Abstract
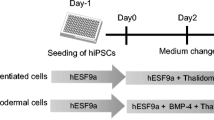
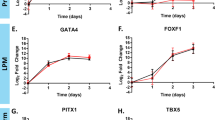
Thalidomide was once administered to pregnant women as a mild sedative; however, it was subsequently shown to be strongly teratogenic. Recently, there has been renewed interest in thalidomide because of its curative effects against intractable diseases. However, the teratogenicity of thalidomide is manifested in various ways and is still not fully understood. In the present study, we evaluated the effects of thalidomide on early mesodermal differentiation by examining the differentiation of human induced pluripotent stem cells (hiPSCs). The most common symptom of thalidomide teratogenicity is limb abnormality, which led us to hypothesize that thalidomide prevents early mesodermal differentiation. Therefore, mesodermal differentiation of hiPSCs was induced over a 6-d period. To induce early mesoderm differentiation, 1 d after seeding, the cells were incubated with the small molecule compound CHIR99021 for 3 d. Thalidomide exposure was initiated at the same time as CHIR99021 treatment. After 5 d of thalidomide exposure, the hiPSCs began expressing a mesodermal marker; however, the number of viable cells decreased significantly as compared to that of control cells. We observed that the proportion of apoptotic and dead cells increased on day 2; however, the proportion of dead cells on day 5 had decreased, suggesting that the cells were damaged by thalidomide during early mesodermal differentiation (days 0–2). Our findings may help elucidate the mechanism underlying thalidomide teratogenicity and bring us closer to the safe use of this drug.








Similar content being viewed by others
References
Aikawa N, Kunisato A, Nagao K, Kusaka H, Takaba K, Ohgami K (2014) Detection of thalidomide embryotoxicity by in vitro embryotoxicity testing based on human iPS cells. J Pharm Sci 124(2):201–207. https://doi.org/10.1254/jphs.13162FP
D'Amato RJ, Loughnan MS, Flynn E, Folkman J (1994) Thalidomide is an inhibitor of angiogenesis. Proc Natl Acad Sci U S A 91(9):4082–4085. https://doi.org/10.1073/pnas.91.9.4082
Franks ME, Macpherson GR, Figg WD (2004) Thalidomide. Lancet 363(9423):1802–1811. https://doi.org/10.1016/S0140-6736(04)16308-3
Fratta ID, Sigg EB, Maiorana K (1965) Teratogenic effects of thalidomide in rabbits, rats, hamsters and mice. Toxicol Appl Pharmacol 7(2):268–286. https://doi.org/10.1016/0041-008X(65)90095-5
Furue MK, Na J, Jackson JP, Okamoto T, Jones M, Baker D, Hata R, Moore HD, Sato JD, Andrews PW (2008) Heparin promotes the growth of human embryonic stem cells in a defined serum-free medium. Proc Natl Acad Sci U S A 105(36):13409–13414. https://doi.org/10.1073/pnas.0806136105
Guo CW, Kawakatsu M, Idemitsu M, Urata Y, Goto S, Ono Y, Hamano K, Li TS (2013) Culture under low physiological oxygen conditions improves the stemness and quality of induced pluripotent stem cells. J Cell Physiol 228(11):2159–2166. https://doi.org/10.1002/jcp.24389
Hayashi Y, Chan T, Warashina M, Fukuda M, Ariizumi T, Okabayashi K, Takayama N, Otsu M, Eto K, Furue MK, Michiue T, Ohnuma K, Nakauchi H, Asashima M (2010) Reduction of N-glycolylneuraminic acid in human induced pluripotent stem cells generated or cultured under feeder- and serum-free defined conditions. PLoS One 5(11):e14099. https://doi.org/10.1371/journal.pone.0014099
Ito T, Ando H, Suzuki T, Ogura T, Hotta K, Imamura Y, Yamaguchi Y, Handa H (2010) Identification of a primary target of thalidomide teratogenicity. Science 327(5971):1345–1350. https://doi.org/10.1126/science.1177319
