Abstract
Insulin-like growth factors (IGFs) are involved in growth and tissue development, including diseases such as type-2 diabetes and cancers. However, their roles in lineage specification, especially in early mammalian neural development, are poorly understood. Here, we analyzed the protein expression of IGF-2 in early mouse embryo, and it was preferentially detected in anterior mesodermal tissue, adjacent to the neural plate. We utilized a self-organizing neural tissue culture system and analyzed the direct effect of IGF-2 on the general neural marker Sox1. Interestingly, using recombinant IGF-2 and a chemical inhibitor of its receptor (IGF-1R), we found that the IGF-2/IGF-1R pathway positively regulated Sox1 expression in embryonic stem (ES) cell-derived neural tissue. Furthermore, to visualize the expression patterns of other neural markers, we used reporter ES cell lines and we found that the IGF-2/IGF-1R signaling upregulated the expression of the posterior neural marker Irx3. In contrast, the anterior neural marker Six3 was downregulated by IGF-2/IGF-1R signaling. Together, our results demonstrate that IGF-2/IGF-1R signaling has different effects on neural marker expression, which may influence the early regional identity of ES cell-derived neural tissues.




Similar content being viewed by others
References
Ajo R, Cacicedo L, Navarro C, Sanchez-Franco F (2003) Growth hormone action on proliferation and differentiation of cerebral cortical cells from fetal rat. Endocrinology 144:1086–1097
Andrabi M, Kuraku S, Takata N, Sasai Y, Love NR (2015) Comparative, transcriptome analysis of self-organizing optic tissues. Sci Data 2:150030
Aubert J, Stavridis MP, Tweedie S, O’Reilly M, Vierlinger K, Li M, Ghazal P, Pratt T, Mason JO, Roy D et al (2003) Screening for mammalian neural genes via fluorescence-activated cell sorter purification of neural precursors from Sox1-gfp knock-in mice. Proc Natl Acad Sci U S A 100(Suppl 1):11836–11841
Beattie J, Allan GJ, Lochrie JD, Flint DJ (2006) Insulin-like growth factor-binding protein-5 (IGFBP-5): a critical member of the IGF axis. Biochem J 395:1–19
Cajal M, Creuzet SE, Papanayotou C, Saberan-Djoneidi D, Chuva de Sousa Lopes SM, Zwijsen A, Collignon J, Camus A (2014) A conserved role for non-neural ectoderm cells in early neural development. Development 141:4127–4138
Cohen DH, LeRoith D (2012) Obesity, type 2 diabetes, and cancer: the insulin and IGF connection. Endocr Relat Cancer 19:F27–45
D’Ercole AJ, Ye P, Calikoglu AS, Gutierrez-Ospina G (1996) The role of the insulin-like growth factors in the central nervous system. Mol Neurobiol 13:227–255
Eiraku M, Takata N, Ishibashi H, Kawada M, Sakakura E, Okuda S, Sekiguchi K, Adachi T, Sasai Y (2011) Self-organizing optic-cup morphogenesis in three-dimensional culture. Nature 472:51–U73
Eivers E, McCarthy K, Glynn C, Nolan CM, Byrnes L (2004) Insulin-like growth factor (IGF) signalling is required for early dorso-anterior development of the zebrafish embryo. Int J Dev Biol 48:1131–1140
Feldman EL, Sullivan KA, Kim B, Russell JW (1997) Insulin-like growth factors regulate neuronal differentiation and survival. Neurobiol Dis 4:201–214
Fernandez AM, Torres-Aleman I (2012) The many faces of insulin-like peptide signalling in the brain. Nat Rev Neurosci 13:225–239
Giovannucci E (2001) Insulin, insulin-like growth factors and colon cancer: a review of the evidence. J Nutr 131:3109S–3120S
Glick RP, Unterman TG, Lacson R (1993) Identification of insulin-like growth factor (IGF) and glucose transporter-1 and -3 mRNA in CNS tumors. Regul Pept 48:251–256
Hartnett L, Glynn C, Nolan CM, Grealy M, Byrnes L (2010) Insulin-like growth factor-2 regulates early neural and cardiovascular system development in zebrafish embryos. Int J Dev Biol 54:573–583
Houweling AC, Dildrop R, Peters T, Mummenhoff J, Moorman AFM, Ruther U, Christoffels VM (2001) Gene and cluster-specific expression of the Iroquois family members during mouse development. Mech Dev 107:169–174
