Abstract
Background
Electronic medical records (EMRs) have the potential to facilitate the design of large cluster-randomized trials (CRTs).
Objective
To describe the design of a CRT of clinical decision support to improve diabetes care and outcomes.
Methods
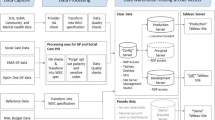
In the Diabetes Improvement Group-Intervention Trial (DIG-IT), we identified and balanced preassignment characteristics of 12,675 diabetic patients cared for by 147 physicians in 24 practices of 2 systems using the same vendor’s EMR. EMR-facilitated disease management was system A’s experimental intervention; system B interventions involved patient empowerment, with or without disease management. For our sample, we: (1) identified characteristics associated with response to interventions or outcomes; (2) summarized feasible partitions of 10 system A practices (2 groups) and 14 system B practices (3 groups) using intra-cluster correlation coefficients (ICCs) and standardized differences; (3) selected (blinded) partitions to effectively balance the characteristics; and (4) randomly assigned groups of practices to interventions.
Results
In System A, 4,306 patients, were assigned to 2 groups of practices; 8,369 patients in system B were assigned to 3 groups of practices. Nearly all baseline outcome variables and covariates were well-balanced, including several not included in the initial design. DIG-IT’s balance was superior to alternative partitions based on volume, geography or demographics alone.
Conclusions
EMRs facilitated rigorous CRT design by identifying large numbers of patients with diabetes and enabling fair comparisons through preassignment balancing of practice sites. Our methods can be replicated in other settings and for other conditions, enhancing the power of other translational investigations.



Similar content being viewed by others
References
Centers for Disease Control and Prevention. National Diabetes fact sheet: general information and national estimates on diabetes in the United States, 2003 Rev. ed. Atlanta: U.S. Department of Health and Human Services, Centers for Disease Control and Prevention; 2004.
Saaddine JB, Cadwell B, Gregg EW, Engelgau MM, Vinicor F, Imperatore G, et al. Improvements in diabetes processes of care and intermediate outcomes: United States, 1988–2002. Ann Intern Med.. 2006;144(7):465–74, Apr 4.
Shojania KG, Ranji S, McDonald KM, et al. Effects of quality improvement strategies for type 2 diabetes on glycemic control: a meta-regression analysis. JAMA. 2006;296(4):427–40, Jul 26.
Meigs JB, Cagliero E, Dubey A, et al. A controlled trial of web-based diabetes disease management: the MGH diabetes primary care improvement project. Diabetes Care. 2003;26(3):750–7, Mar.
O’Connor PJ, Crain AL, Rush WA, Sperl-Hillen JM, Gutenkauf JJ, Duncan JE. Impact of an electronic medical record on diabetes quality of care. Ann Fam Med. 2005;3(4):300–6, Jul–Aug.
Selby JV, Karter AJ, Ackerson LM, Ferrara A, Liu J. Developing a prediction rule from automated clinical databases to identify high-risk patients in a large population with diabetes. Diabetes Care. 2001;24(9):1547–55, Sep.
Murray D. Design and Analysis of Group-Randomized Trials. New York: Oxford University Press; 1998.
Donner A, Klar N. Design and Analysis of Cluster Randomization Trials in Health Research. London: Arnold; 2000.
Eldridge SM, Ashby D, Feder GS, Rudnicka A, Ukoumunne OC. Lessons for cluster randomized trials in the twenty-first century: a systematic review of trials in primary care. Clin Trials. 2004;1(1):80–90, Feb.
Simpson JM, Klar N, Donnor A. Accounting for cluster randomization: a review of primary prevention trials, 1990 through 1993. Am J Public Health. 1995;85(10):1378–83, Oct.
Osheroff JA, Teich JM, Middleton BF, Steen EB, Wright A, Detmer DE. A Roadmap for National Action on Clinical Decision Support. J Am Med Inform Assoc. 2007;14:141–5.
Eccles M, McColl E, Steen N, et al. Effect of computerised evidence based guidelines on management of asthma and angina in adults in primary care: cluster randomised controlled trial. BMJ. 2002;325(7370):941, Oct 26.
Campbell MK, Grimshaw JM, Elbourne DR. Intracluster correlation coefficients in cluster randomized trials: empirical insights into how should they be reported. BMC Med Res Methodol. 2004;4:9, Apr 28.
Campbell MK, Fayers PM, Grimshaw JM. Determinants of the intracluster correlation coefficient in cluster randomized trials: the case of implementation research. Clin Trials. 2005;2(2):99–107.
Lancaster GA, Chellaswamy H, Taylor S, Lyon D, Dowrick C. Design of a clustered observational study to predict emergency admissions in the elderly: statistical reasoning in clinical practice. J Eval Clin Pract. 2007;13(2):169–78, Apr.
Johnson DW, Craig W, Brant , Mitton C, Svenson L, Klassen TP. A cluster randomized controlled trial comparing three methods of disseminating practice guidelines for children with croup. Implement Sci. 2006;1:10.
Freedman LS, Gail MH, Green SB, Corle DK. The efficiency of the matched-pairs design of the Community Intervention Trial for Smoking Cessation (COMMIT). Control Clin Trials. 1997;18(2):131–9, Apr.
Glasgow RE, Nutting PA, King DK, Nelson CC, Cutter G, Gaglio B, et al. Randomized effectiveness trial of a computer-assisted intervention to improve diabetes care. Diabetes Care. 2005;28(1):33–9, Jan.
