Abstract
Background
Crystalloid fluid resuscitation after hemorrhagic shock (HS) that restores/maintains central hemodynamics often culminates in multi-system organ failure and death due to persistent/progressive splanchnic hypoperfusion and end-organ damage. Adjunctive direct peritoneal resuscitation (DPR) using peritoneal dialysis solution reverses HS-induced splanchnic hypoperfusion and improves survival. We examined HS-mediated hepatic perfusion (galactose clearance), tissue injury (histopathology), and dysfunction (liver enzymes).
Methods
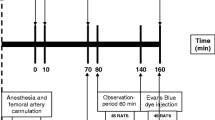
Anesthetized rats were randomly assigned (n = 8/group): (1) sham (no HS); (2) HS (40% mean arterial pressure for 60 min) plus conventional i.v. fluid resuscitation (CR; shed blood + 2 volumes saline); (3) HS + CR + 30 mL intraperitoneal (IP) DPR; or (4) HS + CR + 30 mL IP saline. Hemodynamics and hepatic blood flow were measured for 2 h after CR completion. In duplicate animals, liver and splanchnic tissues were harvested for histopathology (blinded, graded), hepatocellular function (liver enzymes), and tissue edema (wet–dry ratio).
Results
Group 2 decreased liver blood flow, caused liver injuries (focal to submassive necrosis, zones 2 and 3) and tissue edema, and elevated liver enzymes (alanine aminotransferase (ALT), 149 ± 28 μg/mL and aspartate aminotransferase (AST), 234 ± 24 μg/mL; p < 0.05) compared to group 1 (73 ± 9 and 119 ± 10 μg/mL, respectively). Minimal/no injuries were observed in group 3; enzymes were normalized (ALT 89 ± 9 μg/mL and AST 150 ± 17 μg/mL), and tissue edema was similar to sham.
Conclusions
CR from HS restored and maintained central hemodynamics but did not restore or maintain liver perfusion and was associated with significant hepatocellular injury and dysfunction. DPR added to conventional resuscitation (blood and crystalloid) restored and maintained liver perfusion, prevented hepatocellular injury and edema, and preserved liver function.





Similar content being viewed by others
References
Hassoun HT, Kone BC, Mercer DW, Moody FG, Weisbrodt NW, Moore FA. Post-injury multiple organ failure: the role of the gut. Shock 2001;15:1–10.
Fruchterman TM, Spain DA, Wilson MA, Harris PD, Garrison RN. Selective microvascular endothelial cell dysfunction in the small intestine following resuscitated hemorrhagic shock. Shock 1998;10:417–422. doi:10.1097/00024382-199812000-00007.
Wang P, Hauptman JG, Chaudry IH. Hemorrhage produces depression in microvascular blood flow which persists despite fluid resuscitation. Circ Shock 1990;32:307–318.
Zakaria ER, Spain DA, Harris PD, Garrison RN. Resuscitation regimens for hemorrhagic shock must contain blood. Shock 2002;18:567–573. doi:10.1097/00024382-200212000-00014.
Deitch EA, Xu D, Franko L, Ayala A, Chaudry IH. Evidence favoring the role of the gut as a cytokine-generating organ in rats subjected to hemorrhagic shock. Shock 1994;1:141–145.
Wang P, Hauptman JG, Chaudry IH. Hepatocellular dysfunction occurs early after hemorrhage and persists despite fluid resuscitation. J Surg Res 1990;48:464–470.
Wang P, Ba ZF, Chaudry IH. Endothelial cell dysfunction occurs very early following trauma-hemorrhage and persists despite fluid resuscitation. Am J Physiol 1993;265:H973–H979.
Zakaria ER, Garrison RN, Spain DA, Harris PD. Impairment of endothelium-dependent dilation response after resuscitation from hemorrhagic shock involved postreceptor mechanisms. Shock 2004;21:175–181. doi:10.1097/00024382-200402000-00014.
Zweifach BW. Mechanisms of blood flow and fluid exchange in microvessels: hemorrhagic hypotension model. Anesthesiology 1974;41:157–168. doi:10.1097/00000542-197408000-00006.
Zakaria ER, Garrison RN, Spain DA, Matheson PJ, Harris PD, Richardson JD. Intraperitoneal resuscitation improves intestinal blood flow following hemorrhagic shock. Ann Surg 2003;237:704–711. doi:10.1097/00000658-200305000-00013.
