Abstract
Background
We sought to estimate the cost-effectiveness of hepatic resection (HR) (strategy A) relative to surveillance plus 6 months of additional systemic chemotherapy (sCT) (strategy B) for patients with colorectal disappearing liver metastases (DLM).
Methods
A Markov model was developed using data from a systematic literature review. Three base cases were evaluated: (1) a 60-year-old patient with three lesions in the right hemi-liver who underwent 6 months of sCT, had normalized carcinoembryonic antigen (CEA), and was diagnosed with DLM through a computed tomography (CT) scan; (2) a 60-year-old patient with three lesions in the right hemi-liver who underwent 6 months of sCT, had normalized CEA, and was diagnosed with DLM through a magnetic resonance imaging (MRI) scan; and (3) a 60-year-old patient with three lesions in the right hemi-liver who underwent 6 months of sCT plus hepatic artery infusion (HAI), had normalized CEA, and was diagnosed with DLM through a MRI scan. The outcomes evaluated were quality-adjusted life months (QALMs), incremental cost-effectiveness ratio (ICER), and net health benefit (NHB).
Results
The NHB of strategy A versus strategy B was positive in base case 1 (7.7 QALMs, ICER $34.449/quality-adjusted life year (QALY)) and base case 2 (1.6 QALMs, ICER $43,948/QALY). In contrast it was negative (−0.2 QALMs, ICER $72,474/QALY) for base case 3. Monte Carlo simulation showed that strategy B is acceptable only in old patients (>60 years) with normalized CEA and MRI-based diagnosis. In younger patients, strategy B may reach cost-effectiveness only after sCT plus HAI.
Conclusion
Surveillance of DLM after sCT was more beneficial and cost-effective among patients >60 years with multiple factors predictive of true complete pathological response, such as normalization of CEA, HAI therapy, BMI ≤30 kg/m2, and diagnosis of DLM made through MRI.


Similar content being viewed by others
References
Leporrier, J., et al., A population-based study of the incidence, management and prognosis of hepatic metastases from colorectal cancer. Br J Surg, 2006. 93(4): p. 465–74.
Mayo, S.C., et al., Shifting trends in liver-directed management of patients with colorectal liver metastasis: a population-based analysis. Surgery, 2011. 150(2): p. 204–16.
Andres, A., et al., A survival analysis of the liver-first reversed management of advanced simultaneous colorectal liver metastases: a LiverMetSurvey-based study. Ann Surg, 2012. 256(5): p. 772–8; discussion 778–9.
Cardona, K., et al., Detailed pathologic characteristics of the primary colorectal tumor independently predict outcome after hepatectomy for metastases. Ann Surg Oncol, 2013. 20(1): p. 148–54.
Pawlik, T.M. and D. Cosgrove, The role of peri-operative chemotherapy for resectable colorectal liver metastasis: what does the evidence support? J Gastrointest Surg, 2011. 15(3): p. 410–5.
Bischof, D.A., et al., Surgical management of disappearing colorectal liver metastases. Br J Surg, 2013. 100(11): p. 1414–20.
Mayo, S.C. and T.M. Pawlik, Current management of colorectal hepatic metastasis. Expert Rev Gastroenterol Hepatol, 2009. 3(2): p. 131–44.
Mayo, S.C., et al., Surgical management of patients with synchronous colorectal liver metastasis: a multicenter international analysis. J Am Coll Surg, 2013. 216(4): p. 707–16; discussion 716–8.
de Gramont, A., et al., Randomized trial comparing monthly low-dose leucovorin and fluorouracil bolus with bimonthly high-dose leucovorin and fluorouracil bolus plus continuous infusion for advanced colorectal cancer: a French intergroup study. J Clin Oncol, 1997. 15(2): p. 808–15.
Douillard, J.Y., et al., Irinotecan combined with fluorouracil compared with fluorouracil alone as first-line treatment for metastatic colorectal cancer: a multicentre randomised trial. Lancet, 2000. 355(9209): p. 1041–7.
Tournigand, C., et al., FOLFIRI followed by FOLFOX6 or the reverse sequence in advanced colorectal cancer: a randomized GERCOR study. J Clin Oncol, 2004. 22(2): p. 229–37.
Hochster, H.S., et al., Safety and efficacy of oxaliplatin and fluoropyrimidine regimens with or without bevacizumab as first-line treatment of metastatic colorectal cancer: results of the TREE Study. J Clin Oncol, 2008. 26(21): p. 3523–9.
Fuchs, C.S., et al., Randomized, controlled trial of irinotecan plus infusional, bolus, or oral fluoropyrimidines in first-line treatment of metastatic colorectal cancer: results from the BICC-C Study. J Clin Oncol, 2007. 25(30): p. 4779–86.
