Abstract
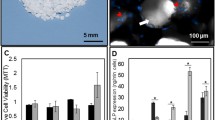
Our laboratory has previously developed scaffoldless engineered bone constructs (EBC). Bone marrow stromal cells (BMSC) were harvested from rat femur and cultured in medium that induced osteogenic differentiation. After reaching confluence, the monolayer of cells contracted around two constraint points forming a cylinder. EBCs were placed in small diameter (0.5905 × 0.0625 in.) or large diameter (0.5905 × 0.125 in.) silicone tubing and implanted intramuscularly in the hind limb of a rat. Bone mineral content (BMC) of the EBC was analyzed before implantation and at 1 and 2 mo following implantation and compared to that of native femur bone at different stages of development. Negligible BMC was observed in E-20 femur or EBCs prior to implantation. One-month implantation in both small and large tubing increased BMC in the EBC. BMC of EBC from large tubing was greater than in 14 d rat neonatal femurs, but was 2% and 3% of BMC content in adult bone after 1 and 2 mo of implantation, respectively. Alizarine Red and osteopontin staining of the EBCs before and after implantation confirmed increased bone mineralization in the implanted EBCs. Implanted EBCs also had extensive vascularization. Our data suggest that BMSC can be successfully used for the generation of scaffoldless EBC, and this model can be potentially used for the generation of autologous bone transplants in humans.








Similar content being viewed by others
References
Akahane M.; Nakamura A.; Ohgushi H. et al. Osteogenic matrix sheet-cell transplantation using osteoblastic cell sheet resulted in bone formation without scaffold at an ectopic site. J. Tissue Eng. Regen. Med. 24: 196–201; 2008.
Augello A.; Tasso R.; Negrini S. M. et al. Bone marrow mesenchymal progenitor cells inhibit lymphocyte proliferation by activation of the programmed death 1 pathway. Eur. J. Immunol. 355: 1482–90; 2005.
Boivin G.; Meunier P. J. The mineralization of bone tissue: a forgotten dimension in osteoporosis research. Osteoporos. Int. 14Suppl 3: S19–24; 2003.
Boivin G.; Bala Y.; Doublier A. et al. The role of mineralization and organic matrix in the microhardness of bone tissue from controls and osteoporotic patients. Bone 43: 532–538; 2008.
Borden M.; Attawia M.; Khan Y. et al. Tissue engineered microsphere-based matrices for bone repair: design and evaluation. Biomaterials 232: 551–9; 2002.
Calve S.; Dennis R. G.; Kosnik P. E. 2nd et al. Engineering of functional tendon. Tissue Eng. 105–6: 755–61; 2004.
Derubeis A. R.; Cancedda R. Bone marrow stromal cells (BMSCs) in bone engineering: limitations and recent advances. Ann. Biomed. Eng. 321: 160–5; 2004.
Follet H.; Boivin G.; Rumelhart C. et al. The degree of mineralization is a determinant of bone strength: a study on human calcanei. Bone 345: 783–9; 2004.
Gitelis S.; Saiz P. What’s new in orthopaedic surgery. J. Am. Coll. Surg. 1946: 788–91; 2002.
Guerkov H. H.; Lohmann C. H.; Liu Y. et al. Pulsed electromagnetic fields increase growth factor release by nonunion cells. Clin. Orthop. Relat. Res. 384: 265–279; 2001.
Hing K. A.; Wilson L. F.; Buckland T. Comparative performance of three ceramic bone graft substitutes. Spine J. 74: 475–90; 2007.
Jaiswal N.; Haynesworth S. E.; Caplan A. I. et al. Osteogenic differentiation of purified, culture-expanded human mesenchymal stem cells in vitro. J. Cell Biochem. 642: 295–312; 1997.
Jiang T.; Abdel-Fattah W. I.; Laurencin C. T. In vitro evaluation of chitosan/poly(lactic acid–glycolic acid) sintered microsphere scaffolds for bone tissue engineering. Biomaterials 2728: 4894–903; 2006.
Khan Y.; Yaszemski M. J.; Mikos A. G. et al. Tissue engineering of bone: material and matrix considerations. J. Bone. Jt. Surg. Am. 90Suppl 1: 36–42; 2008.
