Abstract
Background
Detection of pancreatic ductal adenocarcinoma (PDAC) in asymptomatic patients is very rare, and the clinical implication of early detection of asymptomatic PDAC remains unclear.
Methods
This retrospective study included 569 consecutive patients with PDAC treated in our institution (250 underwent surgical resection and 319 had unresectable PDAC). The patients’ demographics, tumor locations, pathologic stages, treatment, and overall survival (OS) were compared between the asymptomatic and symptomatic patients.
Results
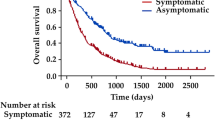
In total, 163 (29%) patients presented without subjective symptoms. These patients had an earlier stage of PDAC on presentation (p < 0.001), higher resectability rate (64 vs. 36%, p < 0.001), and higher 5-year OS rate (18 vs. 7%, p < 0.001) than patients with symptoms. Among the patients who underwent resection, asymptomatic patients did not have a significantly higher chance of complete resection (88 vs. 78%, p = 0.06) or 5-year OS rate (23 vs. 22%, p = 0.09). However, symptomatic patients more often required complex operations such as concomitant vascular resection and reconstruction (57 vs. 29%, p < 0.001).
Conclusions
Asymptomatic PDAC is associated with better long-term outcomes than symptomatic PDAC because of the earlier stage at presentation and higher chance of resectability. Our findings highlight the potential implication of screening programs for early detection of PDAC in selected high-risk populations.


Similar content being viewed by others
References
Jemal A, Siegel R, Xu J, et al. Cancer statistics, 2010. CA Cancer J Clin 2010;60:277–300.
Cancer Research UK. Pancreatic cancer survival statistics. http://www.cancerresearchuk.org/health-professional/cancer-statistics/statistics-by-cancer-type/pancreatic-cancer/survival [Accessed 19 October 2016].
Kalser MH, Barkin J, MacIntyre JM. Pancreatic cancer. Assessment of prognosis by clinical presentation. Cancer 1985;56:397–402.
Winter JM, Cameron JL, Lillemoe KD, et al. Periampullary and pancreatic incidentaloma: a single institution’s experience with an increasingly common diagnosis. Ann Surg 2006;243:673–80; discussion 680-3.
Lahat G, Ben Haim M, Nachmany I, et al. Pancreatic incidentalomas: high rate of potentially malignant tumors. J Am Coll Surg 2009;209:313–9.
National Comprehensive Cancer Network. NCCN Guidelines Version 1.2013 Pancreatic Adenocarcinoma. https://www.tri-kobe.org/nccn/guideline/archive/pancreas2013/english/pancreatic.pdf [Accessed 1 April 2015].
Inoue Y, Saiura A, Yoshioka R, et al. Pancreatoduodenectomy With Systematic Mesopancreas Dissection Using a Supracolic Anterior Artery-first Approach. Ann Surg 2015;262:1092–101.
Yoshioka R, Saiura A, Koga R, et al. Risk factors for clinical pancreatic fistula after distal pancreatectomy: analysis of consecutive 100 patients. World J Surg 2010;34:121–5.
Sato T, Saiura A, Inoue Y, et al. Distal Pancreatectomy with En Bloc Resection of the Celiac Axis with Preservation or Reconstruction of the Left Gastric Artery in Patients with Pancreatic Body Cancer. World J Surg 2016;40:2245–53.
Sobin LH, Gospodarowicz MK, Wittekind C. TNM classification of malignant tumours: Hoboken: Wiley, 2011.
Canto MI, Harinck F, Hruban RH, et al. International Cancer of the Pancreas Screening (CAPS) Consortium summit on the management of patients with increased risk for familial pancreatic cancer. Gut 2013;62:339–47.
Bruzoni M, Johnston E, Sasson AR. Pancreatic incidentalomas: clinical and pathologic spectrum. Am J Surg 2008;195:329–32; discussion 332.
Takamura A. The Present Circumstance of Primary Care in Japan. Qual Prim Care 2015;23:262–6.
Brennan MF, Moccia RD, Klimstra D. Management of adenocarcinoma of the body and tail of the pancreas. Ann Surg 1996;223:506–11; discussion 511-2.
