Abstract
Background
This study aimed to evaluate the additional value of laparoscopic ultrasound (LUS) to staging laparoscopy (SL) for detecting occult liver metastases in patients with potentially resectable pancreatic head cancer.
Methods
A retrospective cohort study was performed including all patients who underwent SL and LUS between 2005 and 2016. LUS was performed during SL to detect liver metastases not found by preoperative imaging or visual inspection of the liver.
Results
Out of 197 patients, visual inspection during SL detected distant metastases in 29 (14.7%) patients. LUS was performed in 127 patients, revealing 3 additional liver metastases. The proportion of patients with unresectable disease after SL and negative LUS was 32.3%, which was similar to 36.6% of patients with unresectable disease after SL without LUS (difference 4.3%; 95% CI − 13–23%; P = 0.61). Sensitivity, specificity, and positive and negative predictive values of LUS to detect liver metastases were 30, 100, 100, and 94%, respectively. The proportion of patients with distant metastases diagnosed at SL significantly increased over time (P = 0.031).
Conclusion
The routine use of LUS during SL for patients with potentially resectable pancreatic head cancer cannot be recommended. Imaging should be repeated when significant delay occurs between index CT and the scheduled surgery.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Pancreatic cancer has a poor prognosis.1,2 Most patients are not amenable for surgery because of infiltration of local structures and disseminated disease or because of significant comorbidities.3 Routine preoperative workup includes computed tomography (CT) to evaluate local resectability and to exclude disseminated disease.4 Despite this, in approximately 10–25% of patients, locally advanced disease or occult distant metastases are identified during exploratory laparotomy.5 Staging laparoscopy (SL) may decrease the rate of futile operations in those found to have resectable disease on CT.5,6
SL can be combined with laparoscopic ultrasound (LUS) to allow a detection of occult liver metastases missed by preoperative CT. Due to the improvement of imaging techniques over the last decade, however, the value of LUS in addition to SL and preoperative CT may have diminished.5,7 Therefore, the aim of this study was to evaluate the additional value of LUS to SL in patients with potentially resectable pancreatic head cancer.
Methods
Design and Participants
A single-center retrospective analysis was performed of all patients who underwent SL for suspected pancreatic head or periampullary cancer at the University Medical Center Groningen, The Netherlands, between January 2005 and December 2016. Patients were identified using the internal database for pancreatic cancer registration in the University Medical Center Groningen. All patients underwent extensive preoperative staging including computed tomography (CT) with iodine contrast. A late arterial phase with a delay of 15 s after bolus tracking and a portal venous phase with a scan delay of 75 s postinjection was performed. Reconstructions were made with a slice thickness of 0.75 and 2 mm in the arterial phase and a slice thickness of 2 mm in the portal venous phase. All CT scans were assessed by a dedicated HPB radiologist.
All patients were discussed in our weekly tumor board meeting by an interdisciplinary group of hepatopancreatobiliary surgeons, radiologists, gastroenterologists, and oncologists. Those patients with suspected pancreatic head cancer who were considered potentially resectable and curable were routinely scheduled for SL. In patients with a history of multiple laparotomies or severe lung disease not tolerating a pneumoperitoneum, SL was contraindicated. In patients with a clear cut preoperative diagnosis of cancer of the ampulla of Vater, the duodenum or the distal bile duct SL was only indicated if indeterminate intra-abdominal lesions were detected on preoperative imaging. Patients with an upfront indication for either resection or (palliative) bypass due to for example subtotal biliary or gastric outlet obstruction untreatable with endoscopy directly underwent surgical exploration. Suspicious lesions on imaging were biopsied preoperatively using percutaneous or endoscopic ultrasound and reviewed for histological characteristics. This study was approved by the Ethics Committee of the University Medical Center Groningen.