Kim I. S.; Song J. K.; Song Y. M. et al. Novel effect of biphasic electric current on in vitro osteogenesis and cytokine production in human mesenchymal stromal cells. Tissue Eng. Part A, 2009 (in press).
Larkin L. M.; Calve S.; Kostrominova T. Y. et al. Structure and functional evaluation of tendon-skeletal muscle constructs engineered in vitro. Tissue Eng. 1211: 3149–58; 2006.
Laurencin C.; Khan Y.; El-Amin S. F. Bone graft substitutes. Expert Rev. Med. Devices 31: 49–57; 2006.
Laurencin C. T.; Ambrosio A. M.; Borden M. D. et al. Tissue engineering: orthopedic applications. Annu. Rev. Biomed. Eng. 1: 19–46; 1999.
Luk J. M.; Wang P. P.; Lee C. K. et al. Hepatic potential of bone marrow stromal cells: development of in vitro co-culture and intra-portal transplantation models. J. Immunol. Methods 3051: 39–47; 2005.
Luna L. G. Masson’s trichrome method. Manual of histologic staining methods of the Armed Forces Institute of Pathology (ed). McGraw-Hill, New York; 1968.
Marks S. C. Jr.; Popoff S. N. Bone cell biology: the regulation of development, structure, and function in the skeleton. Am. J. Anat. 1831: 1–44; 1988.
Mistry A. S.; Mikos A. G. Tissue engineering strategies for bone regeneration. Adv. Biochem. Eng. Biotechnol. 94: 1–22; 2005.
Nair L. S.; Laurencin C. T. Polymers as biomaterials for tissue engineering and controlled drug delivery. Adv. Biochem. Eng. Biotechnol. 102: 47–90; 2006.
Ohgushi H.; Caplan A. I. Stem cell technology and bioceramics: from cell to gene engineering. J. Biomed. Mater. Res. 486: 913–27; 1999.
Perry C. R. Bone repair techniques, bone graft, and bone graft substitutes. Clin. Orthop. Relat. Res. 360: 71–86; 1999.
Scutt A.; Bertram P. Basic fibroblast growth factor in the presence of dexamethasone stimulates colony formation, expansion, and osteoblastic differentiation by rat bone marrow stromal cells. Calcif. Tissue Int. 641: 69–77; 1999.
Stevens B.; Yang Y.; Mohandas A. et al. A review of materials, fabrication methods, and strategies used to enhance bone regeneration in engineered bone tissues. J. Biomed. Mater. Res. B. Appl. Biomater. 852: 573–82; 2008.
Stevenson K. T.; van Tets I. G. Dual-energy X-ray absorptiometry (DXA) can accurately and nondestructively measure the body composition of small, free-living rodents. Physiol. Biochem. Zool. 813: 373–82; 2008.
Syed-Picard F.; Larkin L. M.; Shaw C. M. et al. Three-dimensional engineered bone from bone marrow stromal cells and their autogenous extracellular matrix. Tissue Engineering 151: 187–195; 2009.
Wang Q.; Zhong S.; Ouyang J. et al. Osteogenesis of electrically stimulated bone cells mediated in part by calcium ions. Clin. Orthop. Relat. Res. 348: 259–268; 1998.
Zhang K.; Ma Y.; Francis L. F. Porous polymer/bioactive glass composites for soft-to-hard tissue interfaces. J. Biomed. Mater. Res. 614: 551–63; 2002.
Zhou Y.; Chen F.; Ho S. T. et al. Combined marrow stromal cell-sheet techniques and high-strength biodegradable composite scaffolds for engineered functional bone grafts. Biomaterials 285: 814–24; 2007.
Acknowledgments
This research was supported by DARPA Biomolecular Motors Program, US Air Force, AFOSR award number FA9550-05-1-0015, and by NSF, Civil and Mechanical Systems grant no. CMS9988693.
Author information
Authors and Affiliations
Corresponding author
Additional information
Editor: J. Denry Sato
An erratum to this article can be found at http://dx.doi.org/10.1007/s11626-009-9251-0
Rights and permissions
About this article
Cite this article
Smietana, M.J., Syed-Picard, F.N., Ma, J. et al. The effect of implantation on scaffoldless three-dimensional engineered bone constructs. In Vitro Cell.Dev.Biol.-Animal 45, 512–522 (2009). https://doi.org/10.1007/s11626-009-9216-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11626-009-9216-3