ABSTRACT
Background
Unfavorable results of major studies have led to a large shrinkage of the market for hormone replacement therapy (HRT) in the last 6 years. Some scientists continue to strongly support the use of HRT.
Objectives
We analyzed a sample of partisan editorializing articles on HRT to examine their arguments, the reporting of competing interests, the journal venues and their sponsoring societies.
Data Sources
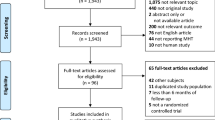
Through Thomson ISI database, we selected articles without primary data written by the five most prolific editorialists that addressed clinical topics pertaining to HRT and that were published in regular journal issues in 2002–2008.
Main Measures
We recorded the number of articles with a partisan stance and their arguments, the number of partisan articles that reported conflicting interests, and the journal venues and their sponsoring societies publishing the partisan editorials.
Key Results
We analyzed 114 eligible articles (58 editorials, 16 guidelines, 37 reviews, 3 letters), of which 110 (96%) had a partisan stance favoring HRT. Typical arguments were benefits for menopausal and related symptoms (64.9%), criticism of unfavorable studies (78.9%), preclinical data that showed favorable effects of HRT (50%), and benefits for major outcomes such as osteoporosis and fractures (49.1%), cardiovascular disease (31.6%), dementia (24.6%) or colorectal cancer (20.2%), but also even breast cancer (4.4%). All 5 prolific editorialists had financial relationships with hormone manufacturers, but these were reported in only 6 of the 110 partisan articles. Four journals published 15–37 partisan articles each. The medical societies of these journals reported on their websites that several pharmaceutical companies sponsored them or their conferences.
Conclusions
There is a considerable body of editorializing articles favoring HRT use and very few of these articles report conflicts of interest. Full disclosure of conflicts of interest is needed, especially for articles without primary data.

Similar content being viewed by others
REFERENCES
Wilson D. Wyeth’s Use of Medical Ghostwriters Questioned. New York Times. December 13, 2008:B1 (Accessed on March 2010 at http://www.nytimes.com/2008/12/13/business/13wyeth.html?_r=1&ref=business)
Rossouw JE, Anderson GL, Prentice RL, et al. Writing Group for the Women’s Health Initiative Investigators. Risks and benefits of estrogen plus progestin in healthy postmenopausal women: principal results From the Women’s Health Initiative randomized controlled trial. JAMA. 2002;288:321–33.
Million Women Study Collaborators. Breast cancer and hormone-replacement therapy in the Million Women Study. Lancet. 2003;362:419–27.
Hersh AL, Stefanick ML, Stafford RS. National use of postmenopausal hormone therapy: annual trends and response to recent evidence. JAMA. 2004;291:47–53.
Watson J, Wise L, Green J. Prescribing of hormone therapy for menopause, tibolone, and bisphosphonates in women in the UK between 1991 and 2005. Eur J Clin Pharmacol. 2007;63:843–9.
ACOG Task Force for Hormone Therapy American College of Obstetricians and Gynecologists Women’s Health Care Physicians. Summary of balancing risks and benefits. Obstet Gynecol. 2004;104:128S–129S.
Mosca L, Appel LJ, Benjamin EJ, et al. Evidence-based guidelines for cardiovascular disease prevention in women. Circulation. 2004;109:672–93.
Recommendations for estrogen and progestogen use in peri-and postmenopausal women: October 2004 position statement of The North American Menopause Society. Menopause. 2004;11:589–600.
Wathen CN, Feig DS, Feightner JW, Abramson BL, Cheung AM. Hormone replacement therapy for the primary prevention of chronic diseases: recommendation statement from the Canadian Task Force on Preventive Health Care. CMAJ. 2004;170:1535–7.
Tatsioni A, Bonitsis NG, Ioannidis JP. Persistence of contradicted claims in the literature. JAMA. 2007;298:2517–26.
Siontis GC, Tatsioni A, Katritsis DG, Ioannidis JP. Persistent reservations against contradicted percutaneous coronary intervention indications: citation content analysis. Am Heart J. 2009;157:695–701.
