Summary
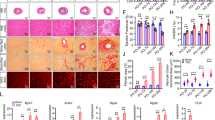
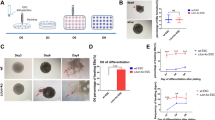
Ventricular septum defects (VSDs) are common types of congenital heart diseases caused by developmental defect; they contribute to 25%-30% of all adult congenital heart diseases. The peroxisome proliferator-activated receptor gamma (PPAR-γ) is widely expressed in mammalian tissues and in the immune system, regulating cell differentiation and immune and inflammatory responses. The PPAR-γ gene has recently been found crucial for heart development, but the mechanism of action is not clear. This study aims to investigate the effects of the PPAR-γ gene in the myocardium on the development of ventricular septation. In this study, we applied Cre-loxP recombination enzyme (CRE) technology to downregulate the expression of the PPAR-γ gene in different cardiac tissues, RT-PCR to examine the expression of the c-fos and TGF-β1 genes, and histology staining to check the defect of embryonic heart at embryonic day 14.5 (E14.5). We found that the downregulation of the PPAR-γ gene resulted in a ventricular membranous septation defect of the embryonic heart at E14.5. Furthermore, only conversion of a Tnt:Cre, but not Mef2c:Cre, Tie2:Cre, or Wnt:Cre PPAR-γ floxed allele to a null allele resulted in VSD. PPAR-γTnt-Cre/+ embryos showed increases in atrioventricular (AV)-cushion cells and the expression of c-fos gene but no change in the expression of TGF-β1 at E10.5. Our study demonstrates PPAR-γ in the myocardium is required for ventricular septation through regulation of AV-cushion cell proliferation by a Tnt/c-fos signal.
Similar content being viewed by others
References
Bruneau BG. The developmental genetics of congenital heart disease. Nature, 2008,451 (7181):943–948
Qin YW. Chinese expert consensus on interventional therapy for common congenital heart disease II. Inter-ventional therapy for VSD. J Intervent Rasiaol, 2011, 20(2):3–9
Anderson RH, Webb S, Brown NA, et al. Development of the heart. Heart, 2003,89 (8):955–957
Srivastava D, Olson EN. A genetic blueprint for cardiac development. Nature, 2000,407 (6801):221–226
Krishnaswami A, Ravikumar S, Lewis JM. Thiazoli-dinediones: a 2010 perspective. Perm J, 2010,14(3):64–72
Barak Y, Nelson MC, Ong ES, et al. PPARγ is required for placental, cardiac, and adipose tissue development. Molecular Cell, 1999,4 (4):585–595
Duan SZ, Ivashchenko CY, Russell MW, et al. Cardiomyocyte-specific knockout and agonist of pero-xisome proliferator-activated receptor-gamma both induce cardiac hypertrophy in mice. Circ Res, 2005,97(4):372–379
Ding G, Fu M, Qin Q, et al. Cardiac peroxisome proliferator-activated receptor δ is essential in protecting cardiomyocytes from oxidative damage. Cardiovasc Res, 2007,76 (2):269–279
Goddeeris MM, Rho S, Petiet A, et al. Intracardiac septation requires hedgehog-dependent cellular contri-butions from outside the heart. Development, 2008,135 (10):1887–1895
Jiao K, Kulessa H, Tompkins K, et al. An essential role of Bmp4 in the atrioventricular septation of the mouse heart. Genes Dev, 2003,17 (19):2362–2367
Dodou E. Mef2c is a direct transcriptional target of ISL1 and GATA factors in the anterior heart field during mouse embryonic development. Development, 2004,131 (16):3931–3942
Szeder V, Grim M, Halata Z, et al. Neural crest origin of mammalian Merkel cells. Dev Biol, 2003,253(2):258–263
Lakso M, Pichel JG, Gorman JR. Efficient in vivo manipulation of mouse genomic sequences at the zygote stage. Proc Natl Acad Sci U S A, 1996,93(12):5860–5865
Lin LC, Hsu SL, Wu CL, et al. TGFβ can stimulate the p38/β-catenin/PPARγ signaling pathway to promote the EMT, invasion and migration of non-small cell lung cancer (H460 cells). Clin Exp Metastasis. 2014,31(8): 881–895.