Surgical Procedures
SL was performed using an open subumbilical introduction of a 10-mm trocar for the 30° angled camera. Capnoperitoneum was established to a pressure of 12–15 mmHg with CO2. One 12-mm trocar was introduced to the left subcostal for the ultrasound probe, and one 5-mm trocar was introduced to the right subcostal. The visceral, parietal, and the left and right subphrenical peritoneum, diaphragm, lower abdomen, and ligament of Treitz were routinely inspected for metastases. The bursa omentalis was left unopened. The liver was visually inspected on all sides and manipulated using a laparoscopic retractor to allow inspection of the inferior surface. The ultrasound probe (Hitachi Aloka Medical Ltd., Tokyo, Japan, Model UST-5550) was introduced, and the liver parenchyma was visualized systematically. Whenever possible, suspicious lesions were biopsied and sent to pathology. The definitive histology of all samples was confirmed postoperatively. When lesions were not readily accessible for biopsy, new postoperative imaging and/or ultrasound guided biopsy were performed to confirm metastatic disease. Exploratory laparotomy was performed when no contraindications were found during preoperative imaging and SL. Laparotomy was followed by routine inspection for distant disease and presence of locally advanced disease. Lesions suspected for metastases were biopsied immediately and sent for frozen section analysis. Para-aortic lymph nodes (station 16) and celiac trunk lymph nodes (station 9) were routinely sampled and sent for frozen section analysis. Positive lymph nodes were considered a contraindication for resection due to reported poor prognosis of this finding.8 Usually, resection was terminated and palliative double bypass was performed. Pancreatoduodenectomy was performed when there was no arterial involvement and/or untreatable involvement of the superior mesenteric or portal vein. All patients followed standardized preoperative and postoperative treatment protocols. All procedures were performed or supervised by an experienced hepatopancreatobiliary surgeon.
Statistical Analysis
Statistical analysis was performed with SPSS (version 23.0, SPSS Inc., Chicago, IL, USA). Normality of distribution was assessed and checked for skewness. Continuous data were expressed in medians with interquartile ranges (IQR) and categorical variables in numbers with percentages. Variables were compared between the LUS and non-LUS groups using the Mann-Whitney U test, chi-squared test, and Fisher’s exact test, where appropriate. A 2 × 2 contingency table and chi-squared test were used to compare proportions. A two-sided P value < 0.05 was considered statistically significant.
Results
A total of 518 patients with suspected pancreatic head cancer were eligible for surgery between January 2005 and December 2016. Because of aforementioned reasons, 321 patients directly underwent exploratory laparotomy and were excluded from analysis. A total of 197 patients underwent SL prior to exploration. During SL, 29 (14.7%) patients proved to have distant metastases by visual inspection, of which 15 patients had liver metastases, 6 with peritoneal metastases, and 8 with liver as well as peritoneal metastases. In 41 patients, no LUS was performed (no LUS group; most commonly because of adhesions or technical reasons), leaving 127 patients screened by LUS (LUS group; Fig. 1).
The median age of the patients that underwent SL was 67 years. There was a predominance of males (53.3%). The median time between CT and SL was 52 days. Most patients that underwent SL were eventually diagnosed with a pancreatic ductal adenocarcinoma at definitive pathology (73.4%). Patient characteristics are presented in Table 1. The median (IQR) operative time of SL with and without LUS was 65 (52–84) min and 60 (41–128) min, respectively (P = 0.541).
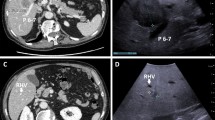
Screening with LUS revealed 3 (2.4%) lesions suspected for liver metastases that were not detected by preoperative imaging or visual inspection (Fig. 1). Metastasis was confirmed using ultrasound-guided percutaneous biopsy in one case and by new CT imaging in two cases (true positives) (Table 2; Fig. 2). After SL and LUS, four patients did not undergo subsequent exploration because of worsening patient factors that made them unfit for explorative laparotomy (Fig. 1).