Grosskurth H, Mosha F, Todd J, Mwijarubi E, Klokke A, Senkoro K, et al. Impact of improved treatment of sexually transmitted diseases on HIV infection in rural Tanzania: randomised controlled trial. Lancet.. 1995;346(8974):530–6, Aug 26.
Gulmezoglu AM, Villar J, Grimshaw J, Piaggio G, Lumbiganon P, Langer A. Cluster randomized trial of an active, multifaceted information dissemination intervention based on The WHO Reproductive health library to change obstetric practices: methods and design issues. BMC Med Res Methodol. 2004;4(1):2, Jan 15.
Thompson SG, Pyke SD, Hardy RJ. The design and analysis of paired cluster randomized trials: an application of meta-analysis techniques. Stat Med. 1997;16(18):2063–79, Sep 30.
American Diabetes Association. Standards of medical care for patients with diabetes mellitus. Diabetes Care. 2003;26(1):S33–50, Jan.
Raab GM, Butcher I. Balance in cluster randomized trials. Stat Med. 2001;20(3):351–65, Feb 15.
Raab GM, Butcher I. Randomization inference for balanced cluster-randomized trials. Clin Trials. 2005;2(2):130–40.
Cohen J. Statistical Power Analysis for the Behavioral Sciences. Toronto: Academic Press, Inc.; 1977.
Ahmed A, Husain A, Love TE, et al. Heart failure, chronic diuretic use, and increase in mortality and hospitalization: an observational study using propensity score methods. Eur Heart J. 2006;27(12):1431–9, Jun.
Normand ST, Landrum MB, Guadagnoli E, et al. Validating recommendations for coronary angiography following acute myocardial infarction in the elderly: a matched analysis using propensity scores. J Clin Epidemiol. 2001;54(4):387–98, Apr.
Kerry SM, Bland JM. The intracluster correlation coefficient in cluster randomisation. BMJ. 1998;316(7142):1455, May 9.
Feng Z, Diehr P, Peterson A, McLerran D. Selected statistical issues in group randomized trials. Annu Rev Public Health. 2001;22:167–87.
Kerry SM, Bland JM. Sample size in cluster randomisation. BMJ. 1998;316(7130):549, Feb 14.
Kerry SM, Bland JM. Trials which randomize practices II: sample size. Fam Pract. 1998;15(1):84–7, Feb.
Campbell MK, Elbourne D, Altman DG. CONSORT statement: extension to cluster randomised trials. BMJ. 2004;328(7441):702–8, Mar 20.
Campbell MK, Mollison J, Steen N, Grimshaw JM, Eccles M. Analysis of cluster randomized trials in primary care: a practical approach. Fam Pract. 2000;17(2):192–6, Apr.
Campbell MK, Mollison J, Grimshaw JM. Cluster trials in implementation research: estimation of intracluster correlation coefficients and sample size. Stat Med. 2001;20(3):391–9, Feb 15.
Cebul RD, Dawson NV, Love TE. Cluster Randomized Trials in Health Care ResearchIn: Machin D, Day S, Green S (eds). The Textbook of Clinical Trials 2nd ed. New York: Wiley; 2006.
StataCorp. Intercooled Stata/SE 9.2 for WindowsCollege Station, TX: StataCorp; 2007.
Puffer S, Torgerson D, Watson J. Evidence for risk of bias in cluster randomised trials: review of recent trials published in three general medical journals. BMJ. 2003;327(7418):785–9, Oct 4.
Eccles M, Hawthorne G, Whitty P, et al. A randomised controlled trial of a patient based Diabetes Recall and Management System: the DREAM trial: a study protocol. BMC Health Serv Res. 2002;2(1):5, Mar 21.
Brown AF, Gregg EW, Stevens M, Karter AJ, Weinberger M, Safford MM, et al. Race, ethnicity, socioeconomic position, and quality of care for adults with diabetes enrolled in managed care: the Translating Research Into Action for Diabetes (TRIAD) study. Diabetes Care. 2005;28(12):2864–70, Dec.
Hsu J, Price M, Huang J, Brand , Fung V, Hui , et al. Unintended consequences of caps on Medicare drug benefits. N Engl J Med. 2006;354(22):2349–59, Jun 1.
Murray DM, Blitstein JL, Hannan PJ, Baker WL, Lytle LA. Sizing a trial to alter the trajectory of health behaviours: methods, parameter estimates, and their application. Stat Med. 2007;26(11):2297–2316, May 20.
Acknowledgments
An earlier version of this work was presented at the Society for Medical Decision Making Annual Meeting in Boston, October 2006. This work is supported in part by grant no. R01 HS15123 from the Agency for Healthcare Research and Quality. The authors wish to thank Mary Jo Roach, Ph.D., and Mark Votruba, Ph.D. for their substantial contributions to DIG-IT.
Conflicts of Interest
Dr. Love has received an honorarium for a short course given to Amgen, Inc. Dr. Einstadter has served as a consultant to Medical Mutual of Ohio. Dr. Dawson has served as a consultant to Gerson Lehrman Group. None of these activities are related to the research described in this report.
Author information
Authors and Affiliations
Consortia
Corresponding author
Rights and permissions
About this article
Cite this article
Love, T.E., Cebul, R.D., Einstadter, D. et al. Electronic Medical Record-Assisted Design of a Cluster-Randomized Trial to Improve Diabetes Care and Outcomes. J GEN INTERN MED 23, 383–391 (2008). https://doi.org/10.1007/s11606-007-0454-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11606-007-0454-3