Abstract
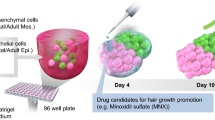
Access to complex in vitro models that recapitulate the unique markers and cell-cell interactions of the hair follicle is rather limited. Creation of scalable, affordable, and relevant in vitro systems which can provide predictive screens of cosmetic ingredients and therapeutic actives for hair health would be highly valued. In this study, we explore the features of the microfollicle, a human hair follicle organoid model based on the spatio-temporally defined co-culture of primary cells. The microfollicle provides a 3D differentiation platform for outer root sheath keratinocytes, dermal papilla fibroblasts, and melanocytes, via epidermal-mesenchymal-neuroectodermal cross-talk. For assay applications, microfollicle cultures were adapted to 96-well plates suitable for medium-throughput testing up to 21 days, and characterized for their spatial and lineage markers. The microfollicles showed hair-specific keratin expression in both early and late stages of cultivation. The gene expression profile of microfollicles was also compared with human clinical biopsy samples in response to the benchmark hair-growth compound, minoxidil. The gene expression changes in microfollicles showed up to 75% overlap with the corresponding gene expression signature observed in the clinical study. Based on our results, the cultivation of the microfollicle appears to be a practical tool for generating testable insights for hair follicle development and offers a complex model for pre-clinical substance testing.






Similar content being viewed by others
References
Aasen T, Izpisua Belmonte JC (2010) Isolation and cultivation of human keratinocytes from skin or plucked hair for the generation of induced pluripotent stem cells. Nat Protoc 5:371–382
Abaci HE, Coffman A, Doucet Y, Chen J, Jackow J, Wang E, Guo Z, Shin JU, Jahoda CA, Christiano AM (2018) Tissue engineering of human hair follicles using a biomimetic developmental approach. Nat Commun 9:5301
Al-Nuaimi Y, Baier G, Watson REB, Chuong C-M, Paus R (2010) The cycling hair follicle as an ideal systems biology research model. Exp Dermatol 19:707–713
Cardona A, Saalfeld S, Schindelin J, Arganda-Carreras I, Preibisch S, Longair M, Tomancak P, Hartenstein V, Douglas RJ (2012) TrakEM2 software for neural circuit reconstruction. PLoS One 7:e38011
Balana ME, Alvarez Roger C, Dugour AV, Kerner NA (2004) Antiandrogen oligonucleotides: active principles in hair- and skin-derived culture cells. J Drugs Dermatol 3:287–294
Casale C, Imparato G, Urciuolo F, Netti PA (2016) Endogenous human skin equivalent promotes in vitro morphogenesis of follicle-like structures. Biomaterials 101:86–95
D'Mello SA, Finlay GJ, Baguley BC, Askarian-Amiri ME (2016) Signaling pathways in melanogenesis. Int J Mol Sci 17:1144
Elliott K, Messenger AG, Stephenson TJ (1999) Differences in hair follicle dermal papilla volume are due to extracellular matrix volume and cell number: implications for the control of hair follicle size and androgen responses. J Investig Dermatol 113:873–877
Huh S, Lee J, Jung E, Kim SC, Kang JI, Lee J, Kim YW, Sung YK, Kang HK, Park D (2009) A cell-based system for screening hair growth-promoting agents. Arch Dermatol Res 301:381–385
Iida M, Ihara S, Matsuzaki T (2007) Hair cycle-dependent changes of alkaline phosphatase activity in the mesenchyme and epithelium in mouse vibrissal follicles. Develop Growth Differ 49:185–195
Korosec A, Lichtenberger BM (2018) 12 - in vitro models to study hair follicle generation. In: in, Skin Tissue Models. Academic Press, Boston
Kwack MH, Kang BM, Kim MK, Kim JC, Sung YK (2011) Minoxidil activates beta-catenin pathway in human dermal papilla cells: a possible explanation for its anagen prolongation effect. J Dermatol Sci 62:154–159
Lachgar S, Charveron M, Gall Y, Bonafe JL (1998) Minoxidil upregulates the expression of vascular endothelial growth factor in human hair dermal papilla cells. Br J Dermatol 138:407–411
Langan EA, Philpott MP, Kloepper JE, Paus R (2015) Human hair follicle organ culture: theory, application and perspectives. Exp Dermatol 24:903–911
Langbein L, Schweizer J (2005) Keratins of the human hair follicle. Int Rev Cytol 243:1–78
Limat A, Breitkreutz D, Stark HJ, Hunziker T, Thikoetter G, Noser F, Fusenig NE (1991) Experimental modulation of the differentiated phenotype of keratinocytes from epidermis and hair follicle outer root sheath and matrix cells. Ann N Y Acad Sci 642:125–146 discussion 46-7
Lindner G, Horland R, Wagner I, Atac B, Lauster R (2011) De novo formation and ultra-structural characterization of a fiber-producing human hair follicle equivalent in vitro. J Biotechnol 152:108–112
Lindner G, Lauster R (2009) Methods for producing hair microfollicles and de novo papillae and their use for in vitro tests and in vivo implantations. In.