Follow-up imaging of the three patients with positive laparoscopic ultrasound (LUS) findings. The arrows point to the liver metastases found during LUS. Percutaneous ultrasound showed a 9-mm lesion in liver segment 4 (a). Computed tomography (CT) confirmed a 10-mm lesion in liver segment 8 and detected two other lesions of 8 and 13 mm in liver segment 8 (b). CT demonstrated an 8-mm lesion in liver segment 5 (c)
Out of 127 patients who underwent SL and LUS (LUS group), 120 patients subsequently underwent exploratory laparotomy (Fig. 1). After surgical exploration, seven patients did not undergo resection due to new found liver metastases (false negatives). In 30 patients, resection was terminated due to locally advanced findings such as vascular involvement or positive distal (para-aortic or celiac trunk) lymph nodes (true negatives). In 83 patients (65.4%), a pancreatoduodenectomy was performed (true negatives).
All 41 patients who underwent SL without LUS (no LUS group) subsequently underwent explorative laparotomy (Fig. 1). After surgical exploration, peritoneal metastases or liver metastases were found in one and three patients, respectively. In 11 patients, resection was terminated due to locally advanced findings or positive distal lymph nodes. In 26 patients, a pancreatoduodenectomy was performed.
The proportion of patients with unresectable disease after SL and negative LUS was 32.3% (41 out of 127 patients), whereas the proportion of patients with unresectable disease after SL without LUS was 36.6% (15 out of 41 patients). This resulted in a proportion difference of 4.3% (95% CI − 13–23%; chi-squared 0.257; 1 df; P = 0.61).
For determination of the effectiveness of LUS as a screening tool, sensitivity, specificity, positive predicted value (PPV), and negative predicted value (NPV) were calculated based on 123 patients with suspected pancreatic head cancer who underwent SL and LUS and were potentially eligible for exploratory laparotomy. Sensitivity was 30% (95% CI 6.7–65%), specificity was 100% (95% CI 97–100%), PPV was 100%, and NPV was 94% (95% CI 92–96%). After excluding 17 patients whose pathology specimen proved benign, sensitivity was 30% (95% CI 6.7–65%), specificity was 100% (95% CI 96–100%), PPV was 100%, and NPV was 93% (95% CI 90–95%).
Because in the three true positive cases the time between CT and SL was 83, 63, and 70 days, respectively, we calculated the median time between CT and SL in all 197 patients who underwent SL. Median time between CT and SL in the 29 patients with metastases detected during SL was 62 days (IQR 46.5–79 days) versus 51.5 days (IQR 34–68.75 days) in the 168 patients who were considered resectable after visual inspection at SL (P = 0.083). When time between CT and SL was within 30 days, 7.9% of patients showed metastases during SL, compared with 12.5% of patients after 60 days, 17.5% of patients after 90 days, and 29.4% of patients when time between CT and SL exceeded 90 days (Fig. 3). The proportion of patients with distant metastases diagnosed at SL significantly increased over time (P = 0.031).
Absolute number of patients in which distant metastases were detected or not during staging laparoscopy (SL) categorized by 30-day intervals following preoperative computed tomography (CT; a). The proportion of patients with distant metastases diagnosed at SL significantly increased over time (P = 0.031; b)
Discussion
In the present cohort, we found that LUS in addition to SL was not of significant additional value to detect radiological occult liver metastases in patients with potentially resectable pancreatic head cancer.
Several studies have investigated LUS as a screening tool for primary tumor resectability. In these studies, the detection of distant metastases was a secondary objective. Overall, the results of these studies have suggested that LUS may improve the sensitivity to detect liver metastases.9,10,11,12,13,14,15
Few studies have investigated the additional value of LUS compared to modern preoperative imaging modalities. However, study cohorts were small (n < 50), and only few patients were diagnosed with occult distant disease (one and two cases, respectively).16,17 The outcomes of our study are more in accordance with the results of a larger study in which the role of intraoperative ultrasound during exploratory laparotomy was investigated. In less than 1% of 470 patients the liver metastases found by intraoperative ultrasound resulted in a significant change of management.18
The two most recently published cohorts regarding this topic included patients from 2002–2007 to 2005–2008. In the first study, a historical cohort of 40 patients who underwent LUS between January 1995 and January 1999 was compared with 9 high-risk patients who underwent LUS between 2002 and 2007. LUS was only performed on a pool of patients who fulfilled certain criteria associated with a higher risk of unresectability, e.g., suspicion of small liver metastases, signs of peritoneal carcinomatosis or incipient ascites, tumors > 4 cm, and lesions of the body and tail, thus increasing the yield of LUS.19 LUS confirmed preoperative CT data in eight out of nine patients (seven patients with liver metastases and one with peritoneal carcinomatosis). In this subgroup of patients, LUS only would have had additional value in one case.20 In the second study of 48 patients with pancreatic head cancer, LUS had additional value in 7 patients (14.6%), because of mesenteric vein infiltration (4 patients), involvement of the celiac or para-aortic nodes (2 patients), or liver metastases (1 patient). All patients preoperatively underwent staging with biphasic contrast-enhanced CT.16 In our study, LUS was performed routinely and not for specifically selected high-risk patients. Furthermore, in our cohort, all patients were preoperatively staged using tri-phasic CT.
