Abstract
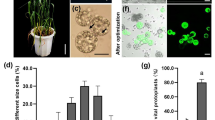
The ability to screen the functionality of gene constructs in a transient system of appropriate tissues informs the researcher of the potential success of stable transformation. For this purpose, we developed a transient system to test the functionality of endosperm-preferred promoters in maize. Two endosperm-preferred promoters from rice (a globulin and a glutelin promoter) were employed. Ears of Zea mays L. were harvested at 17 d after pollination, surface sterilized and the endosperm excised. Using Agrobacterium tumefaciens co-cultivation and sonication, transient expression of the target genes was detected after 4 and 5 d. We demonstrate expression of CBH I and CHB II (both exo-cellulases) up to 1.7% TSP, under the rice globulin and glutelin promoters.







Similar content being viewed by others
References
An G.; Mitra A.; Choi H.; Costa M.; An K.; Thornburg R. Functional analysis of the 3′ control region of the potato wound-inducible proteinase inhibitor II gene. Plant Cell 1: 115–122; 1989.
Armstrong C.; Green C.; Phillips R. Development and availability of germplasm with high type II culture formation response. Maize Genet Coop Newsl 65: 92–93; 1991.
Bradford M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72: 248–254; 1976.
Christensen A. H.; Sharrock R. A.; Quail P. H. Maize polyubiquitin genes: structure, thermal perturbation of expression and transcript splicing, and promoter activity following transfer to protoplasts by electroporation. Plant Molecular Biology 18: 675–689; 1992.
Doehlert D.; Smith L.; Duke E. Gene expression during maize kernel development. Seed Science Research 4: 299–305; 1994.
Hansch R.; Koprek T.; Mendel R. R.; Shulze J. An improved protocol for eliminating endogenous b-glucuronidase background in barley. Plant Sci 105: 63–69; 1995.
Hiei Y.; Ohta S.; Komari T.; Kumashiro T. Efficient transformation of rice (Oryza sativa L.) mediated by Agrobacterium and sequence analysis of the boundaries of the T-DNA. Plant Journal 6: 271–282; 1994.
Hood E.; Bailey M.; Beifuss K.; Magallanes-Lundback M.; Horn M.; Callaway E.; Drees C.; Delaney D.; Clough R.; Howard J. Criteria for high-level expression of a fungal laccase gene in transgenic maize. Plant Biotechnology J 1: 129–140; 2003.
Hood E.; Helmer G.; Fraley R.; Chilton M. The hypervirulence of Agrobacterium tumefaciens A281 is encoded in a region of pTiBo542 outside of T-DNA. J Bacteriol 168: 1291–1301; 1986.
Hood E. E.; Love R.; Lane J.; Bray J.; Clough R.; Pappu K. Subcellular targeting is a key condition for high-level accumulation of cellulase protein in transgenic maize seed. Plant Biotechnology J. 5: 709–719; 2007.
Horn M.; Harkey R.; Vinas A.; Drees C.; Barker D.; Lane J. Use of hi ii-elite inbred hybrids in Agrobacterium-based transformation of maize. In vitro cellular & developmental biology 42: 359–366; 2006.
Ishida Y.; Saito H.; Ohta S.; Hiei Y.; Komari T.; Kumashiro T. High efficiency transformation of maize (Zea mays L.) mediated by Agrobacterium tumefaciens. Nat Biotechnol 14: 745–750; 1996.
Kodrzycki R.; Boston R.; Larkins B. The opaque-2 mutation of maize differentially reduces zein gene transcription. Plant Cell 1: 105–114; 1989.
Rogers J. Two barley alpha-amylase gene families are regulated differently in aleurone cells. J Biol Chem 260: 3731–3738; 1985.
Russell D.; Fromm M. Tissue-specific expression in transgenic maize of four endosperm promoters from maize and rice. Transgenic Research 6: 157–168; 1997.
Shoemaker S.; Schweickart V.; Ladner M.; Gelfand D.; Kwok S.; Myambo K. Molecular cloning of exocellobiohydrolase I derived from Trichoderma reesei strain. Bio/Technology 1: 691–696; 1983.
Streatfield S. J.; Magallanes-Lundback M. E.; Beifuss K. K.; Brooks C. A.; Harkey R. L.; Love R. T.; Bray J.; Howard J. A.; Jilka J. M.; Hood E. E. Analysis of the maize polyubiquitin-1 promoter heat shock elements and generation of promoter variants with modified expression characteristics. Transgenic Res 13: 299–312; 2004.
Taylor M. G.; Vasil V.; Vasil I. K. Enhanced GUS gene expression in cereal/grass cell suspensions and inmature embryos using the maize ubiquitin-based plasmid pAHC25. Plant Cell Reports 12: 491–495; 1993.
Trick H.; Finer J. SAAT: sonication-assisted Agrobacterium-mediated transformation. Transgenic Research 6: 329–336; 1997.
White R.; Chang S.-Y.; Bibb J. A cassette containing the bar gene of Streptomyces hygroscopicus: a selectable marker for plant transformation. Nucl Acids Res 18: 1062; 1992.
Woo Y.; Hu D.; Larkins B.; Jung R. Genomics analysis of genes expressed in maize endosperm identifies novel seed proteins and clarifies patterns of zein gene expression. Plant Cell 13: 2297–2317; 2001.
Acknowledgments
This work received financial support from the US Department of Energy and the Walton Family Foundation. We express our gratitude to Yeun-Kyung Chang and Ashley Flory for their collaboration in providing assistance with this study.
Author information
Authors and Affiliations
Corresponding author
Additional information
Editor: T. J. Jones
Rights and permissions
About this article
Cite this article
Vicuna Requesens, D., Egelkrout, E., Devaiah, S. et al. A method for transient expression in maize endosperm. In Vitro Cell.Dev.Biol.-Plant 46, 485–490 (2010). https://doi.org/10.1007/s11627-010-9305-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11627-010-9305-8