Lim S, Jin CJ, Kim M, et al. PPARgamma gene transfer sustains apoptosis, inhibits vascular smooth muscle cell proliferation, and reduces neointima formation after balloon injury in rats. Arterioscler Thromb Vasc Biol, 2006,26 (4):808–813
Hou J, Wang L, Hou J, et al. Peroxisome proliferator-activated receptor gamma promotes mesenchymal stem cells to express connexin 43 via the inhibition of TGF-β1/Smads signaling in a rat model of myocardial infarction. Stem Cell Rev, 2015,11 (23):885–899
Gittenberger-De Groot AC, Calkoen EE, Poelmann RE, et al. Morphogenesis and molecular considerations on congenital cardiac septal defects. Ann Med, 2014,46(8): 640–652
Wang Q, Reiter RS, Huang QQ, et al. Comparative studies on the expression patterns of three troponin T genes during mouse development. Anat Rec, 2001,263(1): 72–84
Guo Y, Kühl SJ, Pfister AS, et al. Comparative analysis reveals distinct and overlapping functions of Mef2c and Mef2d during cardiogenesis in Xenopus laevis. PLoS One, 2014,9(1):e87294
Kim YJ, Park KJ, Song JK, et al. The PPARγ agonist protects cardiomyocytes from oxidative stress and apop-tosis via thioredoxin overexpression. Biosci Biotechnol Biochem, 2012,76 (12):2181–2187
Mao X, Xing H, Mao A, et al. Netrin-1 attenuates cardiac ischemia reperfusion injury and generates alternatively activated macrophages. Inflammation, 2014,37(2):573–580
Yan L, Zhang JD, Wang B, et al. Quercetin inhibits left ventricular hypertrophy in spontaneously hypertensive rats and inhibits angiotensin II-induced H9C2 cells hypertrophy by enhancing PPAR-γ expression and suppressing AP-1 activity. PLoS One, 2013,8(9):e72548
Maejima Y, Okada H, Haraguchi G, et al. Telmisartan, a unique ARB, improves left ventricular remodeling of infarcted heart by activating PPAR gamma. Lab Invest, 2011,91 (6):932–944
Dong X, Zhang C, Ma, Wen H. High concentrations of mast cell chymase facilitate the transduction of the transforming growth factor- beta1. Smads signaling pathway in skin fibroblasts. Exp Ther Med, 2015,9(3):955–960
Doganci S, Yildirim V, Bolcal C, et al. OP-017 Sodium Nitrite and Cardioprotective Effect in Pig Regional Myocardial Ischemia-Reperfusion Injury Model. Am J Cardiol, 2015,115(S1):S6–S7
Gong K, Xing D, Li P, et al. Hypoxia induces downregulation of PPAR-r in isolated pulmonary arterial smooth muscle cells and in rat lung via transforming growth factor beta signaling. Am J Physiol Lung Cell Mol Physiol, 2011,301(6):L899–L977
Acknowledgments
The authors thank Dr. Linglin Xie from the Department of Pathology, University of North Dakota for her free supply of mice.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare no conflict of interest.
Additional information
This project was supported by Natural Science Foundation of Hubei Province, China (No. 20152015CKB748) and Independent Innovation Foundation of Huazhong University of Science and Technology, China (No. 0118540198).
Rights and permissions
About this article
Cite this article
Zhou, L., Wang, Zz., Xiao, Zc. et al. Effects of PPAR-γ in the Myocardium on the Development of Ventricular Septation. CURR MED SCI 40, 313–319 (2020). https://doi.org/10.1007/s11596-020-2184-2
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-020-2184-2