Abstract
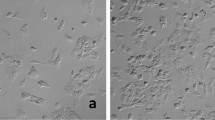
Muscle-invasive bladder cancer is prone to metastasis without a standard organ preference. The current cell lines used to study bladder cancer have primarily been derived from individuals in Western populations, and no human bladder cancer cell line has been established from the Chinese population. A bladder cancer cell line was derived from a female Chinese patient with muscle-invasive bladder cancer, and these cells were then xenografted into the bladders of three nude mice. Five weeks later, these mice were killed to observe local invasion and distant metastasis. The metastatic tumors were also removed and analyzed to assess the metastatic mechanism. This bladder cancer cell line, named T921, was successfully established, as evidenced by karyotype and immunohistochemistry analyses. Multi-organ metastases were observed in all three of the nude mice 5 wk after the orthotopic transfer of the cell line. In addition, epithelial–mesenchymal transition (EMT)-related genes were involved in the tumor metastases. The T921 bladder cancer cell line was successfully established, and EMT was observed to play a role in bladder cancer metastasis.







Similar content being viewed by others
References
Browne G.; Sayan A. E.; Tulchinsky E. ZEB proteins link cell motility with cell cycle control and cell survival in cancer. Cell Cycle 9: 886–891; 2010.
Bubenik J.; Baresova M.; Viklicky V.; Jakoubkova J.; Sainerova H.; Donner J. Established cell line of urinary bladder carcinoma (T24) containing tumour-specific antigen. Int. J. Cancer J. Int. Cancer 11: 765–773; 1973.
Chan K. S.; Espinosa I.; Chao M.; Wong D.; Ailles L.; Diehn M.; Gill H.; Presti Jr. J.; Chang H. Y.; van de Rijn M.; Shortliffe L.; Weissman I. L. Identification, molecular characterization, clinical prognosis, and therapeutic targeting of human bladder tumor-initiating cells. Proc. Natl. Acad. Sci. U. S. A. 106: 14016–14021; 2009.
Chen T.; Wang L. H.; Farrar W. L. Interleukin 6 activates androgen receptor-mediated gene expression through a signal transducer and activator of transcription 3-dependent pathway in LNCaP prostate cancer cells. Cancer Res. 60: 2132–2135; 2000.
Culig Z.; Hobisch A.; Cronauer M. V.; Radmayr C.; Trapman J.; Hittmair A.; Bartsch G.; Klocker H. Androgen receptor activation in prostatic tumor cell lines by insulin-like growth factor-I, keratinocyte growth factor, and epidermal growth factor. Cancer Res. 54: 5474–5478; 1994.
Firulli A. B.; Conway S. J. Phosphoregulation of Twist1 provides a mechanism of cell fate control. Curr. Med. Chem. 15: 2641–2647; 2008.
Fischer O. M.; Hart S.; Gschwind A.; Prenzel N.; Ullrich A. Oxidative and osmotic stress signaling in tumor cells is mediated by ADAM proteases and heparin-binding epidermal growth factor. Mol. Cell. Biol. 24: 5172–5183; 2004.
Hall E. J.; Bedford J. S.; Oliver R. Extreme hypoxia; its effect on the survival of mammalian cells irradiated at high and low dose-rates. Br. J. Radiol. 39: 302–307; 1966.
Hegele A.; Heidenreich A.; Varga Z.; von Knobloch R.; Olbert P.; Kropf J.; Hofmann R. Cellular fibronectin in patients with transitional cell carcinoma of the bladder. Urol. Res. 30: 363–366; 2003.
Ismaili N.; Amzerin M.; Flechon A. Chemotherapy in advanced bladder cancer: current status and future. J. Hematol. Oncol. 4: 35; 2011.
Iwatsuki M.; Mimori K.; Yokobori T.; Ishi H.; Beppu T.; Nakamori S.; Baba H.; Mori M. Epithelial–mesenchymal transition in cancer development and its clinical significance. Cancer Sci. 101: 293–299; 2010.
Jacobs B. L.; Lee C. T.; Montie J. E. Bladder cancer in 2010: how far have we come? CA: Cancer J. Clin. 60:244–272; 2010. 2009. SCR.
Koivisto P. A.; Rantala I. Amplification of the androgen receptor gene is associated with P53 mutation in hormone-refractory recurrent prostate cancer. J. Pathol. 187: 237–241; 1999.