Magerl M, Kauser S, Paus R, Tobin DJ (2002) Simple and rapid method to isolate and culture follicular papillae from human scalp hair follicles. Exp Dermatol 11:381–385
Malgouries S, Thibaut S, Bernard BA (2008) Proteoglycan expression patterns in human hair follicle. Br J Dermatol 158:234–242
Maschmeyer I, Lorenz AK, Schimek K, Hasenberg T, Ramme AP, Hubner J, Lindner M, Drewell C, Bauer S, Thomas A, Sambo NS, Sonntag F, Lauster R, Marx U (2015) A four-organ-chip for interconnected long-term co-culture of human intestine, liver, skin and kidney equivalents. Lab Chip 15:2688–2699
Messenger AG, Rundegren J (2004) Minoxidil: mechanisms of action on hair growth. Br J Dermatol 150:186–194
Miranda BH, Charlesworth MR, Tobin DJ, Sharpe DT, Randall VA (2018) Androgens trigger different growth responses in genetically identical human hair follicles in organ culture that reflect their epigenetic diversity in life. FASEB J 32:795–806
Moll R, Divo M, Langbein L (2008) The human keratins: biology and pathology. Histochem Cell Biol 129:705–733
Ohyama M, Veraitch O (2013) Strategies to enhance epithelial-mesenchymal interactions for human hair follicle bioengineering. J Dermatol Sci 70:78–87
Ojeh N, Akgül B, Tomic-Canic M, Philpott M, Navsaria H (2017) In vitro skin models to study epithelial regeneration from the hair follicle. PLoS One 12:e0174389
Philpott MP (2018) Culture of the human pilosebaceous unit, hair follicle and sebaceous gland. Exp Dermatol 27:571–577
Plowman JE, Harland DP, Deb-Choudhry S (2018) The hair fibre: proteins, structure and development. Springer, Singapore
Randall VA, Sundberg JP, Philpott MP (2003) Animal and in vitro models for the study of hair follicles. J Investig Dermatol Symp Proc 8:39–45
Rompolas P, Greco V (2014) Stem cell dynamics in the hair follicle niche. Semin Cell Dev Biol 25-26:34–42
Schimek K, Hsu HH, Boehme M, Kornet JJ, Marx U, Lauster R, Portner R, Lindner G (2018) Bioengineering of a full-thickness skin equivalent in a 96-well insert format for substance permeation studies and organ-on-a-chip applications. Bioengineering (Basel) 5:43
Schindelin J, Arganda-Carreras I, Frise E, Kaynig V, Longair M, Pietzsch T, Preibisch S, Rueden C, Saalfeld S, Schmid B, Tinevez JY, White DJ, Hartenstein V, Eliceiri K, Tomancak P, Cardona A (2012) Fiji: an open-source platform for biological-image analysis. Nat Methods 9:676–682
Schneider MR, Schmidt-Ullrich R, Paus R (2009) The hair follicle as a dynamic miniorgan. Curr Biol 19:R132–RR42
Schweizer J, Langbein L, Rogers MA, Winter H (2007) Hair follicle-specific keratins and their diseases. Exp Cell Res 313:2010–2020
Steinert PM, Parry DA, Marekov LN (2003) Trichohyalin mechanically strengthens the hair follicle: multiple cross-bridging roles in the inner root shealth. J Biol Chem 278:41409–41419
Suloway C, Pulokas J, Fellmann D, Cheng A, Guerra F, Quispe J, Stagg S, Potter CS, Carragher B (2005) Automated molecular microscopy: the new Leginon system. J Struct Biol 151:41–60