Kameoka S, Babiarz J, Kolaja K, Chiao E (2014) A high-throughput screen for teratogens using human pluripotent stem cells. Toxicol Sci 137(1):76–90. https://doi.org/10.1093/toxsci/kft239
Moore KL, Torchia MG (2015) The developing human, 10th edition Clinically Oriented Embryology. Elsevier
Knobloch J, Jungck D, Koch A (2011) Apoptosis induction by thalidomide: critical for limb teratogenicity but therapeutic potential in idiopathic pulmonary fibrosis? Curr Mol Pharmacol 4:26–61
Knobloch J, Ruther U (2008) Shedding light on an old mystery: thalidomide suppresses survival pathways to induce limb defects. Cell Cycle 7(9):1121–1127. https://doi.org/10.4161/cc.7.9.5793
Knobloch J, Shaughnessy JD Jr, Ruther U (2007) Thalidomide induces limb deformities by perturbing the Bmp/Dkk1/Wnt signaling pathway. FASEB J 21(7):1410–1421. https://doi.org/10.1096/fj.06-7603com
Kumagai A, Suga M, Yanagihara K, Itoh Y, Takemori H, Furue MK (2016) A simple method for labeling human embryonic stem cells destined to lose undifferentiated potency. Stem Cells Transl Med 5(3):275–281. https://doi.org/10.5966/sctm.2015-0145
Kunisada Y, Tsubooka-Yamazoe N, Shoji M, Hosoya M (2012) Small molecules induce efficient differentiation into insulin-producing cells from human induced pluripotent stem cells. Stem Cell Res 8(2):274–284. https://doi.org/10.1016/j.scr.2011.10.002
Lenz W (1962) Thalidomide and congenital abnormalities. Lancet 1:45
McBride WG (1961) Thalidomide and congenital abnormalities. Lancet 2:1358
Melchert M, List A (2007) The thalidomide saga. Int J Biochem Cell Biol 39(7-8):1489–1499. https://doi.org/10.1016/j.biocel.2007.01.022
Mellin GW, Katzenstein M (1962a) The saga of thalidomide. Neuropathy to embryopathy, with case reports of congenital anomalies. N Engl J Med 267(23):1184–1192. https://doi.org/10.1056/NEJM196212062672305
Mellin GW, Katzenstein M (1962b) The saga of thalidomide: neuropathy to embryopathy, with case reports of congenital anomalies. N Engl J Med 267:1238–1244
Miller MT, Stromland K (1999) Teratogen update: thalidomide: a review, with a focus on ocular findings and new potential uses. Teratology 60(5):306–321. https://doi.org/10.1002/(SICI)1096-9926(199911)60:5<306::AID-TERA11>3.0.CO;2-Y
Nichols J, Smith A (2009) Naive and primed pluripotent states. Cell Stem Cell 4(6):487–492. https://doi.org/10.1016/j.stem.2009.05.015
Ninomiya H, Mizuno K, Terada R, Miura T, Ohnuma K, Takahashi S, Asashima M, Michiue T (2015) Improved efficiency of definitive endoderm induction from human induced pluripotent stem cells in feeder and serum-free culture system. In Vitro Cell Dev Biol Anim 51(1):1–8. https://doi.org/10.1007/s11626-014-9801-y
Nowack E (1965) The sensitive phase in thalidomide embryopathy. Humangenetik 1(6):516–536. https://doi.org/10.1007/BF00338341
Ohgushi M, Matsumura M, Eiraku M, Murakami K, Aramaki T, Nishiyama A, Muguruma K, Nakano T, Suga H, Ueno M, Ishizaki T, Suemori H, Narumiya S, Niwa H, Sasai Y (2010) Molecular pathway and cell state responsible for dissociation-induced apoptosis in human pluripotent stem cells. Cell Stem Cell 7(2):225–239
Parman T, Wiley MJ, Wells PG (1999) Free radical-mediated oxidative DNA damage in the mechanism of thalidomide teratogenicity. Nat Med 5(5):582–585. https://doi.org/10.1038/8466
Ring DB, Johnson KW, Henriksen EJ, Nuss JM, Goff D, Kinnick TR, Ma ST, Reeder JW, Samuels I, Slabiak T, Wagman AS, Hammond ME, Harrison SD (2003) Selective glycogen synthase kinase 3 inhibitors potentiate insulin activation of glucose transport and utilization in vitro and in vivo. Diabetes 52(3):588–595. https://doi.org/10.2337/diabetes.52.3.588