Vigano, L., et al., Progression while receiving preoperative chemotherapy should not be an absolute contraindication to liver resection for colorectal metastases. Ann Surg Oncol, 2012. 19(9): p. 2786–96.
Shindoh, J., et al., Optimal morphologic response to preoperative chemotherapy: an alternate outcome end point before resection of hepatic colorectal metastases. J Clin Oncol, 2012. 30(36): p. 4566–72.
Adam, R., et al., Rescue surgery for unresectable colorectal liver metastases downstaged by chemotherapy: a model to predict long-term survival. Ann Surg, 2004. 240(4): p. 644–57; discussion 657–8.
Benoist, S., et al., Complete response of colorectal liver metastases after chemotherapy: does it mean cure? J Clin Oncol, 2006. 24(24): p. 3939–45.
Elias, D., et al., Outcome of posthepatectomy-missing colorectal liver metastases after complete response to chemotherapy: impact of adjuvant intra-arterial hepatic oxaliplatin. Ann Surg Oncol, 2007. 14(11): p. 3188–94.
Elias, D., et al., Evolution of missing colorectal liver metastases following inductive chemotherapy and hepatectomy. J Surg Oncol, 2004. 86(1): p. 4–9.
Auer, R.C., et al., Predictors of a true complete response among disappearing liver metastases from colorectal cancer after chemotherapy. Cancer, 2010. 116(6): p. 1502–9.
Cucchetti, A., et al., Cost-effectiveness of hepatic resection versus percutaneous radiofrequency ablation for early hepatocellular carcinoma. J Hepatol, 2013. 59(2): p. 300–7.
Nordlinger, B., et al., Perioperative FOLFOX4 chemotherapy and surgery versus surgery alone for resectable liver metastases from colorectal cancer (EORTC 40983): long-term results of a randomised, controlled, phase 3 trial. Lancet Oncol, 2013. 14(12): p. 1208–15.
Stinnett, A.A. and J. Mullahy, Net health benefits: a new framework for the analysis of uncertainty in cost-effectiveness analysis. Med Decis Making, 1998. 18(2 Suppl): p. S68-80.
Grosse, S.D., Assessing cost-effectiveness in healthcare: history of the $50,000 per QALY threshold. Expert Rev Pharmacoecon Outcomes Res, 2008. 8(2): p. 165–78.
Tanaka, K., et al., Importance of complete pathologic response to prehepatectomy chemotherapy in treating colorectal cancer metastases. Ann Surg, 2009. 250(6): p. 935–42.
Kanas, G.P., et al., Survival after liver resection in metastatic colorectal cancer: review and meta-analysis of prognostic factors. Clin Epidemiol, 2012. 4: p. 283–301.
Khajanchee, Y.S., et al., Hepatic resection vs minimally invasive radiofrequency ablation for the treatment of colorectal liver metastases: a Markov analysis. Arch Surg, 2011. 146(12): p. 1416–23.
Hyder, O., et al., A Nomogram to Predict Long-term Survival After Resection for Intrahepatic Cholangiocarcinoma: An Eastern and Western Experience. JAMA Surg, 2014.
Spolverato, G., et al., The Relative Net Health Benefit of Liver Resection, Ablation, and Transplantation for Early Hepatocellular Carcinoma. World J Surg, 2015.
Lim, K.C., et al., Cost-effectiveness analysis of liver resection versus transplantation for early hepatocellular carcinoma within the Milan criteria. Hepatology, 2014.
Siegel, J.E., et al., Recommendations for reporting cost-effectiveness analyses. Panel on Cost-Effectiveness in Health and Medicine. JAMA, 1996. 276(16): p. 1339–41.
van Vledder, M.G., et al., Disappearing colorectal liver metastases after chemotherapy: should we be concerned? J Gastrointest Surg, 2010. 14(11): p. 1691–700.
Grant/Support
None
Author information
Authors and Affiliations
Corresponding author
Additional information
Gaya Spolverato and Alessandro Vitale contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
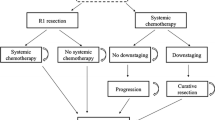
Supplementary Figure 1
The event pathway: decision tree and states of health (GIF 117 kb)
Supplementary Figure 2
Variation of Survival Benefit according to patient age ≤60 (A) or >60 (B) years and rate of true complete pathological response. (GIF 128 kb)
(GIF 128 kb)
Rights and permissions
About this article
Cite this article
Spolverato, G., Vitale, A., Ejaz, A. et al. Hepatic Resection for Disappearing Liver Metastasis: a Cost-Utility Analysis. J Gastrointest Surg 19, 1668–1675 (2015). https://doi.org/10.1007/s11605-015-2873-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11605-015-2873-5