Sperti C, Pasquali C, Pedrazzoli S. Ductal adenocarcinoma of the body and tail of the pancreas. J Am Coll Surg 1997;185:255–9.
Agarwal B, Correa AM, Ho L. Survival in pancreatic carcinoma based on tumor size. Pancreas 2008;36:e15–20.
Shimada K, Sakamoto Y, Sano T, et al. Reappraisal of the clinical significance of tumor size in patients with pancreatic ductal carcinoma. Pancreas 2006;33:233–9.
Katz MH, Wang H, Fleming JB, et al. Long-term survival after multidisciplinary management of resected pancreatic adenocarcinoma. Ann Surg Oncol 2009;16:836–47.
Cleary SP, Gryfe R, Guindi M, et al. Prognostic factors in resected pancreatic adenocarcinoma: analysis of actual 5-year survivors. J Am Coll Surg 2004;198:722–31.
Ferrone CR, Brennan MF, Gonen M, et al. Pancreatic adenocarcinoma: the actual 5-year survivors. J Gastrointest Surg 2008;12:701–6.
Han SS, Jang JY, Kim SW, et al. Analysis of long-term survivors after surgical resection for pancreatic cancer. Pancreas 2006;32:271–5.
Howard TJ, Krug JE, Yu J, et al. A margin-negative R0 resection accomplished with minimal postoperative complications is the surgeon’s contribution to long-term survival in pancreatic cancer. J Gastrointest Surg 2006;10:1338–45; discussion 1345-6.
Riall TS, Cameron JL, Lillemoe KD, et al. Resected periampullary adenocarcinoma: 5-year survivors and their 6- to 10-year follow-up. Surgery 2006;140:764–72.
Schnelldorfer T, Ware AL, Sarr MG, et al. Long-term survival after pancreatoduodenectomy for pancreatic adenocarcinoma: is cure possible? Ann Surg 2008;247:456–62.
Conroy T, Desseigne F, Ychou M, et al. FOLFIRINOX versus gemcitabine for metastatic pancreatic cancer. N Engl J Med 2011;364:1817–25.
Von Hoff DD, Ervin T, Arena FP, et al. Increased survival in pancreatic cancer with nab-paclitaxel plus gemcitabine. N Engl J Med 2013;369:1691–703.
Canto MI, Hruban RH, Fishman EK, et al. Frequent detection of pancreatic lesions in asymptomatic high-risk individuals. Gastroenterology 2012;142:796–804; quiz e14-5.
Poley JW, Kluijt I, Gouma DJ, et al. The yield of first-time endoscopic ultrasonography in screening individuals at a high risk of developing pancreatic cancer. Am J Gastroenterol 2009;104:2175–81.
Vasen HF, Wasser M, van Mil A, et al. Magnetic resonance imaging surveillance detects early-stage pancreatic cancer in carriers of a p16-Leiden mutation. Gastroenterology 2011;140:850–6.
Acknowledgements
This study was supported by a grant from the Project for Development of Innovative Research on Cancer Therapeutics (P-Direct) and the Japan Agency for Medical Research and Development.
Author information
Authors and Affiliations
Contributions
YT—conception or design of the work and drafting the work
AS—conception or design of the work and critical revision of the work
YT—conception or design of the work and critical revision of the work
YI—conception or design of the work and critical revision of the work
TI—conception or design of the work and critical revision of the work
YM—conception or design of the work and critical revision of the work
MM—acquisition of data for the work and critical revision of the work
HI—acquisition of data for the work and critical revision of the work
RM—acquisition of data for the work and critical revision of the work
MT—acquisition of data for the work and critical revision of the work
HI—conception or design of the work and drafting and critical revision of the work
All authors approved the version of the manuscript to be published and agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy and integrity of any part of the work were appropriately investigated and resolved.
Corresponding author
Ethics declarations
This study was approved by the Cancer Institute Hospital Institutional Review Board (2015-1078).
Rights and permissions
About this article
Cite this article
Takeda, Y., Saiura, A., Takahashi, Y. et al. Asymptomatic Pancreatic Cancer: Does Incidental Detection Impact Long-Term Outcomes?. J Gastrointest Surg 21, 1287–1295 (2017). https://doi.org/10.1007/s11605-017-3421-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11605-017-3421-2