A recent systematic review calculated that the sensitivity, specificity, PPV, and NPV in previously published studies were excellent to determine resectability. The two most recent cohort studies reached a sensitivity, specificity, PPV, and NPV of 100%.21 However, these calculations were based on studies that were heterogeneous in resectability criteria, use of multimodal imaging protocols, and the quality of CT technology. More importantly, in the systematic review, sensitivity, specificity, PPV, and NPV were calculated for a combined yield of SL and LUS,21 whereas in our study, we have studied the value of LUS in addition to SL for the detection of liver metastases. This explains the findings of a significantly lower diagnostic yield in our study.
The current study is the first large series on the additional value of LUS during SL. LUS had diagnostic gain in only three cases (4%) in addition to abdominal CT and SL. In all cases, abdominal CT and abdominal ultrasound confirmed metastatic disease shortly after SL and LUS (Table 2). Taking the considerable time delay (63 to 83 days) between preoperative CT and SL/LUS into account, we hypothesize that more recent preoperative imaging would have revealed metastatic disease prior to surgery. Analysis of our data showed that an increasing interval between CT and SL increases the chance that distant metastases were found during SL. Also, longer intervals between preoperative imaging and definitive surgery may have an effect on resectability rates and possible poor survival. One study demonstrated that the resection rate was significantly higher when the imaging-to-resection interval was 32 days or less, when compared to longer waiting times (87 versus 74%).22 Another recent study described that the implementation of a fast track pathway without preoperative biliary drainage for periampullary malignancies leads to a significant reduction in time from index CT to surgery, when compared to those who had been stented before referral (16 versus 65 days). In this study, the resection rate of the group without preoperative biliary drainage was significantly higher, when compared to that of the not drained group (97 versus 75%).23 This underlines the importance to perform surgery shortly after the index scan and to repeat radiological imaging when any delay occurs.
Previous studies have shown that elevated cancer antigen 19-9 levels were associated with an increased yield of metastatic disease found during SL.24,25,26,27 A cancer antigen 19-9 level of 215 U/ml was proposed to select high-risk patients.24 If this cutoff point would have been applied to the current series, about half of the patients with distant metastases would unfairly be excluded for SL (data not shown). The calculation of an optimal threshold for elevated cancer antigen 19-9 levels, however, fell beyond the scope of the current study.
Some limitations need to be taken into consideration. First, this is a cross-sectional analysis of a cohort study; thus, cause-effect relationships cannot be established with certainty. Second, the retrospective design comes with well-known limitations such as selection bias. In our study, we believe this potential bias was limited because the decision to perform LUS or not was not made until SL was commenced. We assume that technical errors followed a random pattern and that severe adhesions were not related to the outcome (i.e., liver metastases).
In conclusion, our findings demonstrate that with current imaging techniques, LUS in addition to SL is of limited value to detect radiologically occult liver metastases. Approximately 25 patients with potentially resectable pancreatic head cancer need to be screened with LUS during SL to prevent one additional exploratory laparotomy. With a sensitivity of only 30%, liver metastases can still be missed by LUS in a majority of patients. Perhaps, more important is that repeated imaging should be performed when significant delay occurs between index CT and the scheduled surgery.
