil, doxorubicin, and cyclophosphamide (FAC) versus FAC followed by weekly paclitaxel as adjuvant therapy for high-risk, node-negative breast cancer: results from the GEICAM/2003-02 study. J Clin Oncol 2013;31:2593–2599.
Higgins CF. Multiple molecular mechanisms for multidrug resistance transporters. Nature 2007;446:749–757.
Zhu MM, Tong JL, Xu Q, Nie F, Xu XT, Xiao SD, et al. Increased JNK1 signaling pathway is responsible for ABCG2-mediated multidrug resistance in human colon cancer. PLoS One 2012;7:e41763.
Szakács G, Paterson JK, Ludwig JA, Booth-Genthe C, Gottesman MM. Targeting multidrug resistance in cancer. Nat Rev Drug Discov 2006;5:219–234.
Thiery JP, Acloque H, Huang RY, Nieto MA. Epithelial-mesenchymal transitions in development and disease. Cell 2009;139:871–890.
Kalluri R, Weinberg RA. The basics of epithelial mesenchymal transition. J Clin Invest 2009;119:1420–1428.
Rosanò L, Cianfrocca R, Spinella F, Di Castro V, Nicotra MR, Lucidi A, et al. Acquisition of chemoresistance and EMT phenotype is linked with activation of the endothelin A receptor pathway in ovarian carcinoma cell. Clin Cancer Res 2011;17:2350–2360.
Singh A, Settleman J. EMT, cancer stem cells and drug resistance: an emerging axis of evil in the war on cancer. Oncogene 2010;29:4741–4751.
Ieri OD, Kars MD, Arpaci F, Atalay C, Pak I, Gündüz U. Drug resistant MCF-7 cells exhibit epithelial-mesenchymal transition gene expression pattern. Biomed Pharmacother 2011;65:40–45.
Turley EA, Veiseh M, Radisky DC, Bissell MJ. Mechanisms of disease: epithelial-mesenchymal transition-dose cellular plasticity fuel neoplastic progression? Nat Clin Pract Oncol 2008;5:280–290.
Xu J, Lamouille S, Derynck R. TGF-β-induced epithelial to mesenchymal transition. Cell Res 2009;19:156–172.
Allendorph GP, Read JD, Kawakami Y, Kelber JA, Isaacs MJ, Choe S. Designer TGF-β superfamily ligands with diversified functionality. PLoS One 2011;6:e26402.
Qi F, Zhao L, Zhou A, Zhang B, Li A, Wang Z, et al. The advantages of using traditional Chinese medicine as an adjunctive therapy in the whole course of cancer treatment instead of only terminal stage of cancer. BioSci Trends 2015;9:16–34.
Chinese Pharmacopoeia Commission. Pharmacopoeia of the Peoples Republic of China. Vol. 1. Beijing: Chinese Medical Science and Technology Press; 2010:573–575.
Lee KK, Kwong WH, Chau FT, Yew DT, Chan WY. Pien Tze Huang protects the liver against carbon tetrachlorideinduced damage. Pharmacol Toxicol 2002;91:185–192.
Chan WY, Chau FT, Lee KK, Kwong WH, Yew DT. Substitution for natural musk in Pien Tze Huang does not affect its hepatoprotective activities. Hum Exp Toxicol 2004;23:35–47.
Lin JM, Wei LH, Chen YQ, Liu XX, Hong ZF, Sferra TJ, et al. Pien Tze Huang induced apoptosis in human colon cancer HT-29 cells is associated with regulation of the Bcl-2 family and activation of caspase 3. Chin J Integr Med 2011;17:685–690.
Zhuang Q, Hong F, Shen A, Zheng L, Zeng J, Lin W, et al. Pien Tze Huang inhibits tumor cell proliferation and promotes apoptosis via suppressing the STAT3 pathway in colorectal cancer mouse. Int J Oncol 2012;40:1569–1574.
Shen AL, Hong F, Liu LY, Lin JM, Zhuang QC, Hong ZF, et al. Effects of Pien Tze Huang on angiogenesis in vivo and in vitro. Chin J Integr Med 2012;18:431–436.