Ikeya M, Kawada M, Nakazawa Y, Sakuragi M, Sasai N, Ueno M, Kiyonari H, Nakao K, Sasai Y (2005) Gene disruption/knock-in analysis of mONT3: vector construction by employing both in vivo and in vitro recombinations. Int J Dev Biol 49:807–823
Kadoshima T, Sakaguchi H, Nakano T, Soen M, Ando S, Eiraku M, Sasai Y (2013) Self-organization of axial polarity, inside-out layer pattern, and species-specific progenitor dynamics in human ES cell-derived neocortex. Proc Natl Acad Sci U S A 110:20284–20289
Lagutin O, Zhu CC, Furuta Y, Rowitch DH, McMahon AP, Oliver G (2001) Six3 promotes the formation of ectopic optic vesicle-like structures in mouse embryos. Dev Dyn 221:342–349
Lancaster MA, Knoblich JA (2014) Generation of cerebral organoids from human pluripotent stem cells. Nat Protoc 9:2329–2340
Lavenius E, Parrow V, Nanberg E, Pahlman S (1994) Basic FGF and IGF-I promote differentiation of human SH-SY5Y neuroblastoma cells in culture. Growth Factors 10:29–39
Maures T, Chan SJ, Xu B, Sun H, Ding J, Duan CM (2002) Structural, biochemical, and expression analysis of two distinct insulin-like growth factor I receptors and their ligands in zebrafish. Endocrinology 143:1858–1871
Miller, R.A., Christoforou, N., Pevsner, J., McCallion, A.S., and Gearhart, J.D. (2008). Efficient Array-Based Identification of Novel Cardiac Genes through Differentiation of Mouse ESCs. Plos One 3
Morrione A, Romano G, Navarro M, Reiss K, Valentinis B, Dews M, Eves E, Rosner MR, Baserga R (2000) Insulin-like growth factor I receptor signaling in differentiation of neuronal H19-7 cells. Cancer Res 60:2263–2272
Mulvihill MJ, Cooke A, Rosenfeld-Franklin M, Buck E, Foreman K, Landfair D, O’Connor M, Pirritt C, Sun Y, Yao Y et al (2009) Discovery of OSI-906: a selective and orally efficacious dual inhibitor of the IGF-1 receptor and insulin receptor. Future Med Chem 1:1153–1171
Nakano T, Ando S, Takata N, Kawada M, Muguruma K, Sekiguchi K, Saito K, Yonemura S, Eiraku M, Sasai Y (2012) Self-formation of optic cups and storable stratified neural retina from human ESCs. Cell Stem Cell 10:771–785
Nishiguchi S, Wood H, Kondoh H, Lovell-Badge R, Episkopou V (1998) Sox1 directly regulates the gamma-crystallin genes and is essential for lens development in mice. Genes Dev 12:776–781
O’Gorman DB, Weiss J, Hettiaratchi A, Firth SM, Scott CD (2002) Insulin-like growth factor-II/mannose 6-phosphate receptor overexpression reduces growth of choriocarcinoma cells in vitro and in vivo. Endocrinology 143:4287–4294
O’Kusky J, Ye P (2012) Neurodevelopmental effects of insulin-like growth factor signaling. Front Neuroendocrinol 33:230–251
Oka Y, Rozek LM, Czech MP (1985) Direct demonstration of rapid insulin-like growth factor II receptor internalization and recycling in rat adipocytes. Insulin stimulates 125I-insulin-like growth factor II degradation by modulating the IGF-II receptor recycling process. J Biol Chem 260:9435–9442
Pera EM, Ikeda A, Eivers E, De Robertis EM (2003) Integration of IGF, FGF, and anti-BMP signals via Smad1 phosphorylation in neural induction. Genes Dev 17:3023–3028
Pera EM, Wessely O, Li SY, De Robertis EM (2001) Neural and head induction by insulin-like growth factor signals. Dev Cell 1:655–665
Pollak MN, Schernhammer ES, Hankinson SE (2004) Insulin-like growth factors and neoplasia. Nat Rev Cancer 4:505–518
Richard-Parpaillon L, Heligon C, Chesnel F, Boujard D, Philpott A (2002) The IGF pathway regulates head formation by inhibiting Wnt signaling in Xenopus. Dev Biol 244:407–417
Rotwein P, Burgess SK, Milbrandt JD, Krause JE (1988) Differential expression of insulin-like growth factor genes in rat central nervous system. Proc Natl Acad Sci U S A 85:265–269
Russo VC, Gluckman PD, Feldman EL, Werther GA (2005) The insulin-like growth factor system and its pleiotropic functions in brain. Endocr Rev 26:916–943