Zakaria ER, Garrison RN, Kawabe T, Harris PD. Direct peritoneal resuscitation from hemorrhagic shock: effect of time delay in therapy initiation. J Trauma 2005;58:499–506. doi:10.1097/01.TA.0000152892.24841.54.
Garrison RN, Conn AA, Harris PD, Zakaria ER. Direct peritoneal resuscitation as adjunct to conventional resuscitation from hemorrhagic shock: a better outcome. Surgery 2004;136:900–908. doi:10.1016/j.surg.2004.06.027.
Zakaria ER, Hurt RT, Matheson PJ, Garrison RN. A novel method of peritoneal resuscitation improves organ perfusion after hemorrhagic shock. Am J Surg 2003;186:443–448. doi:10.1016/j.amjsurg.2003.07.006.
Chojkier M, Fierer J. d-Galactosamine hepatotoxicity is associated with endotoxin sensitivity and mediated by lymphoreticular cells in mice. Gastroenterology 1985;88:115–121.
Shiratori Y, Kawase T, Shiina S, Okano K, Sugimoto T, Teraoka H, Matano S, Matsumoto K, Kamii K. Modulation of hepatotoxicity by macrophages in the liver. Hepatology 1988;8:815–821. doi:10.1002/hep.1840080420.
Wang P, Ba ZF, Biondo A, Chaudry I. Liver endothelial cell dysfunction occurs early following hemorrhagic shock and persists despite crystalloid resuscitation. J Surg Res 1996;63:241–247. doi:10.1006/jsre.1996.0255.
Bauer M, Marzi I, Ziegenfuss T, Seeck G, Buhren V, Larsen R. Comparative effects of crystalloid and small volume hypertonic hyperoncotic fluid resuscitation on hepatic microcirculation after hemorrhagic shock. Circ Shock 1993;40:187–193.
Corso CO, Okamoto S, Leiderer R, Messmer K. Resuscitation with hypertonic saline dextran reduces endothelial cell swelling and improves hepatic microvascular perfusion and function after hemorrhagic shock. J Surg Res 1998;80:210–220. doi:10.1006/jsre.1998.5426.
Dart RC, Liebler DC, Sipes IG. Hepatic injury and lipid peroxidation during hemorrhagic shock and resuscitation. Life Sci 1993;53:1685–1690. doi:10.1016/0024-3205(93)90205-H.
Holliday RL, Illner HP, Shires GT. Liver cell membrane alterations during hemorrhagic shock in the rat. J Surg Res 1981;31:506–515. doi:10.1016/0022-4804(81)90189-X.
Jarrar D, Wang P, Cioffi WG, Bland KI, Chaudry IH. Critical role of oxygen radicals in the initiation of hepatic depression after trauma hemorrhage. J Trauma 2000;49:879–885. doi:10.1097/00005373-200011000-00015.
Flynn WJ, Cryer HG, Garrison RN. Pentoxifylline restores intestinal microvascular blood flow during resuscitated hemorrhagic shock. Surgery 1991;110:350–356.
Flynn WJ, Cryer HG, Garrison RN. Pentoxifylline but not saralasin restores hepatic blood flow after resuscitation from hemorrhagic shock. J Surg Res 1991;50:616–621. doi:10.1016/0022-4804(91)90051-M.
Fruchterman TM, Spain DA, Wilson MA, Harris PD, Garrison RN. Selective microvascular endothelial cell dysfunction in the small intestine following resuscitated hemorrhagic shock. Shock 1998;10:417–422. doi:10.1097/00024382-199812000-00007.
Wang P, Hauptman JG, Chaudry IH. Hemorrhage produces depression in microvascular blood flow which persists despite fluid resuscitation. Circ Shock 1990;32:307–318.
Zakaria ER, Spain DA, Harris PD, Garrison RN. Resuscitation regimens for hemorrhagic shock must contain blood. Shock 2002;18:567–573. doi:10.1097/00024382-200212000-00014.
Flynn WJ Jr, Gosche JR, Garrison RN. Intestinal blood flow is restored with glutamine or glucose suffusion after hemorrhage. J Surg Res 1992;52:499–504. doi:10.1016/0022-4804(92)90318-T.
Flynn WJ Jr, Pilati D, Hoover EL. Effect of allopurinol on venous endothelial dysfunction after resuscitated hemorrhagic shock. Int J Surg Investig 1999;1:11–18.