References
Ryan DP, Hong TS, Bardeesy N. Pancreatic adenocarcinoma. N Engl J Med 2014;371(11): 1039–1049.
Siegel RL, Miller KD, Jemal A. Cancer Statistics, 2017. CA Cancer J Clin 2017;67(1): 7–30.
Bilimoria KY, Bentrem DJ, Ko CY, Stewart AK, Winchester DP, Talamonti MS. National failure to operate on early stage pancreatic cancer. Ann Surg 2007;246(2): 173–180.
Tamburrino D, Riviere D, Yaghoobi M, Davidson BR, Gurusamy KS. Diagnostic accuracy of different imaging modalities following computed tomography (CT) scanning for assessing the resectability with curative intent in pancreatic and periampullary cancer. Cochrane Database Syst Rev 2016;9: CD011515.
Allen VB, Gurusamy KS, Takwoingi Y, Kalia A, Davidson BR. Diagnostic accuracy of laparoscopy following computed tomography (CT) scanning for assessing the resectability with curative intent in pancreatic and periampullary cancer. Cochrane Database Syst Rev 2016;7: CD009323.
Beenen E, Van Roest MHG, Sieders E, Peeters PMJG, Porte RJ, De Boer MT, De Jong KP. Staging laparoscopy in patients scheduled for pancreaticoduodenectomy minimizes hospitalization in the remaining life time when metastatic carcinoma is found. Eur J Surg Oncol 2014;40(8):989–94.
Long EE, Van Dam J, Weinstein S, Jeffrey B, Desser T, Norton JA. Computed tomography, endoscopic, laparoscopic, and intra-operative sonography for assessing resectability of pancreatic cancer. Surg Oncol 2005;14(2): 105–113.
Schwarz L, Lupinacci RM, Svrcek M, Lesurtel M, Bubenheim M, Vuarnesson H, Balladur P, Paye F. Para-aortic lymph node sampling in pancreatic head adenocarcinoma. Br J Surg 2014;101(5): 530–538.
Bemelman WA, de Wit LT, van Delden OM, Smits NJ, Obertop H, Rauws EJ, Gouma DJ. Diagnostic laparoscopy combined with laparoscopic ultrasonography in staging of cancer of the pancreatic head region. Br J Surg 1995;82(6): 820–824.
Callery MP, Strasberg SM, Doherty GM, Soper NJ, Norton JA. Staging laparoscopy with laparoscopic ultrasonography: optimizing resectability in hepatobiliary and pancreatic malignancy. J Am Coll Surg 1997;185(1): 33–39.
Catheline JM, Turner R, Rizk N, Barrat C, Champault G. The use of diagnostic laparoscopy supported by laparoscopic ultrasonography in the assessment of pancreatic cancer. Surg Endosc 1999;13(3): 239–245.
John TG, Greig JD, Carter DC, Garden OJ. Carcinoma of the pancreatic head and periampullary region. Tumor staging with laparoscopy and laparoscopic ultrasonography. Ann Surg 1995;221(2): 156–164.
Menack MJ, Spitz JD, Arregui ME. Staging of pancreatic and ampullary cancers for resectability using laparoscopy with laparoscopic ultrasound. Surg Endosc 2001;15(10): 1129–1134.
Minnard EA, Conlon KC, Hoos A, Dougherty EC, Hann LE, Brennan MF. Laparoscopic ultrasound enhances standard laparoscopy in the staging of pancreatic cancer. Ann Surg 1998;228(2): 182–187.
Thomson BN, Parks RW, Redhead DN, Welsh FK, Madhavan KK, Wigmore SJ, Garden OJ. Refining the role of laparoscopy and laparoscopic ultrasound in the staging of presumed pancreatic head and ampullary tumours. Br J Cancer 2006;94(2): 213–217.
Piccolboni D, Ciccone F, Settembre A, Corcione F. Laparoscopic intra-operative ultrasound in liver and pancreas resection: Analysis of 93 cases. J Ultrasound 2010;13(1): 3–8.