Shen A, Chen Y, Hong F, Lin J, Wei L, Hong Z, et al. Pien Tze Huang suppresses IL-6-inducible STAT3 activation in human colon carcinoma cells through induction of SOCS3. Oncol Rep 2012;28:2125–2130.
Shen A, Lin J, Chen Y, Lin W, Liu L, Hong Z, et al. Pien Tze Huang inhibits tumor angiogenesis in a mouse model of colorectal cancer via suppression of multiple cellular pathways. Oncol Rep 2013;30:1701–1706.
Lin JM, Feng JY, Jin YY, Yan ZK, Lai ZJ, Peng J. Pien Tze Huang suppresses VEGF-C-mediated lymphangiogenesis in colorectal cancer. Oncol Rep 2016;36:3568–3576.
Fu Y, Zhang L, Hong Z, Zheng H, Li N, Gao H, et al. Methanolic extract of Pien Tze Huang hnduces apoptosis signaling in human osteosarcoma MG63 cells via multiple pathways. Molecules 2016;21:283
He F, Wu HN, Cai MY, Li CP, Zhang X, Wan Q, et al. Inhibition of ovarian cancer cell proliferation by Pien Tze Huang via the AKT-mTOR pathway. Oncol Lett 2014;7:2047–2052.
Wei L, Chen P, Chen Y, Shen A, Chen H, Lin W, et al. Pien Tze Huang suppresses the stem-like side population in colorectal cancer cells. Mol Med Rep 2014;9:261–266.
Qi F, Wei L, Shen A, Chen Y, Lin J, Chu J, et al. Pien Tze Huang inhibits the proliferation, and induces the apoptosis and differentiation of colorectal cancer stem cells via suppression of the Notch1 pathway. Oncol Rep 2016;35:511–517.
Lin W, Zhuang Q, Zheng L, Cao Z, Shen A, Li Q, et al. Pien Tze Huang inhibits liver metastasis by targeting TGF-β signaling in an orthotopic model of colorectal cancer. Mol Rep 2015;33:1922–1928.
Shen A, Lin W, Chen Y, Liu L, Chen H, Zhuang Q, et al. Pien Tze Huang inhibits metastasis of human colorectal carcinoma cells via modulation of TGF-β/ZEB/miR-200 signaling network. Int J Oncol 2015;46:685–690.
Shen A, Chen H, Chen Y, Lin J, Lin W, Liu L, et al. Pien Tze Huang overcomes multidrug resistance and epithelialmesenchymal transition in human colorectal carcinoma cells via suppression of TGF-β pathway. Evid Based Complement Alternat Med 2014;2014:679436.
Zhang Y, Wang Q, Niu S, Liu J, Zhang L. Pien Tze Huang induces apoptosis in multidrug-resistant U2OS/ADM cells via downregulation of Bcl-2, survivin and P-gp and upregulation of Bax. Oncol Rep 2014;31:763–770.
Ji XM, Wu ZC, Liu GW, Yu HY, Liu H, Wang ZT, et al. Wenxia Changfu Formula induces apoptosis of lung adenocarcinoma in a transplanted tumor model of drugresistance nude mice. Chin J Integr Med 2016;22:752–758.
Sui H, Pan SF, Feng Y, Jin BH, Liu X, Zhou LH, et al. Zuo Jin Wan reverses P-gp-mediated drug-resistance by inhibiting activation of the PI3K/Akt/NF-κB pathway. BMC Complement Altern Med 2014;14:279.
Li WB, Li Y, Yu C, He YM. Reversal of multidrug resistance by the Chinese medicine Yiqi Jianpi Huaji Decoction and the mechanism of action in human gastric cancer SGC7901/VCR cells. Evid Based Complement Alternat Med 2015;2015:390812.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by the National Natural Science Foundation of China (No. 81673721), Natural Science Foundation of Fujian Province (No. 2018j01352), and the Developmental Fund of Chen Keji Integrative Medicine (No. CKJ2014004)
Rights and permissions
About this article
Cite this article
Chen, X., Qi, F., Shen, Al. et al. Pien Tze Huang (片仔癀) Overcomes Doxorubicin Resistance and Inhibits Epithelial-Mesenchymal Transition in MCF-7/ADR Cells. Chin. J. Integr. Med. 25, 598–603 (2019). https://doi.org/10.1007/s11655-018-2992-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11655-018-2992-4