Li J.; Yen C.; Liaw D.; Podsypanina K.; Bose S.; Wang S. I.; Puc J.; Miliaresis C.; Rodgers L.; McCombie R.; Bigner S. H.; Giovanella B. C.; Ittmann M.; Tycko B.; Hibshoosh H.; Wigler M. H.; Parsons R. PTEN, a putative protein tyrosine phosphatase gene mutated in human brain, breast, and prostate cancer. Science 275: 1943–1947; 1997.
Meng A. X.; Jalali F.; Cuddihy A.; Chan N.; Bindra R. S.; Glazer P. M.; Bristow R. G. Hypoxia down-regulates DNA double strand break repair gene expression in prostate cancer cells. Radiother. Oncol.: J. Eur. Soc. Ther. Radiol. Oncol. 76: 168–176; 2005.
Natalwala A.; Spychal R.; Tselepis C. Epithelial–mesenchymal transition mediated tumourigenesis in the gastrointestinal tract. World J. Gastroenterol.: WJG 14: 3792–3797; 2008.
Niu G.; Wright K. L.; Huang M.; Song L.; Haura E.; Turkson J.; Zhang S.; Wang T.; Sinibaldi D.; Coppola D.; Heller R.; Ellis L. M.; Karras J.; Bromberg J.; Pardoll D.; Jove R.; Yu H. Constitutive Stat3 activity up-regulates VEGF expression and tumor angiogenesis. Oncogene 21: 2000–2008; 2002.
Saad A.; Hanbury D. C.; McNicholas T. A.; Boustead G. B.; Morgan S.; Woodman A. C. A study comparing various noninvasive methods of detecting bladder cancer in urine. BJU Int. 89: 369–373; 2002.
Seabright M. A rapid banding technique for human chromosomes. Lancet 2:971–2; 1971.
Shain K. H.; Dalton W. S. Cell adhesion is a key determinant in de novo multidrug resistance (MDR): new targets for the prevention of acquired MDR. Mol. Cancer Ther. 1: 69–78; 2001.
Soloway M. S.; Sofer M.; Vaidya A. Contemporary management of stage T1 transitional cell carcinoma of the bladder. J. Urol. 167: 1573–1583; 2002.
Thiery J. P.; Acloque H.; Huang R. Y.; Nieto M. A. Epithelial–mesenchymal transitions in development and disease. Cell 139: 871–890; 2009.
Wang T.; Hu H. S.; Feng Y. X.; Shi J.; Li N.; Guo W. X.; Xue J.; Xie D.; Liu S. R.; Wu M. C.; Cheng S. Q. Characterisation of a novel cell line (CSQT-2) with high metastatic activity derived from portal vein tumour thrombus of hepatocellular carcinoma. Br. J. Cancer 102: 1618–1626; 2010.
Wei D.; Le X.; Zheng L.; Wang L.; Frey J. A.; Gao A. C.; Peng Z.; Huang S.; Xiong H. Q.; Abbruzzese J. L.; Xie K. Stat3 activation regulates the expression of vascular endothelial growth factor and human pancreatic cancer angiogenesis and metastasis. Oncogene 22: 319–329; 2003.
Wells A.; Yates C.; Shepard C. R. E-cadherin as an indicator of mesenchymal to epithelial reverting transitions during the metastatic seeding of disseminated carcinomas. Clin. Exp. Metastasis 25: 621–628; 2008.
Yan W.; Fu Y.; Tian D.; Liao J.; Liu M.; Wang B.; Xia L.; Zhu Q.; Luo M. PI3 kinase/Akt signaling mediates epithelial–mesenchymal transition in hypoxic hepatocellular carcinoma cells. Biochem. Biophys. Res. Commun. 382: 631–636; 2009.
Yap A. S.; Crampton M. S.; Hardin J. Making and breaking contacts: the cellular biology of cadherin regulation. Curr. Opin. Cell Biol. 19: 508–514; 2007.
Zolzer F.; Streffer C. Increased radiosensitivity with chronic hypoxia in four human tumor cell lines. Int. J. Radiat. Oncol. Biol. Phys. 54: 910–920; 2002.
Acknowledgments
This study was supported by grants from the National Natural Science Foundation of China (No. 81172425).
Author information
Authors and Affiliations
Corresponding author
Additional information
Editor: T. Okamoto
Chuanliang Xu, Weidong Xu and Shancheng Ren contributed equally to this work.
Rights and permissions
About this article
Cite this article
Xu, C., Xu, W., Ren, S. et al. Establishment of a Chinese bladder cancer cell line (T921) with high metastatic activity. In Vitro Cell.Dev.Biol.-Animal 49, 668–678 (2013). https://doi.org/10.1007/s11626-013-9651-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11626-013-9651-z