Angell M. Drug companies. New York: Oxford University Press; 2004:251.
Rothman DJ, McDonald WJ, Berkowitz CD, et al. Professional medical associations and their relationships with industry: a proposal for controlling conflict of interest. JAMA. 2009;301:1367–72.
Sniderman AD, Furberg CD. Why guidelinemaking requires reform. JAMA. 2009;301:429–31.
Steinbrook R. Financial support of continuing medical education. JAMA. 2008;299:1060–2.
Rothman DJ. Academic medical centers and financial conflicts of interest. JAMA. 2008;299:695–7.
Ehringhaus SH, Weissman JS, Sears JL, Goold SD, Feibelmann S, Campbell EG. Responses of medical schools to institutional conflicts of interest. JAMA. 2008;299:665–71.
Podolsky SH, Greene JA. A historical perspective of pharmaceutical promotion and physician education. JAMA. 2008;300:831–3.
Campbell EG, Gruen RL, Mountford J, Miller LG, Cleary PD, Blumenthal D. A national survey of physician-industry relationships. N Engl J Med. 2007;356:1742–50.
Relman AS. Medical professionalism in a commercialized health care market. JAMA. 2007;298:2668–70.
Brennan TA, Rothman DJ, Blank L, et al. Health industry practices that create conflicts of interest: a policy proposal for academic medical centers. JAMA. 2006;295:429–33.
Peppercorn J, Blood E, Winer E, Partridge A. Association between pharmaceutical involvement and outcomes in breast cancer clinical trials. Cancer. 2007;109:1239–46.
Vlad SC, LaValley MP, McAlindon TE, Felson DT. Glucosamine for pain in osteoarthritis: why do trial results differ? Arthritis Rheum. 2007;56:2267–77.
Ioannidis JP. Why most published research findings are false. PLoS Med. 2005;2:e124.
Ioannidis JP. Effectiveness of antidepressants: an evidence myth constructed from a thousand randomized trials? Philos Ethics Humanit Med. 2008;3:14.
Stelfox HT, Chua G, O’Rourke K, Detsky AS. Conflict of interest in the debate over calcium-channel antagonists. N Engl J Med. 1998;338:101–6.
Choudhry NK, Stelfox HT, Detsky AS. Relationships between authors of clinical practice guidelines and the pharmaceutical industry. JAMA. 2002;287:612–7.
Angell M, Kassirer JP. Editorials and conflicts of interest. N Engl J Med. 1996;335:1055–6.
Kassirer JP, Angell M. Financial conflicts of interest in biomedical research. N Engl J Med. 1993;329:570–1.
Steinman MA, Bero LA, Chren MM, Landefeld CS. Narrative review: the promotion of gabapentin: an analysis of internal industry documents. Ann Intern Med. 2006;145:284–93.
Healy D, Cattell D. Interface between authorship, industry and science in the domain of therapeutics. Br J Psychiatry. 2003;183:22–7.
Ross JS, Hill KP, Egilman DS, Krumholz HM. Guest authorship and ghostwriting in publications related to rofecoxib: a case study of industry documents from rofecoxib litigation. JAMA. 2008;299:1800–12.
Gøtzsche PC, Hróbjartsson A, Johansen HK, Haahr MT, Altman DG, Chan AW. Ghost authorship in industry-initiated randomised trials. PLoS Med. 2007;4:e19.
Conflict of Interest in Medical Research, Education and Practice. Bernard Lo and Marilyn J. Eds. Report prepared by the Committee on Conflict of Interest in Medical Research, Education, and Practice. Washington, DC: Institute of Medicine. National Academy Press; April 28, 2009.
Author Contributions
Dr. Ioannidis had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Funding/Support
None
Conflict of Interest
None disclosed.
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 51 kb)
Rights and permissions
About this article
Cite this article
Tatsioni, A., Siontis, G.C.M. & Ioannidis, J.P.A. Partisan Perspectives in the Medical Literature: A Study of High Frequency Editorialists Favoring Hormone Replacement Therapy. J GEN INTERN MED 25, 914–919 (2010). https://doi.org/10.1007/s11606-010-1360-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11606-010-1360-7