Piccolboni P, Settembre A, Angelini P, Esposito F, Palladino S, Corcione F. Laparoscopic ultrasound: a surgical “must” for second line intra-operative evaluation of pancreatic cancer resectability. G Chir 2015;36(1): 5–8.
Mui LW, Pursell LJ, Botwinick IC, Allendorf JD, Chabot JA, Newhouse JH. Routine intraoperative hepatic sonography does not affect staging or postsurgical hepatic recurrence in pancreatic adenocarcinoma. J Ultrasound Med 2014;33(1): 47–51.
Pisters PWT, Lee JE, Vauthey C, Charnsangavej C, Evans DB. Laparoscopy in the staging of pancreatic cancer. Br J Surg 2001; 88: 325–337.
Barabino M, Santambrogio R, Pisani Ceretti A, Scalzone R, Montorsi M, Opocher E. Is there still a role for laparoscopy combind with laparoscopic ultrasonography in the staging of pancreatic cancer? Surg Endosc 2011;25(1): 160–165.
Levy J, Tahiri M, Vanounou T, Maimon G, Bergman S. Diagnostic laparoscopy with ultrasound still has a role in the staging of pancreatic cancer: a systematic review of the literature. HPB Surg; 2016; 2016:8092109.
Sanjeevi S, Ivanics T, Lundell L, Kartalis N, Andren-Sandberg A, Blomberg J, Del Chiaro M, Ansorge C. Impact of delay between imaging and treatment in patients with potentially curable pancreatic cancer. Br J Surg 2016;103(3): 267–275.
Roberts KJ, Prasad P, Steele Y, Marcon F, Faulkner T, Cilliers H, Dasari B, Abradelo M, Marudanayagam R, Sutcliffe RP, Muiesan P, Mirza DF, Isaac J. A reduced time to surgery within a ‘fast track’ pathway for periampullary malignancy is associated with an increased rate of pancreatoduodenectomy. HPB 2017;19(8):713–20.
Alexakis N, Gomatos IP, Sbarounis S, Toutouzas K, Katsaragakis S, Zografos G, et al. High serum CA 19-9 but not tumor size should select patients for staging laparoscopy in radiological resectable pancreas head and peri-ampullary cancer. Eur J Surg Oncol 2015;41(2): 265–269.
Connor S, Bosonnet L, Alexakis N, Raraty M, Ghaneh P, Sutton R, et al. Serum CA19-9 measurement increases the effectiveness of staging laparoscopy in patients with suspected pancreatic malignancy. Dig Surg 2005;22(1–2): 80–85.
Callery MP, Chang KJ, Fishman EK, Talamonti MS, William Traverso L, Linehan DC. Pretreatment assessment of resectable and borderline resectable pancreatic cancer: expert consensus statement. Ann Surg Oncol 2009;16(7): 1727–1733.
Halloran CM, Ghaneh P, Connor S, Sutton R, Neoptolemos JP, Raraty MGT. Carbohydrate antigen 19-9 accurately selects patients for laparoscopic assessment to determine resectability of pancreatic malignancy. Br J Surg 2007;95(4): 453–459.
Acknowledgements
The authors thank Dr. Wouter J. Derksen for his contribution to the manuscript.
Author information
Authors and Affiliations
Contributions
Each author has participated sufficiently in the work to take public responsibility for appropriate portions of the content as per the guidelines of the International Committee of Medical Journal Editors.
Corresponding author
Ethics declarations
This study was approved by the Ethics Committee of the University Medical Center Groningen.
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
This abstract was presented on the IHPBA 12th World Congress 2016, Sao Paolo, Brazil.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Looijen, G.A., Pranger, B.K., de Jong, K.P. et al. The Additional Value of Laparoscopic Ultrasound to Staging Laparoscopy in Patients with Suspected Pancreatic Head Cancer. J Gastrointest Surg 22, 1186–1192 (2018). https://doi.org/10.1007/s11605-018-3726-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11605-018-3726-9