Tief K, Hahne M, Schmidt A, Beermann F (1996) Tyrosinase, the key enzyme in melanin synthesis, is expressed in murine brain. Eur J Biochem 241:12–16
Upton JH, Hannen RF, Bahta AW, Farjo N, Farjo B, Philpott MP (2015) Oxidative stress-associated senescence in dermal papilla cells of men with androgenetic alopecia. J Invest Dermatol 135:1244–1252
Westgate GE, Craggs RI, Gibson WT (1991a) Immune privilege in hair growth. J Invest Dermatol 97:417–420
Westgate GE, Messenger AG, Watson LP, Gibson WT (1991b) Distribution of proteoglycans during the hair growth cycle in human skin. J Invest Dermatol 96:191–195
Yoo HG, Chang IY, Pyo HK, Kang YJ, Lee SH, Kwon OS, Cho KH, Eun HC, Kim KH (2007) The additive effects of minoxidil and retinol on human hair growth in vitro. Biol Pharm Bull 30:21–26
Acknowledgments
We would like to thank Mike Davis, Xingtao Wei, Rachel Adams, and Jay Tiesman for their valuable help with the microarray analysis. We would also like to thank Chris Kelling, Rudi Lurz, Reyk Horland, and Sarah Tremmel for their valuable contributions in this work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The technology for generating MFs is the subject of a patent from the Technische Universität Berlin that includes G.L. and R.L. as inventors. U.M., G.L., and R.L. are shareholders of TissUse GmbH, and U.M. is CSO of TissUse GmbH, Germany, which is commercializing the MFs. All other authors declare no competing interests. T.D., S.Y., and P.H. are employees of The Procter & Gamble Company.
Additional information
Editor: Tetsuji Okamoto
Supplementary Information
ESM 1
Viability of Microfollicles after Minoxidil (Mx) treatment. Microfollicle viability was measured via fluoresence using PrestoBlue assay at day 4 (D4) before Mx application and every 48 hours following day 8 (D8) during Mx treatment. Data are shown as mean ±SD. The mean comparison was performed using unpaired t-test with Welch’s correction. Statistical significance is shown for corresponding VC versus 1% Mx application with (**) when p<0.01 and (***) when p<0.0001. UTC-Untreated Control, VC-Vehicle Control, 1% Mx-1% minoxidil (n=5-6 MF per timepoint). MF metabolism during minoxidil treatment was examined in 2 donors with similar results. Donor used for microarray experiment is shown. (PNG 36 kb)
ESM 2
Primary antibodies (in 1:100 dilution) and secondary antibodies (in 1:200 dilution) used in immunofluorescence stainings are listed. (PNG 116 kb)
ESM 3
P values < 0.05 are highlighted in red, positive values are highlighted in yellow and negative values are highlighted in green. (XLSX 83 kb)
ESM 4
(MP4 4481 kb)
Rights and permissions
About this article
Cite this article
Ataç, B., Kiss, F.M., Lam, T. et al. The microfollicle: a model of the human hair follicle for in vitro studies. In Vitro Cell.Dev.Biol.-Animal 56, 847–858 (2020). https://doi.org/10.1007/s11626-020-00513-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11626-020-00513-x