Fruchterman TM, Spain DA, Wilson MA, Harris PD, Garrison RN. Complement inhibition prevents gut ischemia and endothelial cell dysfunction after hemorrhage/resuscitation. Surgery 1998;124:782–791. doi:10.1067/msy.1998.91489.
Wang P, Ba ZF, Chaudry IH. Endothelial cell dysfunction occurs after hemorrhage in nonheparinized but not in preheparinized models. J Surg Res 1993;54:499–506. doi:10.1006/jsre.1993.1077.
Watkins JM, Spain DA, Krysztopik RJ, Downard PJ, Wilson MA, Garrison RN. Heparan preserves intestinal perfusion after hemorrhage and resuscitation. J Surg Res 1996;66:154–158. doi:10.1006/jsre.1996.0388.
Zakaria ER, Li N, Garrison RN. Mechanisms of direct peritoneal resuscitation-mediated splanchnic hyperperfusion following hemorrhagic shock. Shock 2007;27:436–442. doi:10.1097/01.shk.0000245017.86117.4e.
Zakaria ER, Li N, Matheson PJ, Garrison RN. Cellular edema regulates tissue capillary perfusion after hemorrhage resuscitation. Surgery 2007;142:487–496. doi:10.1016/j.surg.2007.08.007.
Zakaria ER, Campbell JE, Peyton JC, Garrison RN. Postresuscitation tissue neutrophil infiltration is time-dependent and organ-specific. J Surg Res 2007;143:119–125. doi:10.1016/j.jss.2007.04.008.
Mazzoni MC, Intaglietta M, Cragoe EJ Jr, Arfors KE. Amiloride-sensitive Na+ pathways in capillary endothelial cell swelling during hemorrhagic shock. J Appl Physiol 1992;73:1467–1473.
Chang JX, Chen S, Ma LP, Jiang LY, Chen JW, Chang RM, Wen LQ, Wu W, Jiang ZP, Huang ZT. Functional and morphological changes of the gut barrier during the restitution process after hemorrhagic shock. World J Gastroenterol 2005;11:5485–5491.
Jaeschke H. Mechanisms of Liver Injury. II. Mechanisms of neutrophil-induced liver cell injury during hepatic ischemia-reperfusion and other acute inflammatory conditions. Am J Physiol Gastrointest Liver Physiol 2006;290:G1083–G1088. doi:10.1152/ajpgi.00568.2005.
Chang TW. Improvement of survival from hemorrhagic shock by enterectomy in rats: finding to implicate the role of the gut for irreversibility of hemorrhagic shock. J Trauma 1997;42:223–230. doi:10.1097/00005373-199702000-00007.
Martin C, Boisson C, Haccoun M, Thomachot L, Mege JL. Patterns of cytokine evolution (tumor necrosis factor-alpha and interleukin-6) after septic shock, hemorrhagic shock, and severe trauma. Crit Care Med 1997;25:1813–1819. doi:10.1097/00003246-199711000-00018.
Matsutani T, Kang SC, Miyashita M, Sasajima K, Choudhry MA, Bland KI, Chaudry IH. Liver cytokine production and ICAM-1 expression following bone fracture, tissue trauma, and hemorrhage in middle-aged mice. Am J Physiol Gastrointest Liver Physiol 2007;292:G268–G274. doi:10.1152/ajpgi.00313.2006.
Roumen RM, Hendriks T, Van der Ven-Jongekrijg J, Nieuwenhuijzen GA, Sauerwein RW, van der Meer JW, Goris RJ. Cytokine patterns in patients after major vascular surgery, hemorrhagic shock, and severe blunt trauma. Relation with subsequent adult respiratory distress syndrome and multiple organ failure. Ann Surg 1993;218:769–776. doi:10.1097/00000658-199312000-00011.
Author information
Authors and Affiliations
Corresponding author
Additional information
No conflicts of interest exist.
Grant support: This project was supported by a VA Merit Review grant and by NIH research Grant # 5R01 HL076160-03, funded by the National Heart, Lung, and Blood Institute and the United States Army Medical Resources and Material Command.
Rights and permissions
About this article
Cite this article
Hurt, R.T., Zakaria, E.R., Matheson, P.J. et al. Hemorrhage-Induced Hepatic Injury and Hypoperfusion can be Prevented by Direct Peritoneal Resuscitation. J Gastrointest Surg 13, 587–594 (2009). https://doi.org/10.1007/s11605-008-0796-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11605-008-0796-0