Schardein JL (2000). Chem induced birth defects, Third Edition 41
Seiler AE, Spielmann H (2011) The validated embryonic stem cell test to predict embryotoxicity in vitro. Nat Protoc 6(7):961–978. https://doi.org/10.1038/nprot.2011.348
Sheskin J (1965) Thalidomide in the treatment of lepra reactions. Clin Pharmacol Ther 6(3):303–306. https://doi.org/10.1002/cpt196563303
Singhal S, Mehta J, Desikan R, Ayers D, Roberson P, Eddlemon P, Munshi N, Anaissie E, Wilson C, Dhodapkar M, Zeddis J, Barlogie B (1999) Antitumor activity of thalidomide in refractory multiple myeloma. N Engl J Med 341(21):1565–1571. https://doi.org/10.1056/NEJM199911183412102
Takahashi K, Tanabe K, Ohnuki M, Narita M, Ichisaka T, Tomoda K, Yamanaka S (2007) Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell 131:861–872
Tachikawa S, Nishimura T, Nakauchi H, Ohnuma K (2017) Thalidomide induces apoptosis in undifferentiated human induced pluripotent stem cells. In Vitro Cell Dev Biol Anim 53(9):841–851. https://doi.org/10.1007/s11626-017-0192-8
Takasato M, Er PX, Chiu HS, Maier B, Baillie GJ, Ferguson C, Parton RG, Wolvetang EJ, Roost MS, Lopes SM, Little MH (2016) Kidney organoids from human iPS cells contain multiple lineages and model human nephrogenesis. Nature 536(7615):238. https://doi.org/10.1038/nature17982
Thomson JA, Itskovitz-Eldor J, Shapiro SS, Waknitz MA, Swiergiel JJ, Marshall VS, Jones JM (1998) Embryonic stem cell lines derived from human blastocysts. Science 282(5391):1145–1147. https://doi.org/10.1126/science.282.5391.1145
Watanabe K, Ueno M, Kamiya D, Nishiyama A, Matsumura M, Wataya T, Takahashi J B, Nishikawa S, Nishikawa S, Muguruma K, Sasai Y (2007) A ROCK inhibitor permits survival of dissociated human embryonic stem cells. Nat Biotechnol 25(6):681–686.
Yoshida Y, Takahashi K, Okita K, Ichisaka T, Yamanaka S (2009) Hypoxia enhances the generation of induced pluripotent stem cells. Cell Stem Cell 5:237–241
Yu J, Vodyanik MA, Smuga-Otto K, Antosiewicz-Bourget J, Frane JL, Tian S, Nie J, Jonsdottir GA, Ruotti V, Stewart R, Slukvin II, Thomson JA (2007) Induced pluripotent stem cell lines derived from human somatic cells. Science 318(5858):1917–1920. https://doi.org/10.1126/science.1151526
Funding
This work was supported in part by grants from the Ministry of Health, Labor, and Welfare of Japan, the Japan Agency for Medical Research and Development (AMED) (to K.O.), and the Foundation for Applied Research and Technological Uniqueness at Nagaoka University of Technology (to S.T.). The funding bodies had no role in the study design, data collection and analysis, the decision to publish, or manuscript preparation.
Author information
Authors and Affiliations
Contributions
S.T. and K.O. designed the project. S.T. performed all experiments. M.S. and K.M. assisted with S.T.’ s experiments. S.T. and K.O. wrote the manuscript, and all authors reviewed the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Editor: Tetsuji Okamoto
Electronic supplementary material
ESM 1
(DOCX 105 kb)
Rights and permissions
About this article
Cite this article
Tachikawa, S., Shimizu, M., Maruyama, K. et al. Thalidomide induces apoptosis during early mesodermal differentiation of human induced pluripotent stem cells. In Vitro Cell.Dev.Biol.-Animal 54, 231–240 (2018). https://doi.org/10.1007/s11626-018-0234-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11626-018-0234-x