Sang X, Curran MS, Wood AW (2008) Paracrine insulin-like growth factor signaling influences primordial germ cell migration: in vivo evidence from the zebrafish model. Endocrinology 149:5035–5042
Stewart CE, Rotwein P (1996) Growth, differentiation, and survival: multiple physiological functions for insulin-like growth factors. Physiol Rev 76:1005–1026
Suga H, Kadoshima T, Minaguchi M, Ohgushi M, Soen M, Nakano T, Takata N, Wataya T, Muguruma K, Miyoshi H et al (2011) Self-formation of functional adenohypophysis in three-dimensional culture. Nature 480:57–U215
Taguchi A, White MF (2008) Insulin-like signaling, nutrient homeostasis, and life span. Annu Rev Physiol 70:191–212
Takata, N., Sakakura, E., and Sasai, Y. (2015) Activation of Wnt/ss-catenin signaling in ESC promotes rostral forebrain differentiation in vitro. In Vitro Cell Dev Biol Anim
Torres-Aleman I, Pons S, Arevalo MA (1994) The insulin-like growth factor I system in the rat cerebellum: developmental regulation and role in neuronal survival and differentiation. J Neurosci Res 39:117–126
Vicario-Abejon C, Yusta-Boyo MJ, Fernandez-Moreno C, de Pablo F (2003) Locally born olfactory bulb stem cells proliferate in response to insulin-related factors and require endogenous insulin-like growth factor-I for differentiation into neurons and glia. J Neurosci 23:895–906
Wataya T, Ando S, Muguruma K, Ikeda H, Watanabe K, Eiraku M, Kawada M, Takahashi J, Hashimoto N, Sasai Y (2008) Minimization of exogenous signals in ES cell culture induces rostral hypothalamic differentiation. Proc Natl Acad Sci U S A 105:11796–11801
Wood HB, Episkopou V (1999) Comparative expression of the mouse Sox1, Sox2 and Sox3 genes from pre-gastrulation to early somite stages. Mech Dev 86:197–201
Ye P, D’Ercole AJ (2006) Insulin-like growth factor actions during development of neural stem cells and progenitors in the central nervous system. J Neurosci Res 83:1–6
Acknowledgments
We are grateful to M. Eiraku for the invaluable comments and to the members of the laboratory for the discussion. We also thank M. Kawada for the technical advice on SFEBq and vector construction. This work was supported by grants-in-aid from the Ministry of Education, Culture, Sports, Science, and Technology in Japan (MEXT) (to Y.S.) and the Network Program for Realization of Regenerative Medicine from the Japan Science and Technology Agency (JST) (to Y.S.).
Contributions
N.T. designed the research, N.T. and E.S. performed the experiments, N.T. analyzed the data, N.T. prepared the figures and wrote the paper, and Y.S. supervised the project.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Editor: Tetsuji Okamoto
Dr. Yoshiki Sasai would be corresponding author, if he was alive
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Figure
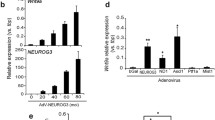
igfbp5 expression in Irx3::Venus+ and Irx3::Venus− cells and distinct effects of IGF-2/IGF-1R signaling on Sox1, Irx3, and Six3 expressions. (A) RT-qPCR analysis of igfbp5 in ES cells and day-5 neuroectoderm-like cells. (B) Schematic diagram of sorting of Irx3::Venus+/− cells via FACS. (C–E) RT-qPCR analysis of igfbp5 in Irx3::Venus+ and Irx3::Venus− cells, detecting mRNA levels of venus, irx3, and igfbp5. gapdh value was used for generating relative expression level of each gene. Error bars indicate standard error of the mean of each experiment. (F) Schematics of IGF-2 effects on Sox1, Irx3, and Six3 expressions. (GIF 31 kb)
Rights and permissions
About this article
Cite this article
Takata, N., Sakakura, E. & Sasai, Y. IGF-2/IGF-1R signaling has distinct effects on Sox1, Irx3, and Six3 expressions during ES cell derived-neuroectoderm development in vitro. In Vitro Cell.Dev.Biol.-Animal 52, 607–615 (2016). https://doi.org/10.1007/s11626-016-0012-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11626-016-0012-6